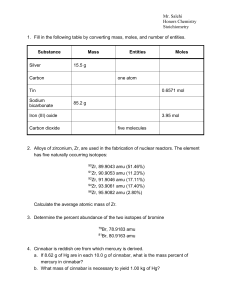
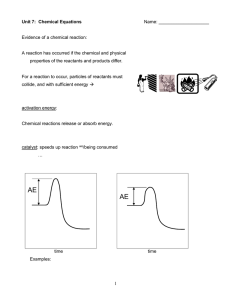
nomenclature review
... _______ tarnishes rapidly in air _______ boiling point of 883 C _______ soft, silver-white _______ reacts violently with water _______ reacts with acid ...
... _______ tarnishes rapidly in air _______ boiling point of 883 C _______ soft, silver-white _______ reacts violently with water _______ reacts with acid ...
Halogens - Cronodon
... fluorine. Fluorine is highly toxic, causing serious burns to the lungs if inhaled! Fluorine may be handled in special metal vessels, which become coated with a protective layer of metal flouride (in a similar way that Mg and Al form protective oxide layers in air). Fluorine may only be handled if HF ...
... fluorine. Fluorine is highly toxic, causing serious burns to the lungs if inhaled! Fluorine may be handled in special metal vessels, which become coated with a protective layer of metal flouride (in a similar way that Mg and Al form protective oxide layers in air). Fluorine may only be handled if HF ...
Review Package
... Propane + oxygen → carbon dioxide + water a) List all reactants in this reaction. b) List all the products in this reaction. 18) The products of neutralization reactions are always __________________ and _____________________. Predict the products and balance the equations. a) ____NaOH (aq) + ____HC ...
... Propane + oxygen → carbon dioxide + water a) List all reactants in this reaction. b) List all the products in this reaction. 18) The products of neutralization reactions are always __________________ and _____________________. Predict the products and balance the equations. a) ____NaOH (aq) + ____HC ...
In-Class Exam - Fayetteville State University
... How many grams of lead oxide will be produced by the decomposition of 2.50 g of lead carbonate? A) 0.00936 B) 2.50 C) 2.61 D) 0.41 E) 2.09 21. When the following equation is balanced, the coefficient of H2O is_____. PCl3 + H2O H3PO3 +HCl A) 1 B) 4 C) 3 D) 2 E) 5 22. How many moles of carbon dioxid ...
... How many grams of lead oxide will be produced by the decomposition of 2.50 g of lead carbonate? A) 0.00936 B) 2.50 C) 2.61 D) 0.41 E) 2.09 21. When the following equation is balanced, the coefficient of H2O is_____. PCl3 + H2O H3PO3 +HCl A) 1 B) 4 C) 3 D) 2 E) 5 22. How many moles of carbon dioxid ...
Formulae/ Equations homework - St Peter the Apostle High School
... (c) When iron is produced in the blast furnace from iron ore, the iron (III) oxide in the ore reacts with carbon monoxide gas. Carbon dioxide is also formed in the reaction. (d) Carbon dioxide gas and black copper oxide powder are formed when green copper carbonate powder is heated in a test tube. ( ...
... (c) When iron is produced in the blast furnace from iron ore, the iron (III) oxide in the ore reacts with carbon monoxide gas. Carbon dioxide is also formed in the reaction. (d) Carbon dioxide gas and black copper oxide powder are formed when green copper carbonate powder is heated in a test tube. ( ...
Formation of Solutions
... When a solution is formed, heat is released or cold is released. When sodium hydroxide dissolved in water, it becomes warmer. Opposite this, if Ammonium Nitrate is dissolved in water, the solution becomes colder. ...
... When a solution is formed, heat is released or cold is released. When sodium hydroxide dissolved in water, it becomes warmer. Opposite this, if Ammonium Nitrate is dissolved in water, the solution becomes colder. ...
1.3.9 Group 7 Elements - uses and halide tests
... Silver chloride, silver bromide and silver iodide are all insoluble salts. They are _________________ when an aqueous solution containing the appropriate halide ion is treated with an aqueous solution of _____________________________. The colours of the three silver salts formed with chloride, bromi ...
