Many Worlds Theory/ `Relative State` formation of Quantum Mechanics
... Many Worlds Theory/ ‘Relative State’ formation of Quantum Mechanics/ Quantum Multiverse: In different universes, this project is going by each/all of these names… and infinite more. What is it? • An interpretation of the universe that proposes that all histories/possible outcomes of a situation are ...
... Many Worlds Theory/ ‘Relative State’ formation of Quantum Mechanics/ Quantum Multiverse: In different universes, this project is going by each/all of these names… and infinite more. What is it? • An interpretation of the universe that proposes that all histories/possible outcomes of a situation are ...
chem6V19_postulates
... where H(t) is the observable associated with the total energy of the system. ...
... where H(t) is the observable associated with the total energy of the system. ...
THE UNCERTAINTY PRINCIPLE The uncertainty principle states
... THE UNCERTAINTY PRINCIPLE MICHAEL G COWLING ...
... THE UNCERTAINTY PRINCIPLE MICHAEL G COWLING ...
There are 4 quantum numbers. - 12S7F-note
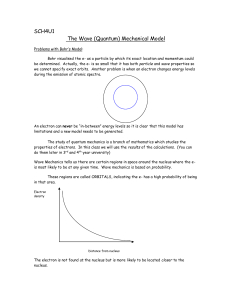
... Atomic Structure Atomic Structure: There are 4 quantum numbers. ...
... Atomic Structure Atomic Structure: There are 4 quantum numbers. ...
Eighth International Conference on Geometry, Integrability and Quantization
... was that elementary particles need not be pointlike. Being extended and non rigid is a better conception. Rather than conceiving the particle as a bulk of fluid, we have supposed that it is composed of pointlike quantum modes. This enabled the construction of our Geometro-Differential Model (G-D-M) ...
... was that elementary particles need not be pointlike. Being extended and non rigid is a better conception. Rather than conceiving the particle as a bulk of fluid, we have supposed that it is composed of pointlike quantum modes. This enabled the construction of our Geometro-Differential Model (G-D-M) ...
Pauli Exclusion Principle Quiz
... Pauli Exclusion Principle Quiz 1. The location of any electron in an atom can be described by ____ unique quantum numbers. ...
... Pauli Exclusion Principle Quiz 1. The location of any electron in an atom can be described by ____ unique quantum numbers. ...