World of matter - Kindle Education
... What happens on heating Matter is made up on small particles which are in constant motion. When you heat matter, the particles of matter absorb the heat energy and begin moving faster (in other words they gain kinetic energy). As more energy is provided the chemical bond between the particles becom ...
... What happens on heating Matter is made up on small particles which are in constant motion. When you heat matter, the particles of matter absorb the heat energy and begin moving faster (in other words they gain kinetic energy). As more energy is provided the chemical bond between the particles becom ...
chemistry_chapter_3
... the mole allows us to count microscopic items (atoms, ion, molecules) on a macroscopic scale. So, 1 mole of any substance is a set number of Items, namely: 6.02 x 1023. ...
... the mole allows us to count microscopic items (atoms, ion, molecules) on a macroscopic scale. So, 1 mole of any substance is a set number of Items, namely: 6.02 x 1023. ...
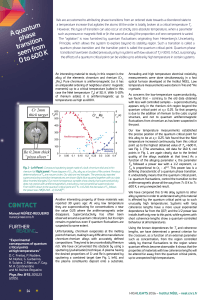
A quantum phase transition seen from 0 to 600 K
... quantum critical point at xc= 0.25. So that property is due to the addition of rhenium to the cubic crystal structure, and not to quantum antiferromagnetic fluctuations from chromium as has been suspected in the past. Our low temperature measurements established the precise position of the quantum c ...
... quantum critical point at xc= 0.25. So that property is due to the addition of rhenium to the cubic crystal structure, and not to quantum antiferromagnetic fluctuations from chromium as has been suspected in the past. Our low temperature measurements established the precise position of the quantum c ...
Chapter 06 Notes (PowerPoint) File
... • If we can find the mass of a particular number of atoms, we can use this information to convert the mass of a element sample to the number of atoms in the sample. • The number of atoms we will use is 6.022 x 1023 and we call this a mole 1 mole = 6.022 x 1023 things Like 1 dozen = 12 things ...
... • If we can find the mass of a particular number of atoms, we can use this information to convert the mass of a element sample to the number of atoms in the sample. • The number of atoms we will use is 6.022 x 1023 and we call this a mole 1 mole = 6.022 x 1023 things Like 1 dozen = 12 things ...
3. Moles and Formulae
... Equal volumes of different gases at the same pressure and temperature will contain equal numbers of particles. For example, if there are 2 moles of O2 in 50 cm3 of oxygen gas, then there will be 2 moles of N2 in 50 cm3 of nitrogen gas and 2 moles of CO2 in 50 cm3 of carbon dioxide gas at the same te ...
... Equal volumes of different gases at the same pressure and temperature will contain equal numbers of particles. For example, if there are 2 moles of O2 in 50 cm3 of oxygen gas, then there will be 2 moles of N2 in 50 cm3 of nitrogen gas and 2 moles of CO2 in 50 cm3 of carbon dioxide gas at the same te ...
3. Moles and Formulae
... Equal volumes of different gases at the same pressure and temperature will contain equal numbers of particles. For example, if there are 2 moles of O2 in 50 cm3 of oxygen gas, then there will be 2 moles of N2 in 50 cm3 of nitrogen gas and 2 moles of CO2 in 50 cm3 of carbon dioxide gas at the same te ...
... Equal volumes of different gases at the same pressure and temperature will contain equal numbers of particles. For example, if there are 2 moles of O2 in 50 cm3 of oxygen gas, then there will be 2 moles of N2 in 50 cm3 of nitrogen gas and 2 moles of CO2 in 50 cm3 of carbon dioxide gas at the same te ...
Chapter 8
... • The molar mass is the mass in grams of one mole of a compound • The relative weights of molecules can be calculated from atomic masses water = H2O = 2(1.008 amu) + 16.00 amu = 18.02 amu • 1 mole of H2O will weigh 18.02 g, therefore the molar mass of H2O is 18.02 g • 1 mole of H2O will contain 16.0 ...
... • The molar mass is the mass in grams of one mole of a compound • The relative weights of molecules can be calculated from atomic masses water = H2O = 2(1.008 amu) + 16.00 amu = 18.02 amu • 1 mole of H2O will weigh 18.02 g, therefore the molar mass of H2O is 18.02 g • 1 mole of H2O will contain 16.0 ...
Stoichiometry …like a beautiful sunset on a serene lake – NOT!
... …Kind of like 1 dozen eggs – 12 eggs A mole is the name of a number! The mole is defined as the number of Carbon atoms in exactly 12 grams of pure Carbon – 12 (think the graphite in a pencil!) It was determined that ...
