The Role of Hydrogen Bond in Designing Molecular Optical Materials
... expansion. For the partition, one can use the quantum theory of atoms in molecules (QTAIM) [6], which is more generally applicable to a theoretical electron density as well. An advantage of QTAIM is that the partitioning is exact (meaning that the sum of atomic contributions exactly reconstruct the ...
... expansion. For the partition, one can use the quantum theory of atoms in molecules (QTAIM) [6], which is more generally applicable to a theoretical electron density as well. An advantage of QTAIM is that the partitioning is exact (meaning that the sum of atomic contributions exactly reconstruct the ...
Equilibrium - District 196
... reactions occur nearly at the same rate before chemical equilibrium is established. Neither reaction is favored • Do not just assume that if you see an equilibrium arrow ( ) that neither side of the system is favored. Use the equilibrium constant to determine for sure! ...
... reactions occur nearly at the same rate before chemical equilibrium is established. Neither reaction is favored • Do not just assume that if you see an equilibrium arrow ( ) that neither side of the system is favored. Use the equilibrium constant to determine for sure! ...
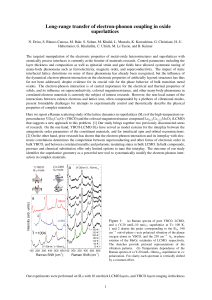
Long-range transfer of electron-phonon coupling in oxide superlattices
... The two low-energy vibrations have been the focus of extensive prior investigations on bulk samples, which have established that they are subject to strong electron-phonon interactions. The influence of the electron-phonon interaction can be recognized in the lineshapes of the Raman profiles and in ...
... The two low-energy vibrations have been the focus of extensive prior investigations on bulk samples, which have established that they are subject to strong electron-phonon interactions. The influence of the electron-phonon interaction can be recognized in the lineshapes of the Raman profiles and in ...
Chapter 13
... because it is a pure liquid. However, if water is a gas rather than a liquid, 2H2O(g) 2H2(g) + O2(g) ...
... because it is a pure liquid. However, if water is a gas rather than a liquid, 2H2O(g) 2H2(g) + O2(g) ...
The effect of confinement on chemical reactions
... significantly higher density than the bulk phase. The combination of these two factors naturally causes an enhancement of the equilibrium yield, as the increased density of the adsorbed phase displaces the equilibrium to the side with a lower number of moles. It would be interesting to consider now ...
... significantly higher density than the bulk phase. The combination of these two factors naturally causes an enhancement of the equilibrium yield, as the increased density of the adsorbed phase displaces the equilibrium to the side with a lower number of moles. It would be interesting to consider now ...
Electronic Structure and Transport Properties of Carbon Based Materials Anders Hansson Link¨
... The size and complexity of molecules, polymers or unit cells of crystals that can be treated with ab initio quantum chemistry or first principles solid state physics methods have increased dramatically. Today we are on a routine base performing ab initio or first principles calculations of molecules ...
... The size and complexity of molecules, polymers or unit cells of crystals that can be treated with ab initio quantum chemistry or first principles solid state physics methods have increased dramatically. Today we are on a routine base performing ab initio or first principles calculations of molecules ...
“No Score” from Exam 1??
... 3. The electron domain geometry corresponds to the number of electron domains 4. The molecular geometry is defined by the positions of only ________________ in the molecules, _______ the nonbonding pairs ...
... 3. The electron domain geometry corresponds to the number of electron domains 4. The molecular geometry is defined by the positions of only ________________ in the molecules, _______ the nonbonding pairs ...
Andrea Marino - Université de Rennes 1
... The ability to photo-switch physical/chemical properties of functional materials through photo induced phase transition opens fascinating perspectives for driving the material towards new state out of thermal equilibrium. However, it is fundamental to disentangle and understand all the dynamical phe ...
... The ability to photo-switch physical/chemical properties of functional materials through photo induced phase transition opens fascinating perspectives for driving the material towards new state out of thermal equilibrium. However, it is fundamental to disentangle and understand all the dynamical phe ...
Correlation between Crystal Structure and Mobility in Organic Field
... in Figure 1, namely bis(ethylenedithio)-tetrathiafulvalene (BEDTTTF), (ethylenethio)(ethylenedithio)-tetrathiafulvalene (ETEDTTTF), bis(ethylenethio)-tetrathiafulvalene (BET-TTF), (ethylenethio)(thiodimethylene)-tetrathiafulvalene (ETTDM-TTF), dithiophene-tetrathiafulvalene (DT-TTF), (thiophene)(thi ...
