6 Chemical Bonding – Orbital Theory
... terms of the orbital theory of atomic structure. Heitler and London believed that electron cloud of the valence orbital on one atom ‘overlaps’ the electron cloud of the other bonding atom to form a covalent linkage. On the contrary, the electrovalent bond formation involves a physical transfer of th ...
... terms of the orbital theory of atomic structure. Heitler and London believed that electron cloud of the valence orbital on one atom ‘overlaps’ the electron cloud of the other bonding atom to form a covalent linkage. On the contrary, the electrovalent bond formation involves a physical transfer of th ...
Bounding the plastic strength of polycrystalline solids by linear
... minimum energy principles, and make use of one-point microstructural statistics only. They have proved useful in the context of high-symmetry polycrystalline solids—like face-centred cubic solids—where the heterogeneity contrast is low, but as crystal anisotropy increases their predictions diverge a ...
... minimum energy principles, and make use of one-point microstructural statistics only. They have proved useful in the context of high-symmetry polycrystalline solids—like face-centred cubic solids—where the heterogeneity contrast is low, but as crystal anisotropy increases their predictions diverge a ...
Test
... Given 1.00 mole of HF(g), 0.386 mole of H2(g), and 0.750 mole of F2(g) are mixed in a 5.00-L flask, determine the reaction quotient, Q. a) b) c) d) e) ...
... Given 1.00 mole of HF(g), 0.386 mole of H2(g), and 0.750 mole of F2(g) are mixed in a 5.00-L flask, determine the reaction quotient, Q. a) b) c) d) e) ...
13 CHEMICAL EQUILIBRIUM W MODULE - 5
... finely is a two phase system. This is because particles of the two solids have different chemical compositions and physical properties. Completely miscible liquids, solutions and all gaseous mixture constitute only one phase each. All the cases of physical equilibrium are in fact the systems in whic ...
... finely is a two phase system. This is because particles of the two solids have different chemical compositions and physical properties. Completely miscible liquids, solutions and all gaseous mixture constitute only one phase each. All the cases of physical equilibrium are in fact the systems in whic ...
Nanoporous molecular crystals - ORCA
... Nanoporous Molecular Crystals (NMCs) are composed of discrete molecules between which there are only non-covalent interactions—i.e. they do not possess an extended framework composed of covalent or coordination bonds. Such materials combine nanoporosity with the ability to be dissolved and then reas ...
... Nanoporous Molecular Crystals (NMCs) are composed of discrete molecules between which there are only non-covalent interactions—i.e. they do not possess an extended framework composed of covalent or coordination bonds. Such materials combine nanoporosity with the ability to be dissolved and then reas ...
The birth of topological insulators
... the next-generation topological insulators is closely related to the Dirac electronic structure of graphene, which has a linear energy–momentum relationship like that of a relativistic particle (and is known as a Dirac cone). Graphene, which consists of a single layer of carbon atoms, has been an ex ...
... the next-generation topological insulators is closely related to the Dirac electronic structure of graphene, which has a linear energy–momentum relationship like that of a relativistic particle (and is known as a Dirac cone). Graphene, which consists of a single layer of carbon atoms, has been an ex ...
The birth of topological insulators
... the next-generation topological insulators is closely related to the Dirac electronic structure of graphene, which has a linear energy–momentum relationship like that of a relativistic particle (and is known as a Dirac cone). Graphene, which consists of a single layer of carbon atoms, has been an ex ...
... the next-generation topological insulators is closely related to the Dirac electronic structure of graphene, which has a linear energy–momentum relationship like that of a relativistic particle (and is known as a Dirac cone). Graphene, which consists of a single layer of carbon atoms, has been an ex ...
Tunneling Transistors Based on Graphene and 2
... Fig. 1 is a schematic representation of the structure of crystals of various dimensions. The bottom row shows the atomic building blocks. The first column shows the ubiquitous 3-D crystal semiconductors. The second column shows the emerging family of 2-D crystals and their many variants. The third a ...
... Fig. 1 is a schematic representation of the structure of crystals of various dimensions. The bottom row shows the atomic building blocks. The first column shows the ubiquitous 3-D crystal semiconductors. The second column shows the emerging family of 2-D crystals and their many variants. The third a ...
The physics of manganites: Structure and transport
... Magnetic systems of great potential are those with a limited ability to transport electricity in zero field, resulting from competing dissimilar ground states. In these systems, magnetic fields produce truly dramatic effects by inducing phase transitions or increasing the temperature of already exis ...
... Magnetic systems of great potential are those with a limited ability to transport electricity in zero field, resulting from competing dissimilar ground states. In these systems, magnetic fields produce truly dramatic effects by inducing phase transitions or increasing the temperature of already exis ...
