Chemistry Quiz #2 Study Guide (Answers)
... • Subscript – The small number in a formula that identifies the number of atoms in each molecule. This cannot be changed. • Coefficient – The larger number in front of a formula that identifies the number of molecules of that specific substance. This can be changed. • Endothermic Reaction – Energy ( ...
... • Subscript – The small number in a formula that identifies the number of atoms in each molecule. This cannot be changed. • Coefficient – The larger number in front of a formula that identifies the number of molecules of that specific substance. This can be changed. • Endothermic Reaction – Energy ( ...
Let’s talk Chemistry!
... During a chemical or physical change, energy may be Converted into another form A liquid changes rapidly into a gas at the liquid’s Boiling point Knowing the chemical properties of a substance will tell you how the substance Reacts with other substances Physical properties are Properties that can b ...
... During a chemical or physical change, energy may be Converted into another form A liquid changes rapidly into a gas at the liquid’s Boiling point Knowing the chemical properties of a substance will tell you how the substance Reacts with other substances Physical properties are Properties that can b ...
Week # 9: Organic Chemistry - Hicksville Public Schools / Homepage
... The hydrocarbons in petroleum are separated from each other by distillation in a “cracking tower,” on the basis of boiling points. The greater the molar mass, the higher the intermolecular forces of attraction between molecules. As a result, melting points and boiling points are higher. EX: Octa ...
... The hydrocarbons in petroleum are separated from each other by distillation in a “cracking tower,” on the basis of boiling points. The greater the molar mass, the higher the intermolecular forces of attraction between molecules. As a result, melting points and boiling points are higher. EX: Octa ...
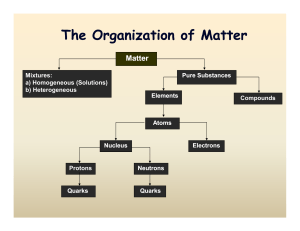
The Organization The Organization of Matter of Matter
... Solid – definite volume and shape; particles packed in fixed positions. Liquid – definite volume but indefinite shape; particles close together but not in fixed positions Gas – neither definite volume nor definite shape; particles are at great distances from one another Plasma – high temperature, io ...
... Solid – definite volume and shape; particles packed in fixed positions. Liquid – definite volume but indefinite shape; particles close together but not in fixed positions Gas – neither definite volume nor definite shape; particles are at great distances from one another Plasma – high temperature, io ...
Spectroscopy, a toolbox for structural information on aerosol particles
... CH-8093 Zurich, Switzerland [email protected] It is well known that light interacts differently with small particles compared with bulk materials or gas phase molecules, producing spectral signatures that strongly depend on particle properties, such as size, shape, or architecture. Even nanosized a ...
... CH-8093 Zurich, Switzerland [email protected] It is well known that light interacts differently with small particles compared with bulk materials or gas phase molecules, producing spectral signatures that strongly depend on particle properties, such as size, shape, or architecture. Even nanosized a ...
Notes on Atoms and Molecules
... A molecule which contains two atoms is called diatomic Example: Hydrogen (H2), Chlorine (Cl2), Nitrogen (N2) etc. Valency: The combining capacity of an element is known as valency. The combining capacity of the atoms to form molecules either with same or different elements is defined as valency. Ato ...
... A molecule which contains two atoms is called diatomic Example: Hydrogen (H2), Chlorine (Cl2), Nitrogen (N2) etc. Valency: The combining capacity of an element is known as valency. The combining capacity of the atoms to form molecules either with same or different elements is defined as valency. Ato ...
Unit 5 Objectives
... Unit 5 – Counting Particles - Objectives Review Concepts a. Types of substances b. Chemical formulas of substances (U 4) 1. State evidence for Avogadro’s Hypothesis. Use Avogadro’s Hypothesis and experimental data to determine the relative mass of molecules. ...
... Unit 5 – Counting Particles - Objectives Review Concepts a. Types of substances b. Chemical formulas of substances (U 4) 1. State evidence for Avogadro’s Hypothesis. Use Avogadro’s Hypothesis and experimental data to determine the relative mass of molecules. ...
The Mole - ETSU.edu
... is a formula unit of an ionic compound? Formula unit is the unit that represents the simplest ratio of cations to anions. A formula unit of NaCl consists of one Na+ and one Cl One mole of an ionic compound consists of 6.022х10²³ of these formula units. One mole of a molecular compound, like H₂O c ...
... is a formula unit of an ionic compound? Formula unit is the unit that represents the simplest ratio of cations to anions. A formula unit of NaCl consists of one Na+ and one Cl One mole of an ionic compound consists of 6.022х10²³ of these formula units. One mole of a molecular compound, like H₂O c ...
section_3.2
... To understand and illustrate the Law of constant composition To learn how a formula describes a compound’s composition ...