... …Kind of like 1 dozen eggs – 12 eggs A mole is the name of a number! The mole is defined as the number of Carbon atoms in exactly 12 grams of pure Carbon – 12 (think the graphite in a pencil!) It was determined that ...
Chapter 8
... • The molar mass is the mass in grams of one mole of a compound • The relative weights of molecules can be calculated from atomic masses water = H2O = 2(1.008 amu) + 16.00 amu = 18.02 amu • 1 mole of H2O will weigh 18.02 g, therefore the molar mass of H2O is 18.02 g • 1 mole of H2O will contain 16.0 ...
... • The molar mass is the mass in grams of one mole of a compound • The relative weights of molecules can be calculated from atomic masses water = H2O = 2(1.008 amu) + 16.00 amu = 18.02 amu • 1 mole of H2O will weigh 18.02 g, therefore the molar mass of H2O is 18.02 g • 1 mole of H2O will contain 16.0 ...
Lecture 3
... Suppose the amounts of reactants: 15 g of C2H6 and 224 g of O2 Which gives the smallest yield of CO2? 15 g/15 g mol-1 of C2H6 = 1 mole; yield of CO2 = 2 moles of CO2 224 g/32 g mol-1 of O2 = 7 moles; yield of CO2 = 4 moles of CO2 Limiting reactant is C2H6. 1 mole of C2H6 (15 g) reacts with 3.5 mole ...
... Suppose the amounts of reactants: 15 g of C2H6 and 224 g of O2 Which gives the smallest yield of CO2? 15 g/15 g mol-1 of C2H6 = 1 mole; yield of CO2 = 2 moles of CO2 224 g/32 g mol-1 of O2 = 7 moles; yield of CO2 = 4 moles of CO2 Limiting reactant is C2H6. 1 mole of C2H6 (15 g) reacts with 3.5 mole ...
7-1 Avogadro`s Number and Molar Conversions Objectives: • Identify
... b.) To find a monatomic element’s molar mass, use the atomic mass, but instead of having units of amu, the molar mass will have units of g/mol. c.) Board 2. The Mole Plays a Central Part in Chemical Conversions - Board 3. Mass Can Be Converted to Amount in Moles - Board 4. Remember to Round Consiste ...
... b.) To find a monatomic element’s molar mass, use the atomic mass, but instead of having units of amu, the molar mass will have units of g/mol. c.) Board 2. The Mole Plays a Central Part in Chemical Conversions - Board 3. Mass Can Be Converted to Amount in Moles - Board 4. Remember to Round Consiste ...
HO #15 Maxwell Distribution
... oxygen is 16. The molecular weight of oxygen is 32 since two oxygen atoms bind together in ordinary gaseous form. This gives us a value of ...
... oxygen is 16. The molecular weight of oxygen is 32 since two oxygen atoms bind together in ordinary gaseous form. This gives us a value of ...
Chapter 2
... An atom contains 66 electrons. Which element is it? An atom of an element contains 14 protons. Which element is it? ...
... An atom contains 66 electrons. Which element is it? An atom of an element contains 14 protons. Which element is it? ...
O - FIU
... DNA nanoparticles for the delivery of genetic materials, nonviral gene vectors` Drug development and delivery on practice of medicine ...
... DNA nanoparticles for the delivery of genetic materials, nonviral gene vectors` Drug development and delivery on practice of medicine ...
Chemical Composition
... 4. Use this as a conversion factor for moles-to-grams Molar Mass The molar mass is the mass in grams of one mole of a compound The relative weights of molecules can be calculated from atomic masses water = H2O = 2(1.008 amu) + 16.00 amu = 18.02 amu 1 mole of H2O will weigh 18.02 g, therefore ...
... 4. Use this as a conversion factor for moles-to-grams Molar Mass The molar mass is the mass in grams of one mole of a compound The relative weights of molecules can be calculated from atomic masses water = H2O = 2(1.008 amu) + 16.00 amu = 18.02 amu 1 mole of H2O will weigh 18.02 g, therefore ...
Chemistry Unit Notes Organizing the Periodic Table All the elements
... identify it. A physical property does not involve a substance becoming a new substance. Example: colour, texture, odour, lustre, clarity, or taste. ...
... identify it. A physical property does not involve a substance becoming a new substance. Example: colour, texture, odour, lustre, clarity, or taste. ...
Statistical Mechanics Basis of Macleod`s Formula
... change in K with respect to the temperature is small because of the fact that for a given liquid/vapor system the change in temperature is not very important compared to the initial temperature. Near the critical temperature, the surface tension remains as given by eq 14. Nevertheless, the constant ...
