Practical Assessment of Sanitizers Steve Gray November
... Dilutions: FDA allowable non-rinse claim at use dilution specified on label ...
... Dilutions: FDA allowable non-rinse claim at use dilution specified on label ...
teaching and learning materials - UNESDOC
... Do not inhale the acids and alcohols deeply! The vapours are easily detectable. Serious respiratory damage and dulled receptiveness may occur if this rule is not adhered to. In particular, the glacial acetic acid, formic acid and isobutanol have sharp, burning odours that also irritate the eyes. Mov ...
... Do not inhale the acids and alcohols deeply! The vapours are easily detectable. Serious respiratory damage and dulled receptiveness may occur if this rule is not adhered to. In particular, the glacial acetic acid, formic acid and isobutanol have sharp, burning odours that also irritate the eyes. Mov ...
www.iitvidya.com salt analysis assignment 1. A compound on
... (iii) Its aqueous solution turns blue litmus to red. (iv) Addition of NH4OH and NaOH separately to a solution of (A) gives white precipitate which is however soluble in excess of NaOH. An inorganic compound (A), transparent like glass is a strong reducing agent. Its hydrolysis in water gives a white ...
... (iii) Its aqueous solution turns blue litmus to red. (iv) Addition of NH4OH and NaOH separately to a solution of (A) gives white precipitate which is however soluble in excess of NaOH. An inorganic compound (A), transparent like glass is a strong reducing agent. Its hydrolysis in water gives a white ...
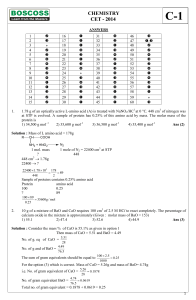
CHEMISTRY CET
... cm3 of methane. With PCC the same alcohol forms a carbonyl compound that answers silver mirror test. The monohydric alcohol is ...
... cm3 of methane. With PCC the same alcohol forms a carbonyl compound that answers silver mirror test. The monohydric alcohol is ...
Groundbreaking Measurement of Free Chlorine Disinfecting Power
... First and foremost, colorimetric tests only report how much chlorine is present, and as we saw in the previous section, knowing "how much" is not at all the same as knowing "how effective". Colorimeters and DPD kits add a reagent or several reagents to the water being tested that causes a color chan ...
... First and foremost, colorimetric tests only report how much chlorine is present, and as we saw in the previous section, knowing "how much" is not at all the same as knowing "how effective". Colorimeters and DPD kits add a reagent or several reagents to the water being tested that causes a color chan ...
- Catalyst
... Figure 4.7: Acetic acid (HC2H3O2) exists in water mostly as undissociated molecules. Weak acids partially dissociate, forming only a small number of anions and hydrated protons. ...
... Figure 4.7: Acetic acid (HC2H3O2) exists in water mostly as undissociated molecules. Weak acids partially dissociate, forming only a small number of anions and hydrated protons. ...
HEAd START TO A LEVEL CHEMISTRY WORKbOOK
... atoms and 16 g of oxygen since the relative atomic mass of hydrogen is 1 (x 2 because there two hydrogen atoms) and that of oxygen is 16. You should not learn large numbers of chemical formulae by heart. However, it is useful to know a few of them and when you do you should be able to work out the r ...
... atoms and 16 g of oxygen since the relative atomic mass of hydrogen is 1 (x 2 because there two hydrogen atoms) and that of oxygen is 16. You should not learn large numbers of chemical formulae by heart. However, it is useful to know a few of them and when you do you should be able to work out the r ...
Solubility and Reactions
... Scientists have carried out a very large number of experiments as they investigated the effects of temperature on the solubility of various solutes. From the results of their experiments, they have developed several useful generalizations about the solubility of solids, liquids, and gases in water. ...
... Scientists have carried out a very large number of experiments as they investigated the effects of temperature on the solubility of various solutes. From the results of their experiments, they have developed several useful generalizations about the solubility of solids, liquids, and gases in water. ...
Question Bank Topic 5
... (2) Reduction occurs at the positive electrode. (3) It is a device for bringing out chemical changes by electricity. A (1) and (2) only B (1) and (3) only C (2) and (3) only D (1), (2) and (3) ...
... (2) Reduction occurs at the positive electrode. (3) It is a device for bringing out chemical changes by electricity. A (1) and (2) only B (1) and (3) only C (2) and (3) only D (1), (2) and (3) ...
stoichiometry - einstein classes
... If it is a difficute to solve the problem through equivalence concept then use the mole concept. ...
... If it is a difficute to solve the problem through equivalence concept then use the mole concept. ...
the pdf of this lesson!