2008 Equilibrium -- without math (PowerPoint 13 MB)
... Chemical Equilibrium • Few chemical reactions proceed in only one direction. Most are reversible, at least to some extent. At the start of a reversible process, the reaction proceeds toward the formation of products. As soon as some product molecules are formed, the reverse process begins to take p ...
... Chemical Equilibrium • Few chemical reactions proceed in only one direction. Most are reversible, at least to some extent. At the start of a reversible process, the reaction proceeds toward the formation of products. As soon as some product molecules are formed, the reverse process begins to take p ...
Document
... Ammonia is prepared industrially by the reaction: N2(g) + 3H2(g) 2NH3(g) For the reaction, H° = – 92.2 kJ and K (at 25°C) = 4.0 × 108. When the temperature of the reaction is increased to 500°C, which of the following is true? A) K for the reaction will be larger at 500°C than at 25°C. B) At equili ...
... Ammonia is prepared industrially by the reaction: N2(g) + 3H2(g) 2NH3(g) For the reaction, H° = – 92.2 kJ and K (at 25°C) = 4.0 × 108. When the temperature of the reaction is increased to 500°C, which of the following is true? A) K for the reaction will be larger at 500°C than at 25°C. B) At equili ...
Physical and chemical properties of glycine sodium nitrate
... Energy dispersive X-ray (EDX) spectroscopy is used for finding elemental composition in the samples. When the sample is bombarded by the Scanning Electron Microscope’s (SEM) electron beam, electrons are ejected from the atoms comprising the sample's surface. The resulting electron vacancies are fill ...
... Energy dispersive X-ray (EDX) spectroscopy is used for finding elemental composition in the samples. When the sample is bombarded by the Scanning Electron Microscope’s (SEM) electron beam, electrons are ejected from the atoms comprising the sample's surface. The resulting electron vacancies are fill ...
Ionic Compound Solubility Nitrates (NO3 ) Ionic Compound
... Patterns of Chemical Reactivity Definition A substance that is able to donate a H+ ion (a proton) and, hence, increases the concentration of H+(aq) when it dissolves in water. ...
... Patterns of Chemical Reactivity Definition A substance that is able to donate a H+ ion (a proton) and, hence, increases the concentration of H+(aq) when it dissolves in water. ...
Equilibrium Booklet - mrstorie
... 3. For the reaction: 9.4 kJ + 2 HI(g) H2(g) + I2(g) a) What is the effect on [HI] if a small amount of H2 is added? b) What is the effect on [HI] if the pressure of the system is increased? c) What is the effect on [HI] if the temperature is increased? d) What is the effect on [HI] if a catalyst ...
... 3. For the reaction: 9.4 kJ + 2 HI(g) H2(g) + I2(g) a) What is the effect on [HI] if a small amount of H2 is added? b) What is the effect on [HI] if the pressure of the system is increased? c) What is the effect on [HI] if the temperature is increased? d) What is the effect on [HI] if a catalyst ...
OC 583- ISOTOPE BIGEOCHEMISTRY
... 3. Since isotopes have the same number of protons and electrons this means isotopes have same chemical behavior, i.e., all isotopic species enter into same chemical reactions and form the same bonds -in this sense, the rare or less abundant isotopic species is an excellent tracer of the abundant iso ...
... 3. Since isotopes have the same number of protons and electrons this means isotopes have same chemical behavior, i.e., all isotopic species enter into same chemical reactions and form the same bonds -in this sense, the rare or less abundant isotopic species is an excellent tracer of the abundant iso ...
IOSR Journal of Applied Physics (IOSRJAP)
... optical second harmonic generation (SHG) because of their applications in opto-electronics and photonics [1, 2]. They have wide applications in the field of telecommunications and optical information storage devices. Amino acid family crystals are promising materials for these applications. In recen ...
... optical second harmonic generation (SHG) because of their applications in opto-electronics and photonics [1, 2]. They have wide applications in the field of telecommunications and optical information storage devices. Amino acid family crystals are promising materials for these applications. In recen ...
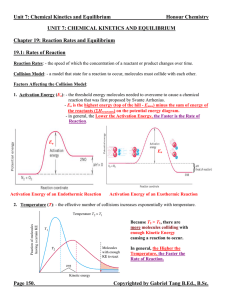
Unit 7 Reaction Rates and Equilibrium Notes
... Equilibrium Position: - the concentrations or pressures of all chemical species at equilibrium state. - depends strongly on the Initial Concentrations of the chemical species. (In contrast, K does NOT depend on initial concentrations, only on temperature and the specific reaction.) - since there all ...
... Equilibrium Position: - the concentrations or pressures of all chemical species at equilibrium state. - depends strongly on the Initial Concentrations of the chemical species. (In contrast, K does NOT depend on initial concentrations, only on temperature and the specific reaction.) - since there all ...
