15-1 Note 15 Properties of Bulk Matter
... phases, solid (ice), liquid (water) and gas (water vapor or steam). An idealized apparatus for the study of the phase changes of water is sketched in Figure 15-2a. A quantity of ice at an initial temperature of –20 ˚C is placed in a sealed container equipped with a thermometer. The container is then ...
... phases, solid (ice), liquid (water) and gas (water vapor or steam). An idealized apparatus for the study of the phase changes of water is sketched in Figure 15-2a. A quantity of ice at an initial temperature of –20 ˚C is placed in a sealed container equipped with a thermometer. The container is then ...
www.XtremePapers.com
... Write your name, Centre number and candidate number on the Answer Sheet in the spaces provided unless this has been done for you. There are forty questions on this paper. Answer all questions. For each question there are four possible answers A, B, C and D. Choose the one you consider correct and re ...
... Write your name, Centre number and candidate number on the Answer Sheet in the spaces provided unless this has been done for you. There are forty questions on this paper. Answer all questions. For each question there are four possible answers A, B, C and D. Choose the one you consider correct and re ...
Chapter 2: Properties of Pure Substances We now turn our attention
... ♦ P-V-T Surface for a Substance that contracts upon freezing ...
... ♦ P-V-T Surface for a Substance that contracts upon freezing ...
Chapter 1 Fluid dynamics and moist thermodynamics
... The momentum equations (1.1), the thermodynamic equation (1.12) or one of its equivalent forms (Eqs. (1.13) or (1.14)), the equation of state (1.8), and the continuity equation (1.2) or one of the approximations to it (Eqs. (1.6) or (1.7)) form a complete set for solving problems of dry air flow. For ...
... The momentum equations (1.1), the thermodynamic equation (1.12) or one of its equivalent forms (Eqs. (1.13) or (1.14)), the equation of state (1.8), and the continuity equation (1.2) or one of the approximations to it (Eqs. (1.6) or (1.7)) form a complete set for solving problems of dry air flow. For ...
CYU 1: (a) (b) CYU 2:
... has a boiling point above 100 ⬚C. CYU 17: Boiling water has a vapor pressure of one atmosphere, and the cool water in the sealed jar has a lower vapor pressure. The excess external pressure crates a net force pushing on the lid, making it hard to unscrew. CYU 18: Under pressure in the sealed bottle, ...
... has a boiling point above 100 ⬚C. CYU 17: Boiling water has a vapor pressure of one atmosphere, and the cool water in the sealed jar has a lower vapor pressure. The excess external pressure crates a net force pushing on the lid, making it hard to unscrew. CYU 18: Under pressure in the sealed bottle, ...
Content Benchmark E
... in heating occurring between the equator and the poles. Maximum insolation, also known as incoming solar radiation, occurs where the sun heats Earth’s surface at the equatorial belts causes warm air to rise in the atmosphere. At minimum insolation, where the sun heats Earth’s surface at the north an ...
... in heating occurring between the equator and the poles. Maximum insolation, also known as incoming solar radiation, occurs where the sun heats Earth’s surface at the equatorial belts causes warm air to rise in the atmosphere. At minimum insolation, where the sun heats Earth’s surface at the north an ...
Content Benchmark E
... in heating occurring between the equator and the poles. Maximum insolation, also known as incoming solar radiation, occurs where the sun heats Earth’s surface at the equatorial belts causes warm air to rise in the atmosphere. At minimum insolation, where the sun heats Earth’s surface at the north an ...
... in heating occurring between the equator and the poles. Maximum insolation, also known as incoming solar radiation, occurs where the sun heats Earth’s surface at the equatorial belts causes warm air to rise in the atmosphere. At minimum insolation, where the sun heats Earth’s surface at the north an ...
Course Solution Set 18-24
... Finally, we return to the previous arrangement at 35º C., with V = 2520 cm.3 and P’ = 108,300 Pa. We now wish to keep this volume constant by increasing the pressure on the gas in!the chamber through the use of additional weight placed on the piston. The comparison ratio here will be similar to the ...
... Finally, we return to the previous arrangement at 35º C., with V = 2520 cm.3 and P’ = 108,300 Pa. We now wish to keep this volume constant by increasing the pressure on the gas in!the chamber through the use of additional weight placed on the piston. The comparison ratio here will be similar to the ...
