chemical bonding and molecular structure
... crystalline compounds. His views have proved to be of great value in the understanding and systematisation of the ionic compounds. At the same time he did recognise the fact that a large number of compounds did not fit into these concepts. 4.1.1 Octet Rule Kössel and Lewis in 1916 developed an impor ...
... crystalline compounds. His views have proved to be of great value in the understanding and systematisation of the ionic compounds. At the same time he did recognise the fact that a large number of compounds did not fit into these concepts. 4.1.1 Octet Rule Kössel and Lewis in 1916 developed an impor ...
AP Chem Chapter 13 Homework
... a. In the absence of a phase change, the viscosity of a liquid increases as temperature decreases. b. All other factors being equal, if adhesive forces are strong, capillary action is likely to occur less readily than if adhesive forces are weak. c. The shape of a meniscus depends on the difference ...
... a. In the absence of a phase change, the viscosity of a liquid increases as temperature decreases. b. All other factors being equal, if adhesive forces are strong, capillary action is likely to occur less readily than if adhesive forces are weak. c. The shape of a meniscus depends on the difference ...
Structure and Properties of Polymers
... Macromolecules, i.e., big molecules, are molecules with large or very large molecular mass. Molecules of common low-molecular-weight substances, such as water - H2O, oxygen - O2, carbon dioxide - CO2, toluene - C6H5CH3, etc., have relative molecular masses of the order of magnitude of tens or hundre ...
... Macromolecules, i.e., big molecules, are molecules with large or very large molecular mass. Molecules of common low-molecular-weight substances, such as water - H2O, oxygen - O2, carbon dioxide - CO2, toluene - C6H5CH3, etc., have relative molecular masses of the order of magnitude of tens or hundre ...
CHEMICAL EQUILIBRIUM (Part II)II)
... What do you think will happen if more NH3 is added? The equilibrium of system will be disturbed, and Qc > Kc . In order for the system to reestablish equilibrium, more NH3 must be consumed and more H2 and N2 must be produced until Qc = Kc . o ...
... What do you think will happen if more NH3 is added? The equilibrium of system will be disturbed, and Qc > Kc . In order for the system to reestablish equilibrium, more NH3 must be consumed and more H2 and N2 must be produced until Qc = Kc . o ...
L9 Tensor properties, anisotropy, part 2
... • Symmetry becomes important whenever a thermodynamic function depends on the gradient of a property; in such cases, symmetry theory can determine the possible mathematical expressions that will correctly describe this property. There is thus an intimate relation between the symmetry of a material a ...
... • Symmetry becomes important whenever a thermodynamic function depends on the gradient of a property; in such cases, symmetry theory can determine the possible mathematical expressions that will correctly describe this property. There is thus an intimate relation between the symmetry of a material a ...
In_Class_Practice Chapter 17 PreAP
... 6. Use Le Châtelier’s principle to predict how each of the following changes would affect this equilibrium. C2H4O(g) CH4(g) + CO(g) a. adding CH4(g) to the system b. removing CO(g) from the system c. removing C2H4O(g) from the system 7. How would decreasing the volume of the reaction vessel affect ...
... 6. Use Le Châtelier’s principle to predict how each of the following changes would affect this equilibrium. C2H4O(g) CH4(g) + CO(g) a. adding CH4(g) to the system b. removing CO(g) from the system c. removing C2H4O(g) from the system 7. How would decreasing the volume of the reaction vessel affect ...
CHEM 1212 Module Ten-Chapter 16 Name
... __________________ 1. If all the reactants and the products are in the same phase (such as gases), then the we say the system is in _______________equilibrium. __________________ 2. If at least one of the reactants or one of the products is not in the same phase, then we say the system is in _______ ...
... __________________ 1. If all the reactants and the products are in the same phase (such as gases), then the we say the system is in _______________equilibrium. __________________ 2. If at least one of the reactants or one of the products is not in the same phase, then we say the system is in _______ ...
The chemistry of two-dimensional layered transition metal
... for 2H polymorphs. A single TMD can be found in multiple polymorphs or polytypes, depending on the history of its formation. For example, natural MoS2 is commonly found in the ‘2H phase’ where the stacking sequence is AbA BaB (The capital and lower case letters denote chalcogen and metal atoms, resp ...
... for 2H polymorphs. A single TMD can be found in multiple polymorphs or polytypes, depending on the history of its formation. For example, natural MoS2 is commonly found in the ‘2H phase’ where the stacking sequence is AbA BaB (The capital and lower case letters denote chalcogen and metal atoms, resp ...
Computational investigations of the electronic structure of molecular
... I would first like to thank my supervisor Nik Kaltsoyannis for his patience and support in helping me finish this PhD project. There were times when results were not forthcoming or needed explanations but he provided stability, calm and guidance at those times. I would like to thank Jonas Häller for ...