Unit 5: Chemical Kinetics and Equilibrium
... a. At 400 K, PNH3 = 0.1024 atm, PN 2 = 2.8084 atm and PH 2 = 0.0102 atm. Write the equilibrium expression in terms of pressure and calculate KP. b. Convert the calculated KP above to K. a. Equilibrium Expression: ...
... a. At 400 K, PNH3 = 0.1024 atm, PN 2 = 2.8084 atm and PH 2 = 0.0102 atm. Write the equilibrium expression in terms of pressure and calculate KP. b. Convert the calculated KP above to K. a. Equilibrium Expression: ...
Chap15 - Bakersfield College
... Chemical Equilibrium • When compounds react, they eventually form a mixture of products and unused reactants, in a dynamic equilibrium. – A dynamic equilibrium consists of a forward reaction, in which substances react to give products, and a reverse reaction, in which products react to give the ori ...
... Chemical Equilibrium • When compounds react, they eventually form a mixture of products and unused reactants, in a dynamic equilibrium. – A dynamic equilibrium consists of a forward reaction, in which substances react to give products, and a reverse reaction, in which products react to give the ori ...
Asp_calcite_FCM_Main_NatMat_Symplectic author accepted
... expanding the crystal axes independently. The calculated configurational energies rose much faster when the crystal was strained along the a-axis than the c-axis (Fig. 5c), as is consistent with the elastic anisotropy of calcite35. For crystals containing 2.3 mol% Asp2- and 2.8 mol% Gly0 (to corresp ...
... expanding the crystal axes independently. The calculated configurational energies rose much faster when the crystal was strained along the a-axis than the c-axis (Fig. 5c), as is consistent with the elastic anisotropy of calcite35. For crystals containing 2.3 mol% Asp2- and 2.8 mol% Gly0 (to corresp ...
Peter Ertl - American Chemical Society
... where nacti is the number of drugs which contain substituent i, ninacti is the number of nondrugs which contain substituent i, nacttotal is the total number of drugs, and ninacttotal is the total number of nondrugs processed. In several in-house studies we compared the performance of various indices ...
... where nacti is the number of drugs which contain substituent i, ninacti is the number of nondrugs which contain substituent i, nacttotal is the total number of drugs, and ninacttotal is the total number of nondrugs processed. In several in-house studies we compared the performance of various indices ...
Introduction to Alloy Phase Diagrams
... is a hypereutectic alloy (meaning "greater than"). In the eutectic system described above, the two components of the system have the same crystal structure. This, and other factors, allows complete miscibilitybetween them. Eutectic systems, however, also can be formed by two components having differ ...
... is a hypereutectic alloy (meaning "greater than"). In the eutectic system described above, the two components of the system have the same crystal structure. This, and other factors, allows complete miscibilitybetween them. Eutectic systems, however, also can be formed by two components having differ ...
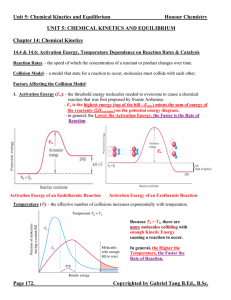
Chapter 14: Chemical Equilibrium
... as being in a state of dynamic equilibrium? What are the characteristics and requirements of dynamic equilibrium? ...
... as being in a state of dynamic equilibrium? What are the characteristics and requirements of dynamic equilibrium? ...
A preliminary DFT study of the adsorption and dissociation of $ CH_
... to form hydroxyl species, methyl groups adsorbed on sulfur and/or methoxy group (R − O − CH3 ) atoms. SO2 adsorption leads to the formation of sulfite species involving surface or preadsorbed oxygen atoms. No sulfate species formation was observed. O2 adsorption in the presence of SO2 favors oxygen ...
... to form hydroxyl species, methyl groups adsorbed on sulfur and/or methoxy group (R − O − CH3 ) atoms. SO2 adsorption leads to the formation of sulfite species involving surface or preadsorbed oxygen atoms. No sulfate species formation was observed. O2 adsorption in the presence of SO2 favors oxygen ...
IOSR Journal of Applied Physics (IOSR-JAP)
... a function of frequency at temperatures 373 K, 393 K and 413 K respectively (Fig 4 and Fig 5). The high value of dielectric constant at low frequency can be attributed to the presence of all four types of polarizations namely ionic, electronic, space-charge and orientation and the low value at highe ...
... a function of frequency at temperatures 373 K, 393 K and 413 K respectively (Fig 4 and Fig 5). The high value of dielectric constant at low frequency can be attributed to the presence of all four types of polarizations namely ionic, electronic, space-charge and orientation and the low value at highe ...