Chapter 3: Properties of Pure Substances
... for a substance that contracts on freezing and a substance that expands on freezing. Constant pressure curves on a temperature-volume diagram are shown in Figure 311. ...
... for a substance that contracts on freezing and a substance that expands on freezing. Constant pressure curves on a temperature-volume diagram are shown in Figure 311. ...
Chapter 1-3: Weather Forecasting A. Define, Describe, or Identify: 1
... 2. __________________________ are made using balloons that are tracked by instruments as they rise. 3. Storm detection __________________________________ gathers data showing the distribution of rain, hail, thunderstorms, tornadoes, and hurricanes. 4. A solar observation network monitors the events ...
... 2. __________________________ are made using balloons that are tracked by instruments as they rise. 3. Storm detection __________________________________ gathers data showing the distribution of rain, hail, thunderstorms, tornadoes, and hurricanes. 4. A solar observation network monitors the events ...
Name
... Yes, because both states of matter can exist at zero degree Celsius. 5. If you have 450 grams of water and the temperature is raised from 5oC to 95oC. How much heat was gained by the water? (5pts) 40500 calories 6. Explain how a heat pump functions. (4pts) A liquid is evaporated forming a cold gas. ...
... Yes, because both states of matter can exist at zero degree Celsius. 5. If you have 450 grams of water and the temperature is raised from 5oC to 95oC. How much heat was gained by the water? (5pts) 40500 calories 6. Explain how a heat pump functions. (4pts) A liquid is evaporated forming a cold gas. ...
CYL110 2012-2013 Classical Thermodynamics Sample Problems
... your thermodynamic reasoning. If the statement is false, you may either state which law or laws of thermodynamics it violates or provide a physical counter example or any other plausible physical reason. Finally, correct the false statement with a clarifying phrase that makes the statement true. (i) ...
... your thermodynamic reasoning. If the statement is false, you may either state which law or laws of thermodynamics it violates or provide a physical counter example or any other plausible physical reason. Finally, correct the false statement with a clarifying phrase that makes the statement true. (i) ...
Hurricanes get their energy via a fairly straightforward energy
... vapor molecules (or the phase/vaporization energy of the water vapor) to the thermal and kinetic energy of the air over the sea. As the air over the sea gains thermal and kinetic energy, its temperature and also its speed of motion increase. 4. As the cycle continues, the result can be very high win ...
... vapor molecules (or the phase/vaporization energy of the water vapor) to the thermal and kinetic energy of the air over the sea. As the air over the sea gains thermal and kinetic energy, its temperature and also its speed of motion increase. 4. As the cycle continues, the result can be very high win ...
Changes in Pet Food
... If the outside of the bin (assuming it is not insulated) is subjected to temperatures equal or below the dew point temperature of the internal air, condensation will occur on the inside of the bin. Condensation will form on the internal walls and will drip down and contaminate the food. ...
... If the outside of the bin (assuming it is not insulated) is subjected to temperatures equal or below the dew point temperature of the internal air, condensation will occur on the inside of the bin. Condensation will form on the internal walls and will drip down and contaminate the food. ...
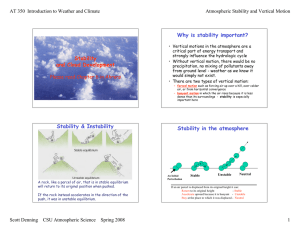
Atmospheric Stability
... – forced motion such as forcing air up over a hill, over colder air, or from horizontal convergence – buoyant motion in which the air rises because it is less dense than its surroundings - stability is especially ...
... – forced motion such as forcing air up over a hill, over colder air, or from horizontal convergence – buoyant motion in which the air rises because it is less dense than its surroundings - stability is especially ...
State Equations The Thermodynamics of State An Isentropic
... Relative Pressure and Relative Specific Volume • typically we assume specific heat to be constant with respect to temperature • but when temperature swings are significant, this assumption can lead to inaccuracies, i.e. T (K) ...
... Relative Pressure and Relative Specific Volume • typically we assume specific heat to be constant with respect to temperature • but when temperature swings are significant, this assumption can lead to inaccuracies, i.e. T (K) ...
Water Cycle in a Jar Lab
... water that you drink today might be the same water that your little brother took a bath in last year. It might be the same water that people on the other side of the world used to wash their clothes or cook their vegetables one thousand years ago. It might even be the same water that a tyrannosaurus ...
