Fuel Cell Formulary
... 1.5.1 Kinetic of the H2 /O2 electrochemical device The kinetics of electrochemical reaction is relevant as soon as the electric circuit of any electrochemical device is closed and it starts providing electric power. As soon as electrons and ions start moving we get voltage losses (overvoltage). This ...
... 1.5.1 Kinetic of the H2 /O2 electrochemical device The kinetics of electrochemical reaction is relevant as soon as the electric circuit of any electrochemical device is closed and it starts providing electric power. As soon as electrons and ions start moving we get voltage losses (overvoltage). This ...
CH1 INTRODUCTION AND BASIC CONCEPTS
... tank of a car is about 50 L. Therefore, a car needs to be refueled once every 10 days. Also, the density of gasoline ranges from 0.68 to 0.78 kg/L, and its lower heating value is about 44,000 kJ/kg (that is, 44,000 Kj of heat is released when 1 kg of gasoline is completely burned). Suppose all the p ...
... tank of a car is about 50 L. Therefore, a car needs to be refueled once every 10 days. Also, the density of gasoline ranges from 0.68 to 0.78 kg/L, and its lower heating value is about 44,000 kJ/kg (that is, 44,000 Kj of heat is released when 1 kg of gasoline is completely burned). Suppose all the p ...
Academic Earth/Space Science Date: March 19, 2014 ET Topic: U
... Describe how changes in concentration of minor components (e.g., O2, CO2, ozone, dust, pollution) in Earth's atmosphere are linked to climate change. S11.D.2.1.2 -- Important Compare the transmission, reflection, absorption, and radiation of solar energy to and by the Earth's surface under different ...
... Describe how changes in concentration of minor components (e.g., O2, CO2, ozone, dust, pollution) in Earth's atmosphere are linked to climate change. S11.D.2.1.2 -- Important Compare the transmission, reflection, absorption, and radiation of solar energy to and by the Earth's surface under different ...
variable specific heat theory
... specific heat depends on temperature changes. Specific heat of real gases also to some extent depends upon the pressure. For real gases the specific heats and hence y are not constant with temperature as in the case of perfect gases (y = 1-67 for monatomic, 7 = 1-4 for diatomic, and y = 1-3 for poly ...
... specific heat depends on temperature changes. Specific heat of real gases also to some extent depends upon the pressure. For real gases the specific heats and hence y are not constant with temperature as in the case of perfect gases (y = 1-67 for monatomic, 7 = 1-4 for diatomic, and y = 1-3 for poly ...
Page 1 of 3 Curriculum: Susquenita Curriculum PENNSYLVANIA
... Describe how changes in concentration of minor components (e.g., O2, CO2, ozone, dust, pollution) in Earth's atmosphere are linked to climate change. S11.D.2.1.2 -- Important Compare the transmission, reflection, absorption, and radiation of solar energy to and by the Earth's surface under different ...
... Describe how changes in concentration of minor components (e.g., O2, CO2, ozone, dust, pollution) in Earth's atmosphere are linked to climate change. S11.D.2.1.2 -- Important Compare the transmission, reflection, absorption, and radiation of solar energy to and by the Earth's surface under different ...
Activity 4a Part 1 - Mr. Nap`s Excellent Earth Science Page
... temperatures by wind motion is referred to as warm air advection. Conversely, the transport of air from a region of relatively low temperatures to a region of relatively high temperatures by wind motion is called cold air advection. Although on a global and annual average basis, there is a net polew ...
... temperatures by wind motion is referred to as warm air advection. Conversely, the transport of air from a region of relatively low temperatures to a region of relatively high temperatures by wind motion is called cold air advection. Although on a global and annual average basis, there is a net polew ...
Changes of State
... Le Chatelier's principle can be used to predict how the liquid-vapor equilibrium in the system just discussed changes when the temperature of the system is increased from 20 ºC to 50 ºC. Le Chatelier's principle can be used to predict it. how does the liquid-vapor equilibrium (in the system) (just d ...
... Le Chatelier's principle can be used to predict how the liquid-vapor equilibrium in the system just discussed changes when the temperature of the system is increased from 20 ºC to 50 ºC. Le Chatelier's principle can be used to predict it. how does the liquid-vapor equilibrium (in the system) (just d ...
Overview of dosimetry - National Physical Laboratory
... earlier than water calorimetry. NPL’s absorbed dose standards are at present based on graphite calorimeters. In graphite calorimetry, lattice impurities and chemical reactions with dissolved oxygen have been proposed as mechanisms for a potential heat defect, but they are in general assumed to be ne ...
