Chapter 3: Mass Relationships in Chemical Reactions – Khan
... I. The Mole A The mole is a unit of measure that measures quantity. It is the same concept as a dozen. You can have a dozen doughnuts, oranges, golf balls, etc. and you always have a quantity of 12. The same goes for a mole, we can have a mole of doughnuts, oranges or golf balls and would always hav ...
... I. The Mole A The mole is a unit of measure that measures quantity. It is the same concept as a dozen. You can have a dozen doughnuts, oranges, golf balls, etc. and you always have a quantity of 12. The same goes for a mole, we can have a mole of doughnuts, oranges or golf balls and would always hav ...
Molecular Mass - Teacher Notes
... How many molecules are in 3.5 moles of H2O? How many moles are present in 465 molecules of NO2? How many atoms of nitrogen are in 3.15 moles of NH3? How many atoms of chlorine are in .862 moles of MgCl2? ...
... How many molecules are in 3.5 moles of H2O? How many moles are present in 465 molecules of NO2? How many atoms of nitrogen are in 3.15 moles of NH3? How many atoms of chlorine are in .862 moles of MgCl2? ...
- Bright Star Schools
... 2. Describe what an atom looks like in your own words (use the vocabulary) ...
... 2. Describe what an atom looks like in your own words (use the vocabulary) ...
Name: Chapter 3 Reading Guide: Molecules, Compounds, and
... 3.5 Ionic Compounds: Formulas and Names (p. 95-101) Ionic compounds always contain ___________________________________________. In a chemical formula, the sum of the charges of the positive ions (______________________) must equal the ____________ of the charges of the negative ions (______________ ...
... 3.5 Ionic Compounds: Formulas and Names (p. 95-101) Ionic compounds always contain ___________________________________________. In a chemical formula, the sum of the charges of the positive ions (______________________) must equal the ____________ of the charges of the negative ions (______________ ...
Chapter 11.1
... who studied quantities of gases and discovered that no matter what the gas was, there were the same number of molecules present ...
... who studied quantities of gases and discovered that no matter what the gas was, there were the same number of molecules present ...
Module 1 in 10 minutes
... There is a persistent current in the superconductor that causes a magnetic field to be set up that repels the magnetic field of the permanent magnet. ...
... There is a persistent current in the superconductor that causes a magnetic field to be set up that repels the magnetic field of the permanent magnet. ...
Properties of Matter - Red Clay Secondary Science Wiki
... arrangement, motion, and interaction of these particles determine the three states of matter (solid, liquid, and gas). Particles in all three states are in constant motion. In the solid state, tightly packed particles have a limited range of motion. In the liquid state, particles are loosely packed ...
... arrangement, motion, and interaction of these particles determine the three states of matter (solid, liquid, and gas). Particles in all three states are in constant motion. In the solid state, tightly packed particles have a limited range of motion. In the liquid state, particles are loosely packed ...
Defining the Atom
... To convert from grams to moles use the formula: 1 mol moles = number of grams x -------------------------------Gram formula mass ...
... To convert from grams to moles use the formula: 1 mol moles = number of grams x -------------------------------Gram formula mass ...
Zumdahl Chapter
... First Year Chemistry Podcast DVD Featuring Jonathan Bergmann and Aaron Sams from Peak Educational Consulting LLC All Rights Reserved © This is an interactive page that allows you to get to all of the content on this DVD. Click to each unit packet or podcast. The podcasts require Quicktime and the pa ...
... First Year Chemistry Podcast DVD Featuring Jonathan Bergmann and Aaron Sams from Peak Educational Consulting LLC All Rights Reserved © This is an interactive page that allows you to get to all of the content on this DVD. Click to each unit packet or podcast. The podcasts require Quicktime and the pa ...
Lecture 8
... A 30.5-g sample of acrylic acid, used in the manufacture of acrylic plastics, is found to contain 15.25 g C, 1.71 g H, and 13.54 g O. In a separate mass spectrometer experiment, the acrylic acid is found to have a molar mass of approximately 72 g/mol. What are the empirical and molecular formulas of ...
... A 30.5-g sample of acrylic acid, used in the manufacture of acrylic plastics, is found to contain 15.25 g C, 1.71 g H, and 13.54 g O. In a separate mass spectrometer experiment, the acrylic acid is found to have a molar mass of approximately 72 g/mol. What are the empirical and molecular formulas of ...
Document
... mass (Ar) of the element. It allows chemists to calculate chemical formulae The relative atomic mass scale is used to compare the masses of different atoms. The reference that used is the carbon-12 isotope (C12) which is assigned a relative atomic mass of exactly 12 every other atom is measured agai ...
... mass (Ar) of the element. It allows chemists to calculate chemical formulae The relative atomic mass scale is used to compare the masses of different atoms. The reference that used is the carbon-12 isotope (C12) which is assigned a relative atomic mass of exactly 12 every other atom is measured agai ...
Chapter 3 - pennsaukenchemistry2
... The formula of a compound that expresses the _________________ ratio of the atoms present. Ionic formula are always ____________ formula • _______ Formula The formula that states the _____ number of each kind of atom found in _________ of the compound. ...
... The formula of a compound that expresses the _________________ ratio of the atoms present. Ionic formula are always ____________ formula • _______ Formula The formula that states the _____ number of each kind of atom found in _________ of the compound. ...
Chem 1a Midterm Review
... Boundary conditions: (r) 0 as r (this is same as standing wave and makes probability finite) and (r) ; at all times (r)2 dV is probability of finding an electron in small volume dV around r Only solutions with specific values of the quantum numbers, n, l, ml (and ms added later ...