... I would first like to thank my supervisor Nik Kaltsoyannis for his patience and support in helping me finish this PhD project. There were times when results were not forthcoming or needed explanations but he provided stability, calm and guidance at those times. I would like to thank Jonas Häller for ...
"Fundamentals of Rotation--Vibration Spectra" in
... form, there are consequently no simple analytical expressions available for the energy levels or rovibrational term values except for model potentials. Thus, in practical spectroscopy, one uses approximations leading to simple formulae for the term values, which can be expressed by tables of constan ...
... form, there are consequently no simple analytical expressions available for the energy levels or rovibrational term values except for model potentials. Thus, in practical spectroscopy, one uses approximations leading to simple formulae for the term values, which can be expressed by tables of constan ...
Comparison of the electronic structures of four crystalline phases of
... the delithiated form of LiFePO4 in connection with Li ion battery cathodes.1 The mineral names “heterosite” and “olivine” have been used to describe this orthorhombic form of iron phosphate which has the space group symmetry P nma (No. 62 in the International Tables for Crystallography2 ). Although ...
... the delithiated form of LiFePO4 in connection with Li ion battery cathodes.1 The mineral names “heterosite” and “olivine” have been used to describe this orthorhombic form of iron phosphate which has the space group symmetry P nma (No. 62 in the International Tables for Crystallography2 ). Although ...
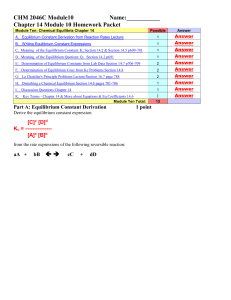
Document
... Equilibrium Constant (concentrations) = Kc = [A]m[B]n …. A more accurate equilibrium constant is Keq, which will be discussed in detail in Physical Chemistry I (59-240). Thermodynamic Equilibrium constant = Keq = ...
... Equilibrium Constant (concentrations) = Kc = [A]m[B]n …. A more accurate equilibrium constant is Keq, which will be discussed in detail in Physical Chemistry I (59-240). Thermodynamic Equilibrium constant = Keq = ...
1165 KB - Max-Planck-Institut für Kolloid
... monovalent (alkali) counter ions was investigated. It was found that the surface activity of surface chemically pure decylsulfate significantly increases with decreasing hydrated size of the counter ion. Fig. 1 presents the dependence of the surface tension of decylsulfate on its counter ion in solu ...
... monovalent (alkali) counter ions was investigated. It was found that the surface activity of surface chemically pure decylsulfate significantly increases with decreasing hydrated size of the counter ion. Fig. 1 presents the dependence of the surface tension of decylsulfate on its counter ion in solu ...
exercise on Chapter 13 - Louisiana Tech University
... chemical reactions are dynamic because the forward and reverse reactions rates are equal. Le Chaterlier’s Principle provides a simple qualitative rule for analyzing the effects of perturbations on equilibrium. You can use it in evaluating the direction and extent of change that will take place. Equi ...
... chemical reactions are dynamic because the forward and reverse reactions rates are equal. Le Chaterlier’s Principle provides a simple qualitative rule for analyzing the effects of perturbations on equilibrium. You can use it in evaluating the direction and extent of change that will take place. Equi ...
DX4301741751
... the organic molecules with physical properties of the inorganics has been found to be overwhelmingly successful in the recent search. Hence the recent search is concentrated on semiorganic materials due to their large nonlinearity, high resistance to laser induced damage, low angular sensitivity and ...
... the organic molecules with physical properties of the inorganics has been found to be overwhelmingly successful in the recent search. Hence the recent search is concentrated on semiorganic materials due to their large nonlinearity, high resistance to laser induced damage, low angular sensitivity and ...
Equilibrium
... *Le Chatelier's Principle: If a "____________" is applied to a system at equilibrium, the equilibrium condition is _________; a net reaction (“shift”) occurs in that direction which tends to ___________ the "stress," and a ___________ equilibrium is obtained. initial equilibrium state “non-equi ...
... *Le Chatelier's Principle: If a "____________" is applied to a system at equilibrium, the equilibrium condition is _________; a net reaction (“shift”) occurs in that direction which tends to ___________ the "stress," and a ___________ equilibrium is obtained. initial equilibrium state “non-equi ...
Multiwalled Boron Nitride Nanotubes: Growth, Properties, and
... Laser heating is one of the possible methods to synthesize BNNTs as demonstrated by Golberg et al. [44]. Single crystal c-BN specimens were laser heated in a diamond anvil cell under high nitrogen pressures. TEM and other characterization techniques confirmed the product to be BNNTs. Another laser-b ...
... Laser heating is one of the possible methods to synthesize BNNTs as demonstrated by Golberg et al. [44]. Single crystal c-BN specimens were laser heated in a diamond anvil cell under high nitrogen pressures. TEM and other characterization techniques confirmed the product to be BNNTs. Another laser-b ...