... earlier than water calorimetry. NPL’s absorbed dose standards are at present based on graphite calorimeters. In graphite calorimetry, lattice impurities and chemical reactions with dissolved oxygen have been proposed as mechanisms for a potential heat defect, but they are in general assumed to be ne ...
DIFFUSION OF WATER MOLECULES IN HYDRATES AND NMR
... average over all the different hi in accordance with the general formula (5). It is important to note that (h) is directly connected with the aggregate of ei and Pi, which can be obtained from the NMR spectra in the temperature region below the transition temperature. For a sufficiently low temperat ...
... average over all the different hi in accordance with the general formula (5). It is important to note that (h) is directly connected with the aggregate of ei and Pi, which can be obtained from the NMR spectra in the temperature region below the transition temperature. For a sufficiently low temperat ...
Chapter 4: Energy Analysis of Closed Systems
... One kilogram of water is contained in a piston-cylinder device at 100 C. The piston rests on lower stops such that the volume occupied by the water is 0.835 m3. The cylinder is fitted with an upper set of stops. When the piston rests against the upper stops, the volume enclosed by the piston-cylind ...
... One kilogram of water is contained in a piston-cylinder device at 100 C. The piston rests on lower stops such that the volume occupied by the water is 0.835 m3. The cylinder is fitted with an upper set of stops. When the piston rests against the upper stops, the volume enclosed by the piston-cylind ...
Fundamentals of Thermodynamics Applied to Thermal
... point as seen in Fig. 1. Therefore, above the critical pressure and temperature there will be no liquid–vapor phase change. The process illustrated in Fig. 1 takes place at a constant pressure, known as isobaric, which is imposed on the system by the piston weight plus local atmospheric pressure. Ot ...
... point as seen in Fig. 1. Therefore, above the critical pressure and temperature there will be no liquid–vapor phase change. The process illustrated in Fig. 1 takes place at a constant pressure, known as isobaric, which is imposed on the system by the piston weight plus local atmospheric pressure. Ot ...
Topic # 3
... We use Dalton’s Law of partial pressures: Total pressure = sum of individual pressures (as long as the gases do not interact chemically!) ...
... We use Dalton’s Law of partial pressures: Total pressure = sum of individual pressures (as long as the gases do not interact chemically!) ...
Physics Final Exam Study Guide 3
... 8. Consider an air-filled balloon, weighted with a stone, that just barely floats in water. When the balloon is pushed beneath the surface, say about a meter deep, and then released, it will _____. a. stay where it is b. sink c. float back to the surface 9. Which has the larger buoyant force, a ball ...
... 8. Consider an air-filled balloon, weighted with a stone, that just barely floats in water. When the balloon is pushed beneath the surface, say about a meter deep, and then released, it will _____. a. stay where it is b. sink c. float back to the surface 9. Which has the larger buoyant force, a ball ...
Tables from chapter 17
... The meter is the length of the path traveled by light in vacuum during a time interval of 1/299 792 458 of a second. The kilogram is equal to the mass of the international prototype of the kilogram. The second is the duration of 9 192 631 770 periods of the radiation corresponding to the transition ...
... The meter is the length of the path traveled by light in vacuum during a time interval of 1/299 792 458 of a second. The kilogram is equal to the mass of the international prototype of the kilogram. The second is the duration of 9 192 631 770 periods of the radiation corresponding to the transition ...
PDF only - at www.arxiv.org.
... in the universe.” (SLAC Press Release July 23, 2002). Theorists found it hard to see why CP symmetry should be broken at all and even harder to understand why any imperfection should be so small[2]. “Such rare decays are very challenging to identify, but over the past few years we have learned to cl ...
... in the universe.” (SLAC Press Release July 23, 2002). Theorists found it hard to see why CP symmetry should be broken at all and even harder to understand why any imperfection should be so small[2]. “Such rare decays are very challenging to identify, but over the past few years we have learned to cl ...
Heat Engine 0.5
... 3.25 cm). The second term, “88.5”, must be replaced with the volume calculated in the table above. Enter the number you calculated and click on OK to store the correct equation. 7. Lift the platform so that the position is near 50 mm. Press the rubber stopper into the aluminum can. Make sure that th ...
... 3.25 cm). The second term, “88.5”, must be replaced with the volume calculated in the table above. Enter the number you calculated and click on OK to store the correct equation. 7. Lift the platform so that the position is near 50 mm. Press the rubber stopper into the aluminum can. Make sure that th ...
basic thermodynamics
... the law of degradation of energy and the energy is said to run down hill. ...
... the law of degradation of energy and the energy is said to run down hill. ...