... 1. You may try other sources of calcium to form the gel with sodium alginate. Try a “Tums”. You will need an antacid tablet of the type that has calcium in it such as “Tums” and a little vinegar. The active ingredient in “Tums” is calcium carbonate. Place half of a “Tums” tablet in a cup and add 2 t ...
... 1. You may try other sources of calcium to form the gel with sodium alginate. Try a “Tums”. You will need an antacid tablet of the type that has calcium in it such as “Tums” and a little vinegar. The active ingredient in “Tums” is calcium carbonate. Place half of a “Tums” tablet in a cup and add 2 t ...
Precision, accuracy and significant figures
... For a quantity to have an exact value, it must either be defined or obtained by counting. All measured quantities have an inherent uncertainty because all instruments used to make measurements have limitations, and the people operating the instruments have varying skills. The accuracy of a measureme ...
... For a quantity to have an exact value, it must either be defined or obtained by counting. All measured quantities have an inherent uncertainty because all instruments used to make measurements have limitations, and the people operating the instruments have varying skills. The accuracy of a measureme ...
1.24 calculations and chemical reactions
... A 25.0 cm3 sample of this solution required 22.80 cm3 of 0.150 mol dm–3 aqueous NaOH for complete reaction. Calculate the relative molecular mass, Mr, of H2A 4.2) Sodium carbonate forms several hydrates of general formula Na2CO3.xH2O. A 2.98 g sample of one of these hydrates was dissolved in water a ...
... A 25.0 cm3 sample of this solution required 22.80 cm3 of 0.150 mol dm–3 aqueous NaOH for complete reaction. Calculate the relative molecular mass, Mr, of H2A 4.2) Sodium carbonate forms several hydrates of general formula Na2CO3.xH2O. A 2.98 g sample of one of these hydrates was dissolved in water a ...
w_4-3 Chemistry of Nitrogen Compounds
... free available chlorine to form combined chlorine compounds. Because these compounds do not readily hydrolyze to hypochlorous acid, they are poor disinfectants. In effect, these combined chlorine compounds adversely affect disinfection by consuming free available chlorine. Ammonia is readily oxidize ...
... free available chlorine to form combined chlorine compounds. Because these compounds do not readily hydrolyze to hypochlorous acid, they are poor disinfectants. In effect, these combined chlorine compounds adversely affect disinfection by consuming free available chlorine. Ammonia is readily oxidize ...
85 Q.2 Pure water has a low electricity conductivity because A. it
... sulphur dioxide bromine water B. sulphur dioxide calcium hydroxide C. carbon dioxide bromine water D. carbon dioxide calcium hydroxide 90 Q.31 16.1g of a hydrated metal sulphate was heated to constant mass. After cooling to room temperature, the residual anhydrous metal sulphate weighed 7.1g How man ...
... sulphur dioxide bromine water B. sulphur dioxide calcium hydroxide C. carbon dioxide bromine water D. carbon dioxide calcium hydroxide 90 Q.31 16.1g of a hydrated metal sulphate was heated to constant mass. After cooling to room temperature, the residual anhydrous metal sulphate weighed 7.1g How man ...
Discussion Questions
... molecular equation? Write the balanced molecular equation for each reaction. a. potassium perchlorate b. cesium nitrate c. calcium iodide 60. Carminic acid, a naturally occurring red pigment extracted from the cochineal insect, contains only carbon, hydrogen, and oxygen. It was commonly used ...
... molecular equation? Write the balanced molecular equation for each reaction. a. potassium perchlorate b. cesium nitrate c. calcium iodide 60. Carminic acid, a naturally occurring red pigment extracted from the cochineal insect, contains only carbon, hydrogen, and oxygen. It was commonly used ...
1.24 calculations and chemical reactions
... A 25.0 cm3 sample of this solution required 22.80 cm3 of 0.150 mol dm–3 aqueous NaOH for complete reaction. Calculate the relative molecular mass, Mr, of H2A 4.2) Sodium carbonate forms several hydrates of general formula Na2CO3.xH2O. A 2.98 g sample of one of these hydrates was dissolved in water a ...
... A 25.0 cm3 sample of this solution required 22.80 cm3 of 0.150 mol dm–3 aqueous NaOH for complete reaction. Calculate the relative molecular mass, Mr, of H2A 4.2) Sodium carbonate forms several hydrates of general formula Na2CO3.xH2O. A 2.98 g sample of one of these hydrates was dissolved in water a ...