Graphene: carbon in two dimensions
... Fig. 5 Scanning electron micrograph of a graphene device. The graphene crystal is contacted by Au electrodes and patterned into Hall bar geometry by e-beam lithography with subsequent reactive plasma etching. The width of the channel is 1 µm. (Courtesy of K. Novoselov and A. Geim.) ...
... Fig. 5 Scanning electron micrograph of a graphene device. The graphene crystal is contacted by Au electrodes and patterned into Hall bar geometry by e-beam lithography with subsequent reactive plasma etching. The width of the channel is 1 µm. (Courtesy of K. Novoselov and A. Geim.) ...
- University of Bath Opus
... the DOS of mo-1 and or-1 structures, we find that they are very similar, even in the band gaps which are 0.84 eV for mo-1 and 0.88 eV for or-1 structure. This similarity is also observed for the calculated DOS of other structures with only Cu3 Sn and Cu2 Sn2 clusters around the S anions, indicating t ...
... the DOS of mo-1 and or-1 structures, we find that they are very similar, even in the band gaps which are 0.84 eV for mo-1 and 0.88 eV for or-1 structure. This similarity is also observed for the calculated DOS of other structures with only Cu3 Sn and Cu2 Sn2 clusters around the S anions, indicating t ...
Principles of Reactivity: Chemical Equilibria
... and [isobutane] = 2.18 M? If it is not at equilibrium, in which direction will the reaction proceed in order to achieve equilibrium? Is the system at equilibrium when [butane] = 0.75 M and [isobutane] = 2.60 M? If it is not at equilibrium, in which direction will the reaction proceed in order to ach ...
... and [isobutane] = 2.18 M? If it is not at equilibrium, in which direction will the reaction proceed in order to achieve equilibrium? Is the system at equilibrium when [butane] = 0.75 M and [isobutane] = 2.60 M? If it is not at equilibrium, in which direction will the reaction proceed in order to ach ...
Ferroelectric Ceramics - Universiti Sains Malaysia
... This is because of the formation of multiple domains in a single grain, the motion of whose walls increases the dielectric constant at the Curie point. For a BaTiO3 ceramic with fine grains (~ 1 m m), a single domain forms inside each grain. The movement of domain walls are restricted by the grain b ...
... This is because of the formation of multiple domains in a single grain, the motion of whose walls increases the dielectric constant at the Curie point. For a BaTiO3 ceramic with fine grains (~ 1 m m), a single domain forms inside each grain. The movement of domain walls are restricted by the grain b ...
Materials Chemistry B
... these pigment classes feature H-bonding between –NH hydrogens (donor) and carbonyl groups (acceptor), reminiscent of the same type of H-bonding interactions present in proteins. The interplay of H-bonding and p-stacking results in high lattice energies. This fact is reected in the remarkable therma ...
... these pigment classes feature H-bonding between –NH hydrogens (donor) and carbonyl groups (acceptor), reminiscent of the same type of H-bonding interactions present in proteins. The interplay of H-bonding and p-stacking results in high lattice energies. This fact is reected in the remarkable therma ...
Classification of polymers
... Polymers containing amide or carbonyl groups can form hydrogen bonds between adjacent chains. Polyesters have dipole –dipole bonding between the oxygen atoms in C=Ogroups and the hydrogen ato.ms in C-H groups. Dipole bonding is not as strong as hydrogen bonding ...
... Polymers containing amide or carbonyl groups can form hydrogen bonds between adjacent chains. Polyesters have dipole –dipole bonding between the oxygen atoms in C=Ogroups and the hydrogen ato.ms in C-H groups. Dipole bonding is not as strong as hydrogen bonding ...
Molecular Recognition of Trans-Chiral Schiff Base
... tures (including thermally-induced structural phase transition and polymorphism by sol‐ vents) of trans-type chiral Schiff base metal complexes and extract important features of chiral molecular recognition in the solid states. As mentioned in Abstract section, we have tested observation of some nov ...
... tures (including thermally-induced structural phase transition and polymorphism by sol‐ vents) of trans-type chiral Schiff base metal complexes and extract important features of chiral molecular recognition in the solid states. As mentioned in Abstract section, we have tested observation of some nov ...
Order date : 24-07-2010
... Coordination chemistry formation and stability of complexes, chelate and macrocyclic effect. The crystal field and ligand field theories, orbital splitting in octahedral, tetrahedral and square planar fields, spectrochemical- and nephelauxetic series, Jahn-Teller effect. M.O. theory- composition of ...
... Coordination chemistry formation and stability of complexes, chelate and macrocyclic effect. The crystal field and ligand field theories, orbital splitting in octahedral, tetrahedral and square planar fields, spectrochemical- and nephelauxetic series, Jahn-Teller effect. M.O. theory- composition of ...