Heat and Properties of Matter
... A good definition of a phase of a system is a region in the parameter space of the system’s thermodynamic variables in which the free energy F is a continuous well-behaved function of external conditions such as temperature or pressure. At certain conditions, the stable state is the one which minimi ...
... A good definition of a phase of a system is a region in the parameter space of the system’s thermodynamic variables in which the free energy F is a continuous well-behaved function of external conditions such as temperature or pressure. At certain conditions, the stable state is the one which minimi ...
Pure Substances
... cylinder contains one kg of ice at –200C and one bar. When heat is transferred to the ice the pressure remains constant the specific volume increases slightly and the temperature increases until it reaches 0 0C, at which point the ice melts and temperature remains constant. This state is called satu ...
... cylinder contains one kg of ice at –200C and one bar. When heat is transferred to the ice the pressure remains constant the specific volume increases slightly and the temperature increases until it reaches 0 0C, at which point the ice melts and temperature remains constant. This state is called satu ...
IV. Adiabatic Processes
... If a material undergoes a change in its physical state (e.g., its pressure, volume, or temperature) without any heat being added to it or withdrawn from it, the change is said to be adiabatic. Suppose that the initial state of a material is represented by the point A on the thermodynamic diagram bel ...
... If a material undergoes a change in its physical state (e.g., its pressure, volume, or temperature) without any heat being added to it or withdrawn from it, the change is said to be adiabatic. Suppose that the initial state of a material is represented by the point A on the thermodynamic diagram bel ...
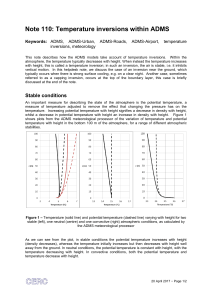
Note 110: Temperature inversions within ADMS
... During very low wind stable conditions in hilly terrain, horizontal gradients in density can cause katabatic (downslope) winds, which may influence the background flow in deep valleys. These effects are not specifically accounted for in ADMS; please contact CERC to discuss specific cases. ...
... During very low wind stable conditions in hilly terrain, horizontal gradients in density can cause katabatic (downslope) winds, which may influence the background flow in deep valleys. These effects are not specifically accounted for in ADMS; please contact CERC to discuss specific cases. ...
PDF only - at www.arxiv.org.
... pumped to a vacuum pressure P≤10-8 Pa for 20 hours. The aim of the operation is to remove impurities such as oxygen adsorbed on the inner walls of the container. We measured the total capacitance C = 52.4pF and C’’ = 1632pF, the vacuum capacitance is C’0 =54.1 pF according to (6). The mass of a sodi ...
... pumped to a vacuum pressure P≤10-8 Pa for 20 hours. The aim of the operation is to remove impurities such as oxygen adsorbed on the inner walls of the container. We measured the total capacitance C = 52.4pF and C’’ = 1632pF, the vacuum capacitance is C’0 =54.1 pF according to (6). The mass of a sodi ...
Physical Vapor Deposition
... PVD is used for metals. Dielectrics can be deposited using specialized equipment ...
... PVD is used for metals. Dielectrics can be deposited using specialized equipment ...
Chapter 12
... phenomenon that the freezing point of a liquid,is depressed when another compound is added, meaning that a solution has a lower freezing point than a pure solvent. This happens whenever a solute is added to a pure solvent, such as water. This phenomenon may be observed in sea water, which due to i ...
... phenomenon that the freezing point of a liquid,is depressed when another compound is added, meaning that a solution has a lower freezing point than a pure solvent. This happens whenever a solute is added to a pure solvent, such as water. This phenomenon may be observed in sea water, which due to i ...
Document
... However, this now underestimates the recombination, because we have not considered the more complex shape of the charge densities – space charge alteration of the electric field (see blue lines): -- Note that we specify charge density at the electrodes by the potential: ...
... However, this now underestimates the recombination, because we have not considered the more complex shape of the charge densities – space charge alteration of the electric field (see blue lines): -- Note that we specify charge density at the electrodes by the potential: ...
Humidity

Humidity is the amount of water vapor in the air. Water vapor is the gaseous state of water and is invisible. Humidity indicates the likelihood of precipitation, dew, or fog. Higher humidity reduces the effectiveness of sweating in cooling the body by reducing the rate of evaporation of moisture from the skin. This effect is calculated in a heat index table or humidex.There are three main measurements of humidity: absolute, relative and specific. Absolute humidity is the water content of air at a given temperature expressed in gram per cubic metre. Relative humidity, expressed as a percent, measures the current absolute humidity relative to the maximum (highest point) for that temperature. Specific humidity is a ratio of the water vapor content of the mixture to the total air content on a mass basis.