- Kendriya Vidyalaya Jhunjhunu
... c. When a nail of iron is added to a solution of copper Sulphate, iron (ll) Sulphate and copper metal are formed. 2. A chemical reaction which is both combination as well as exothermic, is used by us for white washing purposes. Write the equation for the same. 3. Answer the following: a. Why cannot ...
... c. When a nail of iron is added to a solution of copper Sulphate, iron (ll) Sulphate and copper metal are formed. 2. A chemical reaction which is both combination as well as exothermic, is used by us for white washing purposes. Write the equation for the same. 3. Answer the following: a. Why cannot ...
(NH 3 ) 2 - GZ @ Science Class Online
... When adding one ionic solution to another we use a solubility grid to decide if a precipitate has formed or not. ...
... When adding one ionic solution to another we use a solubility grid to decide if a precipitate has formed or not. ...
Menstruation Workshop Notes for Chemicals in
... o Rayon comes from cellulose fibers derived from wood pulp o Wood pulp is bleached - there is no reason to bleach wood pulp other than the perception that white=c1ean=better=pure o Bleaching process used to use chlorine as part of the bleaching stages o This was the source of dioxins as dioxins bypr ...
... o Rayon comes from cellulose fibers derived from wood pulp o Wood pulp is bleached - there is no reason to bleach wood pulp other than the perception that white=c1ean=better=pure o Bleaching process used to use chlorine as part of the bleaching stages o This was the source of dioxins as dioxins bypr ...
Example of Lab Notebook
... to the theoretical value of 113–115oC. Although reasonably pure, the wider range and depressed values indicate the presence of impurities. Improper drying and removal of water could account for this result. In addition, incomplete washing the product during the filtration process could have allowed ...
... to the theoretical value of 113–115oC. Although reasonably pure, the wider range and depressed values indicate the presence of impurities. Improper drying and removal of water could account for this result. In addition, incomplete washing the product during the filtration process could have allowed ...
Chemistry SAM
... CCEA has developed new specifications which comply with criteria for GCE qualifications. The specimen assessment materials accompanying new specifications are provided to give centres guidance on the structure and character of the planned assessments in advance of the first assessment. It is intende ...
... CCEA has developed new specifications which comply with criteria for GCE qualifications. The specimen assessment materials accompanying new specifications are provided to give centres guidance on the structure and character of the planned assessments in advance of the first assessment. It is intende ...
«Классы и номенклатура неорганических соединений»
... exes of solution of alkalis the following compounds are formed: A. *hydroxo-complexes B. oxides C. hydroxides D. neutral salts E. basic salts 20. Compounds of chlorine are used as a disinfectant. What formula corresponds to hypochlorous acid: A. * HClO B. HClO4 C. HClO2 D. HClO3 E. HCl 21. Amphoteri ...
... exes of solution of alkalis the following compounds are formed: A. *hydroxo-complexes B. oxides C. hydroxides D. neutral salts E. basic salts 20. Compounds of chlorine are used as a disinfectant. What formula corresponds to hypochlorous acid: A. * HClO B. HClO4 C. HClO2 D. HClO3 E. HCl 21. Amphoteri ...
Review: The preparation of Hydantoins
... remaining sodium sulfate is cleaned and cooled with ice to give a deposit. The deep red-brown oily liquid is then extracted several times with a large amount of diethyl ether (5-10 times if necessary). The ether extracts are evaporated after dehydration with sodium sulfate to leave 65 g of α-aminois ...
... remaining sodium sulfate is cleaned and cooled with ice to give a deposit. The deep red-brown oily liquid is then extracted several times with a large amount of diethyl ether (5-10 times if necessary). The ether extracts are evaporated after dehydration with sodium sulfate to leave 65 g of α-aminois ...
selected experiments in organic chemistry
... edge of a small hot flame. The dry solid is ground to a fine powder on a piece of paper with a spatula. The open end of the capillary is then pushed into the powder which is forced down the capillary tube by gently tapping the closed end on the bench top. This is repeated several times until the sol ...
... edge of a small hot flame. The dry solid is ground to a fine powder on a piece of paper with a spatula. The open end of the capillary is then pushed into the powder which is forced down the capillary tube by gently tapping the closed end on the bench top. This is repeated several times until the sol ...
Sodium hypochlorite

Sodium hypochlorite is a chemical compound with the formula NaClO. It is composed of a sodium cation (Na+) and a hypochlorite anion (ClO−); it may also be viewed as the sodium salt of hypochlorous acid. When dissolved in water it is commonly known as bleach, or liquid bleach. Sodium hypochlorite is practically and chemically distinct from chlorine. Sodium hypochlorite is frequently used as a disinfectant or a bleaching agent.