... Boundary conditions: (r) 0 as r (this is same as standing wave and makes probability finite) and (r) ; at all times (r)2 dV is probability of finding an electron in small volume dV around r Only solutions with specific values of the quantum numbers, n, l, ml (and ms added later ...
Chemical Foundations: Elements, Atoms and Ions
... Elements are made of tiny particles called atoms All atoms of a given element are identical Atoms of a given element are different from those of any other element 4. Atoms of one element can combine with atoms of other elements to form compounds 5. Atoms are indivisible and can not be created or des ...
... Elements are made of tiny particles called atoms All atoms of a given element are identical Atoms of a given element are different from those of any other element 4. Atoms of one element can combine with atoms of other elements to form compounds 5. Atoms are indivisible and can not be created or des ...
Catalyst Activity (in your notebook)
... • specifies the relative number of moles and molecules involved in the reaction • used to balance the equation ...
... • specifies the relative number of moles and molecules involved in the reaction • used to balance the equation ...
111 Exam I Outline
... When most reactions are performed, some of the reactants is usually present in excess of the amount needed. If the reaction goes to completion, then some of this excess reactant will be left-over. The limiting reactant is the reactant used-up completely and it "limits" the reaction. For example: ...
... When most reactions are performed, some of the reactants is usually present in excess of the amount needed. If the reaction goes to completion, then some of this excess reactant will be left-over. The limiting reactant is the reactant used-up completely and it "limits" the reaction. For example: ...
Pre-AP Chemistry Kinetic Theory and Heat Quiz
... 1. According to the kinetic theory, the primary difference in the phases of matter (of the same substance) is the __speed_ of the particles. Thus, the two factors that influence which state of matter exists are __pressure_ and temperature. 2. The higher the energy of the particles, the faster they m ...
... 1. According to the kinetic theory, the primary difference in the phases of matter (of the same substance) is the __speed_ of the particles. Thus, the two factors that influence which state of matter exists are __pressure_ and temperature. 2. The higher the energy of the particles, the faster they m ...
Section 3.2 Atoms and Compounds
... which the number and kind of atoms present is shown by using the element symbols and subscripts. Example: the simple sugar, glucose ...
... which the number and kind of atoms present is shown by using the element symbols and subscripts. Example: the simple sugar, glucose ...
Chapter 2 cont’
... when atoms gain or lose electrons, they acquire a charge charged particles are called ions when atoms gain electrons, they become negatively charged ions, ...
... when atoms gain or lose electrons, they acquire a charge charged particles are called ions when atoms gain electrons, they become negatively charged ions, ...
CYL110
... When a system is at equilibrium, its state is defined entirely by the state variables, and not by the history of the system. The properties of the system can be described by an equation of state which specifies the relationship between these variables. ...
... When a system is at equilibrium, its state is defined entirely by the state variables, and not by the history of the system. The properties of the system can be described by an equation of state which specifies the relationship between these variables. ...
Solid State Physics
... When atoms are brought close together, an electron state is created for each core state of an atom – some bonding (lower energy) and some antibonding (higher energy). These states are all occupied. Good thing we have the Pauli exclusion principle. Atoms are attracted by the outer electrons, occupyin ...
... When atoms are brought close together, an electron state is created for each core state of an atom – some bonding (lower energy) and some antibonding (higher energy). These states are all occupied. Good thing we have the Pauli exclusion principle. Atoms are attracted by the outer electrons, occupyin ...
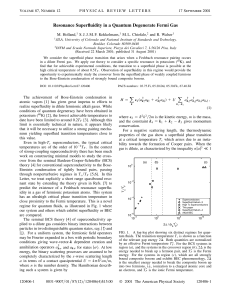
Resonance Superfluidity in a Quantum Degenerate Fermi Gas
... atomic resonance. For a temperature above Tc , the grand potential surface is shaped like a bowl, and the value of fm which minimizes the grand potential is fm 苷 0, associated with the self-consistent solution p 苷 0. For T , Tc , the grand potential surface is shaped like a Mexican hat, and its mini ...
... atomic resonance. For a temperature above Tc , the grand potential surface is shaped like a bowl, and the value of fm which minimizes the grand potential is fm 苷 0, associated with the self-consistent solution p 苷 0. For T , Tc , the grand potential surface is shaped like a Mexican hat, and its mini ...
Unit 6 Study Guide – Chemical Bonding 1. A _ chemical
... 22. An __ionic____________ compound is composed of positive and negative ions that are combined so that the numbers of positive and negative charges are equal. 23. Most ionic compounds exist as ___crystalline___________ __solids______________. 24. What does a formula unit of an ionic compound indic ...
... 22. An __ionic____________ compound is composed of positive and negative ions that are combined so that the numbers of positive and negative charges are equal. 23. Most ionic compounds exist as ___crystalline___________ __solids______________. 24. What does a formula unit of an ionic compound indic ...
Bose–Einstein condensate

A Bose–Einstein condensate (BEC) is a state of matter of a dilute gas of bosons cooled to temperatures very close to absolute zero (that is, very near 5000000000000000000♠0 K or 5000000000000000000♠−273.15 °C). Under such conditions, a large fraction of bosons occupy the lowest quantum state, at which point macroscopic quantum phenomena become apparent.This state was first predicted, generally, in 1924–25 by Satyendra Nath Bose and Albert Einstein.