... in Figure 1, namely bis(ethylenedithio)-tetrathiafulvalene (BEDTTTF), (ethylenethio)(ethylenedithio)-tetrathiafulvalene (ETEDTTTF), bis(ethylenethio)-tetrathiafulvalene (BET-TTF), (ethylenethio)(thiodimethylene)-tetrathiafulvalene (ETTDM-TTF), dithiophene-tetrathiafulvalene (DT-TTF), (thiophene)(thi ...
Chapter 3. Materials and Film Preparation - diss.fu
... Cu, Zn, or 2H. A set of these compounds is similar to that formed by the tetraazaporphyrin (or porphyrazine) (TAP), phthalocyanine (Pc), naphthalocyanine (Nc) and anthracyanine (Ac) (see Fig. 1). The chemical structures of P0-Me and MeTAP, P1-Me and MePc, or P2Me and MeNc differ by the substitution ...
... Cu, Zn, or 2H. A set of these compounds is similar to that formed by the tetraazaporphyrin (or porphyrazine) (TAP), phthalocyanine (Pc), naphthalocyanine (Nc) and anthracyanine (Ac) (see Fig. 1). The chemical structures of P0-Me and MeTAP, P1-Me and MePc, or P2Me and MeNc differ by the substitution ...
Chemistry 12 Keq WORKSHEET #1
... A reaction vessel with a capacity of 1.0 litre, in which the following reaction : SO2 (g) + NO2 (g) <===> SO3 (g) + NO (g) had reached a state of equilibrium, was found to contain 0.40 moles of SO3 , 0.30 moles of NO, 0.15 moles of NO2 , and 0.20 moles of SO2. Calculate the equilibrium constant for ...
... A reaction vessel with a capacity of 1.0 litre, in which the following reaction : SO2 (g) + NO2 (g) <===> SO3 (g) + NO (g) had reached a state of equilibrium, was found to contain 0.40 moles of SO3 , 0.30 moles of NO, 0.15 moles of NO2 , and 0.20 moles of SO2. Calculate the equilibrium constant for ...
Assessing carbon-based anodes for lithium-ion
... found for pristine graphene.[30] Although we have specifically developed the SFM to explain the physical principles underlying Li binding on the π manifold of sp2 C, we emphasize that it should be generalizable to other systems and applications where charge transfer dominates the adsorption behavior ...
... found for pristine graphene.[30] Although we have specifically developed the SFM to explain the physical principles underlying Li binding on the π manifold of sp2 C, we emphasize that it should be generalizable to other systems and applications where charge transfer dominates the adsorption behavior ...
Solid State Physics (I)
... overlapping between the replica. • Primitive unit cell: unit cell with the smallest volume • Unit cell can form from fundamental vectors, but it can also be constructed by other methods (e. g. Wigner-Seitz primitive cell). • As long as the fundamental vectors are fixed, all the unit cells have the s ...
... overlapping between the replica. • Primitive unit cell: unit cell with the smallest volume • Unit cell can form from fundamental vectors, but it can also be constructed by other methods (e. g. Wigner-Seitz primitive cell). • As long as the fundamental vectors are fixed, all the unit cells have the s ...
Word - chemmybear.com
... 12. Bromine and chlorine react to produce bromine monochloride according to the equation. Kc = 36.0 under the conditions of the experiment. Br2(g) + Cl2(g) 2 BrCl(g) If 0.180 moles of bromine gas and 0.180 moles of chlorine gas are introduced into a 3.0 Liter flask and allowed to come to equilibrium ...
... 12. Bromine and chlorine react to produce bromine monochloride according to the equation. Kc = 36.0 under the conditions of the experiment. Br2(g) + Cl2(g) 2 BrCl(g) If 0.180 moles of bromine gas and 0.180 moles of chlorine gas are introduced into a 3.0 Liter flask and allowed to come to equilibrium ...
South Pasadena · AP Chemistry
... 12. Bromine and chlorine react to produce bromine monochloride according to the equation. Kc = 36.0 under the conditions of the experiment. Br2(g) + Cl2(g) 2 BrCl(g) If 0.180 moles of bromine gas and 0.180 moles of chlorine gas are introduced into a 3.0 Liter flask and allowed to come to equilibrium ...