... To understand and illustrate the Law of constant composition To learn how a formula describes a compound’s composition ...
View - Workshops+SJCOE Workshop Management
... By the end of grade 2. Different kinds of matter exist (e.g., wood, metal, water), and many of them can be either solid or liquid, depending on temperature. Matter can be described and classified by its observable properties (e.g., visual, aural, textural), by its uses, and by whether it occurs natu ...
... By the end of grade 2. Different kinds of matter exist (e.g., wood, metal, water), and many of them can be either solid or liquid, depending on temperature. Matter can be described and classified by its observable properties (e.g., visual, aural, textural), by its uses, and by whether it occurs natu ...
Chemistry Scavenger Hunt
... material of the _______________. 2. There are three main phases of matter: _____________, ____________, and _____________. There is also a fourth phase, ______________, but it exists at very high temperatures. Science Is Fun 2 Go to the “ChemTime Clock” area to find the answers to these questions. h ...
... material of the _______________. 2. There are three main phases of matter: _____________, ____________, and _____________. There is also a fourth phase, ______________, but it exists at very high temperatures. Science Is Fun 2 Go to the “ChemTime Clock” area to find the answers to these questions. h ...
Document
... • Equal volumes of different gases at the same T and P contain equal numbers of molecules (or, equal numbers of moles of gas). Neglecting history (Avogadro’s elegant experiments!), we can apply the Ideal Gas Law Equation to two gases (Gas 1 and Gas 2). • n1 = P1V1/RT1 and n2 = P2V2/RT2 • If P1=P2 an ...
... • Equal volumes of different gases at the same T and P contain equal numbers of molecules (or, equal numbers of moles of gas). Neglecting history (Avogadro’s elegant experiments!), we can apply the Ideal Gas Law Equation to two gases (Gas 1 and Gas 2). • n1 = P1V1/RT1 and n2 = P2V2/RT2 • If P1=P2 an ...
Chemical Reactions
... 2. Find the number of atoms for each element on the left side. Compare those against the number of the atoms of the same element on the right side. 3. Determine where to place coefficients in front of formulas so that the left side has the same number of atoms as the right side for EACH element in o ...
... 2. Find the number of atoms for each element on the left side. Compare those against the number of the atoms of the same element on the right side. 3. Determine where to place coefficients in front of formulas so that the left side has the same number of atoms as the right side for EACH element in o ...
Mole Unit Review Packet
... I hope you all had a wonderful Memorial Day weekend. Sadly I am sick today and cannot make it to school. In my absence, you will practice some of the topics that we have learned recently. This packet is due in class on WEDNESDAY, May 28 (tomorrow). ...
... I hope you all had a wonderful Memorial Day weekend. Sadly I am sick today and cannot make it to school. In my absence, you will practice some of the topics that we have learned recently. This packet is due in class on WEDNESDAY, May 28 (tomorrow). ...
Unit 11: The Mole
... MOLAR MASS The molar mass of an element is the mass in grams of one mole of any pure substance. ...
... MOLAR MASS The molar mass of an element is the mass in grams of one mole of any pure substance. ...
Unit 1B1 - Uddingston Grammar School
... Atoms P and Q have the same number of protons Atoms Q and R have the same number of electrons Atoms P and S have the same number of neutrons Atoms R and S are isotopes of each other Atoms S and T have different chemical properties. ...
... Atoms P and Q have the same number of protons Atoms Q and R have the same number of electrons Atoms P and S have the same number of neutrons Atoms R and S are isotopes of each other Atoms S and T have different chemical properties. ...
POGIL: Kinetic Molecular Theory
... Gases consist of tiny particles (atoms or molecules). These particles are so small compared with the distance between them that the volume (size) of the individual particles can be assumed to be negligible (zero). The particles are in constant random motion, colliding with the walls of the container ...
... Gases consist of tiny particles (atoms or molecules). These particles are so small compared with the distance between them that the volume (size) of the individual particles can be assumed to be negligible (zero). The particles are in constant random motion, colliding with the walls of the container ...
Bose–Einstein condensate

A Bose–Einstein condensate (BEC) is a state of matter of a dilute gas of bosons cooled to temperatures very close to absolute zero (that is, very near 5000000000000000000♠0 K or 5000000000000000000♠−273.15 °C). Under such conditions, a large fraction of bosons occupy the lowest quantum state, at which point macroscopic quantum phenomena become apparent.This state was first predicted, generally, in 1924–25 by Satyendra Nath Bose and Albert Einstein.