... Silver chloride, silver bromide and silver iodide are all insoluble salts. They are _________________ when an aqueous solution containing the appropriate halide ion is treated with an aqueous solution of _____________________________. The colours of the three silver salts formed with chloride, bromi ...
2016 Pre Course CHEMISTRY - Calday Grange Grammar School
... Diamond is able to scratch almost all other substances, whereas graphite may be used as a lubricant. Diamond and graphite both have high melting points. Explain each of these properties of diamond and graphite in terms of structure and bonding. Give one other difference in the properties of diamond ...
... Diamond is able to scratch almost all other substances, whereas graphite may be used as a lubricant. Diamond and graphite both have high melting points. Explain each of these properties of diamond and graphite in terms of structure and bonding. Give one other difference in the properties of diamond ...
Stoichiometry - Cloudfront.net
... 7. A 105.5 mg sample of a white substance is suspected to be cocaine, C 17H21NO4. The substance formed 279.3 mg of CO2 and 66.46 mg H2O on combustion. The compound contains 4.680% N by mass. Is the white solid cocaine? 8. An unknown compound (molar mass = 176 g/mol) contains 68.2 mass % C, 6.86 mass ...
... 7. A 105.5 mg sample of a white substance is suspected to be cocaine, C 17H21NO4. The substance formed 279.3 mg of CO2 and 66.46 mg H2O on combustion. The compound contains 4.680% N by mass. Is the white solid cocaine? 8. An unknown compound (molar mass = 176 g/mol) contains 68.2 mass % C, 6.86 mass ...
C1a - Mr Corfe
... Q1 . Salts can be made by reacting acids with alkalis. This reaction is an example of? Q2. when potassium reacts with water the colour of the flame is? Q3. When baking powder is heated, it breaks down to form new substances. This is? Q4. A coin was reacted to form a solution. Sodium hydroxide soluti ...
... Q1 . Salts can be made by reacting acids with alkalis. This reaction is an example of? Q2. when potassium reacts with water the colour of the flame is? Q3. When baking powder is heated, it breaks down to form new substances. This is? Q4. A coin was reacted to form a solution. Sodium hydroxide soluti ...
Unit A Remediation Review
... 12. What are five clues that will allow you to conclude that a chemical change has occurred? 13. Describe what occurs in the following reaction types, the general equation and an example for each: a) Formation b) Decomposition c) Single Replacement d) Double Replacement e) Combustion 14. Write a bal ...
... 12. What are five clues that will allow you to conclude that a chemical change has occurred? 13. Describe what occurs in the following reaction types, the general equation and an example for each: a) Formation b) Decomposition c) Single Replacement d) Double Replacement e) Combustion 14. Write a bal ...
UNIT 1 - MATTER AND CHEMICAL BONDING
... f) ferrous iodide l) cobalt(III) sulphate 5. Classify each of the following reactions as synthesis, single displacement, double displacement, combustion or decomposition. a) iron + copper(I) nitrate iron(II) nitrate + copper b) phosphorus + oxygen diphosphorus pentoxide c) calcium carbonate ca ...
... f) ferrous iodide l) cobalt(III) sulphate 5. Classify each of the following reactions as synthesis, single displacement, double displacement, combustion or decomposition. a) iron + copper(I) nitrate iron(II) nitrate + copper b) phosphorus + oxygen diphosphorus pentoxide c) calcium carbonate ca ...
students - Teach-n-Learn-Chem
... For single-replacement reactions, use Activity Series. In general, elements above replace ...
... For single-replacement reactions, use Activity Series. In general, elements above replace ...
Sodium hypochlorite

Sodium hypochlorite is a chemical compound with the formula NaClO. It is composed of a sodium cation (Na+) and a hypochlorite anion (ClO−); it may also be viewed as the sodium salt of hypochlorous acid. When dissolved in water it is commonly known as bleach, or liquid bleach. Sodium hypochlorite is practically and chemically distinct from chlorine. Sodium hypochlorite is frequently used as a disinfectant or a bleaching agent.