... 12. Bromine and chlorine react to produce bromine monochloride according to the equation. Kc = 36.0 under the conditions of the experiment. Br2(g) + Cl2(g) 2 BrCl(g) If 0.180 moles of bromine gas and 0.180 moles of chlorine gas are introduced into a 3.0 Liter flask and allowed to come to equilibrium ...
Chapter 4 Power Point Quiz
... How many grams of glucose (C6H12O6) form when 4.40 g of CO2 react? a) b) c) d) ...
... How many grams of glucose (C6H12O6) form when 4.40 g of CO2 react? a) b) c) d) ...
Plastic nanocomposite insulation material enabling reliable
... to be modified in such a way that the smallest accessible units (normally called silica aggregates) are exposed to the modifying agent. On the other hand, the polar character within the primary particles of the silica should remain in order to guarantee a high electrical resistivity. The idea is to ...
... to be modified in such a way that the smallest accessible units (normally called silica aggregates) are exposed to the modifying agent. On the other hand, the polar character within the primary particles of the silica should remain in order to guarantee a high electrical resistivity. The idea is to ...
4.5 Physical properties of molecular covalent
... Glycerol (HOCH2CH(OH)CH2OH) has high and water (H2O) has low viscosity. The more viscous fluid flows more slowly because the intermolecular forces between the molecules are stronger increasing the attraction the molecules have for one another. Viscosity increases with temperature. ...
... Glycerol (HOCH2CH(OH)CH2OH) has high and water (H2O) has low viscosity. The more viscous fluid flows more slowly because the intermolecular forces between the molecules are stronger increasing the attraction the molecules have for one another. Viscosity increases with temperature. ...
Introduction to Organic Electronics
... Organic materials can form very highly ordered crystals Van der Waals forces keep these crystals together. These crystals can have a band structure just like any other semiconductor if the crystals are highly order and the concentration of impurities is very low. ...
... Organic materials can form very highly ordered crystals Van der Waals forces keep these crystals together. These crystals can have a band structure just like any other semiconductor if the crystals are highly order and the concentration of impurities is very low. ...
Fulltext PDF - Indian Academy of Sciences
... charged species at ROB3LYP level using a smaller basis and then by single point PCM calculations at the same level but using the larger basis set. 2.3 Thermal energy and molecular entropy Thermal energy and entropy contribution towards the free energy change of the reductive process are obtained for ...
... charged species at ROB3LYP level using a smaller basis and then by single point PCM calculations at the same level but using the larger basis set. 2.3 Thermal energy and molecular entropy Thermal energy and entropy contribution towards the free energy change of the reductive process are obtained for ...
Organic Semiconductors - Physik Uni
... delocalization of electronic wavefunctions amongst neighboring molecules, which has direct implications for optical properties and charge carrier transport. The situation in polymers is somewhat different since the morphology of polymer chains can lead to improved mechanical properties. Nevertheless ...
... delocalization of electronic wavefunctions amongst neighboring molecules, which has direct implications for optical properties and charge carrier transport. The situation in polymers is somewhat different since the morphology of polymer chains can lead to improved mechanical properties. Nevertheless ...
Chapter 4: Chemical Reaction Dynamics
... 4.4.2 Effect of vibrational and kinetic energy: Polanyi rules For asymmetric reactions, the transition state is usually located closer to either the reactant or the products (early or late barrier). From an inspection of the favourable reaction trajectories, it can be seen that: For an early barrie ...
... 4.4.2 Effect of vibrational and kinetic energy: Polanyi rules For asymmetric reactions, the transition state is usually located closer to either the reactant or the products (early or late barrier). From an inspection of the favourable reaction trajectories, it can be seen that: For an early barrie ...
Week 10 Day 2-Plastics - Washington State University
... strain • Damping—ability to absorb energy Properties are often temperature and rate dependent (Viscoelastic) ...
... strain • Damping—ability to absorb energy Properties are often temperature and rate dependent (Viscoelastic) ...
Ideal two-dimensional electron systems with a giant Rashba
... almost the same band structures as FLEUR. In the case of BiTeI, the obtained electronic bands are in good agreement with earlier WIEN-code calculations [19]: both the conduction band minimum (CBM) and the valence band maximum (VBM) demonstrate giant Rashba-type spin splitting with kR ≈ 0.055 Å-1 in ...
... almost the same band structures as FLEUR. In the case of BiTeI, the obtained electronic bands are in good agreement with earlier WIEN-code calculations [19]: both the conduction band minimum (CBM) and the valence band maximum (VBM) demonstrate giant Rashba-type spin splitting with kR ≈ 0.055 Å-1 in ...