... water that you drink today might be the same water that your little brother took a bath in last year. It might be the same water that people on the other side of the world used to wash their clothes or cook their vegetables one thousand years ago. It might even be the same water that a tyrannosaurus ...
CHAPTER 13 LEARNING OBJECTIVES - crypt
... gas molecules are colliding with the walls. Q1. If there are N molecules in the gas, how many, on average, will be travelling in the x direction? Q2. Using your expression for the pressure due to one molecule and your answer to the previous question, show that the pressure due to all molecules is gi ...
... gas molecules are colliding with the walls. Q1. If there are N molecules in the gas, how many, on average, will be travelling in the x direction? Q2. Using your expression for the pressure due to one molecule and your answer to the previous question, show that the pressure due to all molecules is gi ...
FEED PREPARATION FACTORS AFFECTING THE
... inception voltage is reduced as humidity increases. At high voltages, the corona current decreases as relative humidity increases. In general, the effect of humidity is less notable in high-tension separation than in electrostatic separation, primarily because of the higher charge levels utilised [3 ...
... inception voltage is reduced as humidity increases. At high voltages, the corona current decreases as relative humidity increases. In general, the effect of humidity is less notable in high-tension separation than in electrostatic separation, primarily because of the higher charge levels utilised [3 ...
Communication: Evidence of hydrated electrons injected by a
... and an electric current is registered. The electric current indicates a flow of electrons and a simple deprotonation or polarization as the redistribution of the superficial charge on the particle does not justify the experimental observations. Deprotonation allows a polarization of the particles ge ...
... and an electric current is registered. The electric current indicates a flow of electrons and a simple deprotonation or polarization as the redistribution of the superficial charge on the particle does not justify the experimental observations. Deprotonation allows a polarization of the particles ge ...
Chapter 3
... Consequently, shortly after sunset, the earth’s surface is slightly cooler than the air directly above it. The surface air transfers some energy to the ground by conduction, which the ground, in turn, ...
... Consequently, shortly after sunset, the earth’s surface is slightly cooler than the air directly above it. The surface air transfers some energy to the ground by conduction, which the ground, in turn, ...
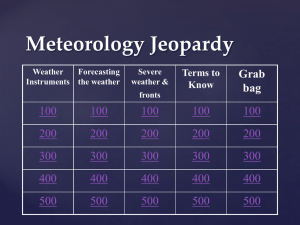
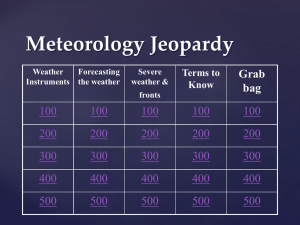
Meteorology Jeopardy Review
... the air sinks and flows outward from a center of high pressure that brings dry weather Answer ...
... the air sinks and flows outward from a center of high pressure that brings dry weather Answer ...
Meteorology Jeopardy
... the air sinks and flows outward from a center of high pressure that brings dry weather Answer ...
... the air sinks and flows outward from a center of high pressure that brings dry weather Answer ...
4th Grade Weather Read and answer each question carefully. 1
... 12) Compare the data for Utah and Colorado. Which is the most likely explanation for the difference in average high temperatures? A) Utah has more humidity. B) Colorado has more humidity. C) Utah has higher altitudes. D) Colorado has higher altitudes. ...
... 12) Compare the data for Utah and Colorado. Which is the most likely explanation for the difference in average high temperatures? A) Utah has more humidity. B) Colorado has more humidity. C) Utah has higher altitudes. D) Colorado has higher altitudes. ...
Humidity

Humidity is the amount of water vapor in the air. Water vapor is the gaseous state of water and is invisible. Humidity indicates the likelihood of precipitation, dew, or fog. Higher humidity reduces the effectiveness of sweating in cooling the body by reducing the rate of evaporation of moisture from the skin. This effect is calculated in a heat index table or humidex.There are three main measurements of humidity: absolute, relative and specific. Absolute humidity is the water content of air at a given temperature expressed in gram per cubic metre. Relative humidity, expressed as a percent, measures the current absolute humidity relative to the maximum (highest point) for that temperature. Specific humidity is a ratio of the water vapor content of the mixture to the total air content on a mass basis.