Nuclear Reactions - Kelso High School
... state that in fission a nucleus of large mass number splits into two nuclei of small mass numbers along with several neutrons. ...
... state that in fission a nucleus of large mass number splits into two nuclei of small mass numbers along with several neutrons. ...
AP Revision Guide Ch 18
... Beta radiation Both electrons and positrons are known as beta particles, sometimes written β– and β+ respectively. In beta decay, (anti) neutrinos (or neutrinos) are also emitted, and carry away energy and momentum. The energies of beta particles therefore vary, up to the maximum available from the ...
... Beta radiation Both electrons and positrons are known as beta particles, sometimes written β– and β+ respectively. In beta decay, (anti) neutrinos (or neutrinos) are also emitted, and carry away energy and momentum. The energies of beta particles therefore vary, up to the maximum available from the ...
1 0 +1 0 - davis.k12.ut.us
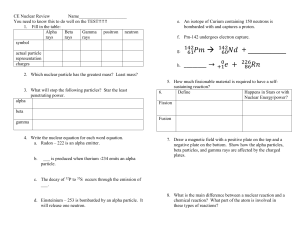
... Alpha, beta, gamma 20. Define Free Radicals and describe why they are dangerous. ...
... Alpha, beta, gamma 20. Define Free Radicals and describe why they are dangerous. ...
Chapter 29
... • Nuclei with mass numbers greater than or less than 60 are not as strongly bound as those near the middle of the periodic table • The curve is slowly varying at A > 40, suggesting that the nuclear force saturates • A particular nucleon can interact with only a limited number of other nucleons ...
... • Nuclei with mass numbers greater than or less than 60 are not as strongly bound as those near the middle of the periodic table • The curve is slowly varying at A > 40, suggesting that the nuclear force saturates • A particular nucleon can interact with only a limited number of other nucleons ...
Nuclear Decay
... Fusion is a process where nuclei collide and join together to form a heavier atom, usually deuterium and tritium. When this happens a considerable amount of energy gets released at extremely high temperatures: nearly 150 million degrees Celsius. At extreme temperatures, electrons are separated from ...
... Fusion is a process where nuclei collide and join together to form a heavier atom, usually deuterium and tritium. When this happens a considerable amount of energy gets released at extremely high temperatures: nearly 150 million degrees Celsius. At extreme temperatures, electrons are separated from ...
Objective 2 Average Atomic Mass
... Another term used to describe the process by which one element spontaneously changes into another element is (14) ____________________. Any isotope that undergoes such changes is called a(n) (15)___________________. There are three common forms of radiation. One type is a form of energy known as (16 ...
... Another term used to describe the process by which one element spontaneously changes into another element is (14) ____________________. Any isotope that undergoes such changes is called a(n) (15)___________________. There are three common forms of radiation. One type is a form of energy known as (16 ...
Remediation_unit 2_standard
... Another term used to describe the process by which one element spontaneously changes into another element is (14) ____________________. Any isotope that undergoes such changes is called a(n) (15)___________________. There are three common forms of radiation. One type is a form of energy known as (16 ...
... Another term used to describe the process by which one element spontaneously changes into another element is (14) ____________________. Any isotope that undergoes such changes is called a(n) (15)___________________. There are three common forms of radiation. One type is a form of energy known as (16 ...
Chapter 29
... • These forces should cause the nucleus to fly apart • The nuclei are stable because of the presence of another, short-range force, called the nuclear force • This is an attractive force that acts between all nuclear particles • The nuclear attractive force is stronger than the Coulomb repulsive for ...
... • These forces should cause the nucleus to fly apart • The nuclei are stable because of the presence of another, short-range force, called the nuclear force • This is an attractive force that acts between all nuclear particles • The nuclear attractive force is stronger than the Coulomb repulsive for ...
Radiation and Radioactive Decay
... more nucleons, however the distance between them is increasing and the mutual attraction due to the (short-range) strong nuclear force is weakened. But the proton pairs are still feeling the electromagnet repulsion force. As we approach elements such as radium, polonium, and uranium, the binding ene ...