... change in K with respect to the temperature is small because of the fact that for a given liquid/vapor system the change in temperature is not very important compared to the initial temperature. Near the critical temperature, the surface tension remains as given by eq 14. Nevertheless, the constant ...
SLIB quantitative chemistry homework
... The pressure of 4.0 L of an ideal gas in a flexible container is decreased to one-third of its original pressure and its absolute temperature is decreased by one-half. The volume then is a) 1.0 L b) 4.0 L c) 6.0 L d) 8.0 L e) 24 L ...
... The pressure of 4.0 L of an ideal gas in a flexible container is decreased to one-third of its original pressure and its absolute temperature is decreased by one-half. The volume then is a) 1.0 L b) 4.0 L c) 6.0 L d) 8.0 L e) 24 L ...
THE UNIVERSITY OF LETHBRIDGE DEPARTMENT OF CHEMISTRY
... easilly polarizable iodine molecule, these dispersion forces are sufficiently strong to cause it to be a solid. c) Remove the water of hydration from MnCl2.4H2O Ion-DP forces dominate, because the water in this formula is most likely coordinated directly to the Mn, and is not merely water of crystal ...
... easilly polarizable iodine molecule, these dispersion forces are sufficiently strong to cause it to be a solid. c) Remove the water of hydration from MnCl2.4H2O Ion-DP forces dominate, because the water in this formula is most likely coordinated directly to the Mn, and is not merely water of crystal ...
Elements, Compounds, and Molecules
... Carbon Dioxide CO2 Carbon dioxide molecules are made from one carbon and two oxygen atoms joined together by covalent bonds. The chemical symbol is CO2. ...
... Carbon Dioxide CO2 Carbon dioxide molecules are made from one carbon and two oxygen atoms joined together by covalent bonds. The chemical symbol is CO2. ...
Ang. bindningstyper och elektronegativitet
... A coordinate covalent bond is one where both bonding electrons are from one of the atoms involved in the bond. These bonds give rise to Lewis acids and bases. The electrons are shared roughly equally between the atoms in contrast to ionic bonding. Such bonding occurs in molecules such as the ammoniu ...
... A coordinate covalent bond is one where both bonding electrons are from one of the atoms involved in the bond. These bonds give rise to Lewis acids and bases. The electrons are shared roughly equally between the atoms in contrast to ionic bonding. Such bonding occurs in molecules such as the ammoniu ...
Chapter 2
... Dalton’s Atomic Theory Examine Dalton's Atomic Theory. ❶All matter is composed of atoms, indivisible particles that are exceedingly small. ❷All atoms of a given element are identical, both in mass and in chemical properties. However, they are different from atoms of other elements. ❸Atoms are not cr ...
... Dalton’s Atomic Theory Examine Dalton's Atomic Theory. ❶All matter is composed of atoms, indivisible particles that are exceedingly small. ❷All atoms of a given element are identical, both in mass and in chemical properties. However, they are different from atoms of other elements. ❸Atoms are not cr ...
Exam 1 Review Sheet
... Use scientific notation correctly (know that the N in N x 10n should be between 1 and 10) Pertinent problems from the book: ...
... Use scientific notation correctly (know that the N in N x 10n should be between 1 and 10) Pertinent problems from the book: ...
AP Unit 1 Test Review
... 8. Represents an atom that is chemically unreactive 9. Represents an atom in an excited state 10. Represents an atom that has four valence electrons. 11. Represents an atom of a transition metal. ...
... 8. Represents an atom that is chemically unreactive 9. Represents an atom in an excited state 10. Represents an atom that has four valence electrons. 11. Represents an atom of a transition metal. ...
The Mole: A Measurement of Matter
... The Mole and Avogadro’s Number SI unit that measures the amount of substance 1 mole = 6.022 x 1023 representative particles Representative particles are usually atoms, molecules, or formula units (ions) ...
... The Mole and Avogadro’s Number SI unit that measures the amount of substance 1 mole = 6.022 x 1023 representative particles Representative particles are usually atoms, molecules, or formula units (ions) ...
Bose–Einstein condensate

A Bose–Einstein condensate (BEC) is a state of matter of a dilute gas of bosons cooled to temperatures very close to absolute zero (that is, very near 5000000000000000000♠0 K or 5000000000000000000♠−273.15 °C). Under such conditions, a large fraction of bosons occupy the lowest quantum state, at which point macroscopic quantum phenomena become apparent.This state was first predicted, generally, in 1924–25 by Satyendra Nath Bose and Albert Einstein.