... more nucleons, however the distance between them is increasing and the mutual attraction due to the (short-range) strong nuclear force is weakened. But the proton pairs are still feeling the electromagnet repulsion force. As we approach elements such as radium, polonium, and uranium, the binding ene ...
- Physics
... each other. If the uranium-235 is in the shape of a sphere about 13 pounds of uranium form a critical mass where a runaway chain reaction (bomb) can occur. I would restate the last paragraph on page 20-4. In the process of fission some mass becomes kinetic energy of the fission fragments. It is true ...
... each other. If the uranium-235 is in the shape of a sphere about 13 pounds of uranium form a critical mass where a runaway chain reaction (bomb) can occur. I would restate the last paragraph on page 20-4. In the process of fission some mass becomes kinetic energy of the fission fragments. It is true ...
Lab 77 Nuclear Radiation Detection
... are called radioisotopes. All elements with atomic numbers greater than 82 (and some with smaller atomic numbers) possess naturally radioactive isotopes. In addition, artificial radioactive isotopes can be created by bombarding certain stable isotopes with particles. To date, over 2,000 radioisotope ...
... are called radioisotopes. All elements with atomic numbers greater than 82 (and some with smaller atomic numbers) possess naturally radioactive isotopes. In addition, artificial radioactive isotopes can be created by bombarding certain stable isotopes with particles. To date, over 2,000 radioisotope ...
Ionizing radiation
Ionizing (or ionising in British English) radiation is radiation that carries enough energy to free electrons from atoms or molecules, thereby ionizing them. Ionizing radiation is made up of energetic subatomic particles, ions or atoms moving at relativistic speeds, and electromagnetic waves on the high-energy end of the electromagnetic spectrum.Gamma rays, X-rays, and the higher ultraviolet part of the electromagnetic spectrum are ionizing, whereas the lower ultraviolet part of the electromagnetic spectrum, visible light (including nearly all types of laser light), infrared, microwaves, and radio waves are considered non-ionizing radiation. The boundary between ionizing and non-ionizing electromagnetic radiation that occurs in the ultraviolet is not sharply defined, since different molecules and atoms ionize at different energies. Conventional definition places the boundary at a photon energy between 10 eV and 33 eV in the ultraviolet (see definition boundary section below).Typical ionizing subatomic particles from radioactivity include alpha particles, beta particles and neutrons. Almost all products of radioactive decay are ionizing because the energy of radioactive decay is typically far higher than that required to ionize. Other subatomic ionizing particles which occur naturally are muons, mesons, positrons, neutrons and other particles that constitute the secondary cosmic rays that are produced after primary cosmic rays interact with Earth's atmosphere. Cosmic rays may also produce radioisotopes on Earth (for example, carbon-14), which in turn decay and produce ionizing radiation.Cosmic rays and the decay of radioactive isotopes are the primary sources of natural ionizing radiation on Earth referred to as background radiation.In space, natural thermal radiation emissions from matter at extremely high temperatures (e.g. plasma discharge or the corona of the Sun) may be ionizing. Ionizing radiation may be produced naturally by the acceleration of charged particles by natural electromagnetic fields (e.g. lightning), although this is rare on Earth. Natural supernova explosions in space produce a great deal of ionizing radiation near the explosion, which can be seen by its effects in the glowing nebulae associated with them.Ionizing radiation can also be generated artificially using X-ray tubes, particle accelerators, and any of the various methods that produce radioisotopes artificially.Ionizing radiation is invisible and not directly detectable by human senses, so radiation detection instruments such as Geiger counters are required. However, ionizing radiation may lead to secondary emission of visible light upon interaction with matter, such as in Cherenkov radiation and radioluminescence.Ionizing radiation is applied constructively in a wide variety of fields such as medicine, research, manufacturing, construction, and many other areas, but presents a health hazard if proper measures against undesired exposure aren't followed. Exposure to ionizing radiation causes damage to living tissue, and can result in mutation, radiation sickness, cancer, and death.