Surface Tension Measurements
... where l is the length along the slide and the factor of “2” is required to account for the film’s two sides. (Note the analogy between the intensive parameter , defined as a force per unit length, and the intensive parameter pressure, defined as a force per area.) Then we relate the surface tension ...
... where l is the length along the slide and the factor of “2” is required to account for the film’s two sides. (Note the analogy between the intensive parameter , defined as a force per unit length, and the intensive parameter pressure, defined as a force per area.) Then we relate the surface tension ...
Unit 3: Properties and States of Matter
... • You and your lab partner must go around to each station and identify whether the property being demonstrated is a physical or chemical property and state why. ...
... • You and your lab partner must go around to each station and identify whether the property being demonstrated is a physical or chemical property and state why. ...
MSTA WOW Chemistry
... Disposal The foam and solution left in the cylinder may be rinsed down the drain with excess water. ...
... Disposal The foam and solution left in the cylinder may be rinsed down the drain with excess water. ...
Abstract - Institute of Sound and Vibration Research
... Considering the physical conditions of the experiment, it seems likely that the growth of the bubble is due to rectified diffusion of dissolved gas in the liquid phase transferring into the gas phase of the bubble. In order to measure the rate of growth of the bubble by rectified diffusion, an exper ...
... Considering the physical conditions of the experiment, it seems likely that the growth of the bubble is due to rectified diffusion of dissolved gas in the liquid phase transferring into the gas phase of the bubble. In order to measure the rate of growth of the bubble by rectified diffusion, an exper ...
Type Of Chemical Reaction
... 42. The escape of high energy molecules from the liquid phase below the boiling point is called? ...
... 42. The escape of high energy molecules from the liquid phase below the boiling point is called? ...
Type Of Chemical Reaction
... 42. The escape of high energy molecules from the liquid phase below the boiling point is called? ...
... 42. The escape of high energy molecules from the liquid phase below the boiling point is called? ...
Name:_____________ Chemistry 114 Second Hour Exam
... van’t Hoff factor : A number that tell how many particles are formed when an ionic compound is dissolved in solution. heat of solution: The heat energy that is gained or lost when two compounds are mixed to form a solution. Henry’s Law: C=kP; C = concentration of a gas dissolved in a liquid, P the p ...
... van’t Hoff factor : A number that tell how many particles are formed when an ionic compound is dissolved in solution. heat of solution: The heat energy that is gained or lost when two compounds are mixed to form a solution. Henry’s Law: C=kP; C = concentration of a gas dissolved in a liquid, P the p ...
EXPERIMENT NO: 2
... surrounded by other molecules, and as a consequence it experiences only an attraction towards the body of the liquid. This latter attraction tends to draw the surface molecules inward and in doing so makes the liquid behave as if it were surrounded by an invisible membrane. This behavior of the surf ...
... surrounded by other molecules, and as a consequence it experiences only an attraction towards the body of the liquid. This latter attraction tends to draw the surface molecules inward and in doing so makes the liquid behave as if it were surrounded by an invisible membrane. This behavior of the surf ...
Physical concept of the surface tension of the liquid until some time
... As a result of calculations for molecules, similar in the symmetric spherical form , the coefficient n must be equal to unity. The processing of the reference data of the thermophysical properties of most substances has confirmed the theoretical formulas with an accuracy of 15%. Some substances with ...
... As a result of calculations for molecules, similar in the symmetric spherical form , the coefficient n must be equal to unity. The processing of the reference data of the thermophysical properties of most substances has confirmed the theoretical formulas with an accuracy of 15%. Some substances with ...
CHEM 2: Exam 3
... Imagine you are a scuba diver at a depth of 20 meters below the water surface where the pressure is 3 atm. If you fill your lungs to a total capacity of 6 Liters at this depth and hold your breath, then rise to the surface where the pressure is 1 atm, what would be the final volume of the air in you ...
... Imagine you are a scuba diver at a depth of 20 meters below the water surface where the pressure is 3 atm. If you fill your lungs to a total capacity of 6 Liters at this depth and hold your breath, then rise to the surface where the pressure is 1 atm, what would be the final volume of the air in you ...
9/21 properties of matter ppt
... to remove undesirable particles. One familiar example of filtration is the purification of water in a swimming pool. This is done by continuously pumping water through the pool's filter. As the water passes through the filters, dirt and any other types of debris that might be harmful are removed. An ...
... to remove undesirable particles. One familiar example of filtration is the purification of water in a swimming pool. This is done by continuously pumping water through the pool's filter. As the water passes through the filters, dirt and any other types of debris that might be harmful are removed. An ...
CHEM 2: Exam 3
... Imagine you are a scuba diver at a depth of 20 meters below the water surface where the pressure is 3 atm. If you fill your lungs to a total capacity of 6 Liters at this depth and hold your breath, then rise to the surface where the pressure is 1 atm, what would be the final volume of the air in you ...
... Imagine you are a scuba diver at a depth of 20 meters below the water surface where the pressure is 3 atm. If you fill your lungs to a total capacity of 6 Liters at this depth and hold your breath, then rise to the surface where the pressure is 1 atm, what would be the final volume of the air in you ...
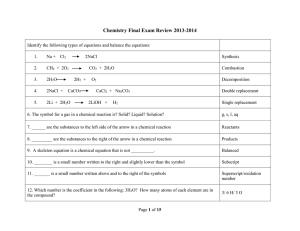
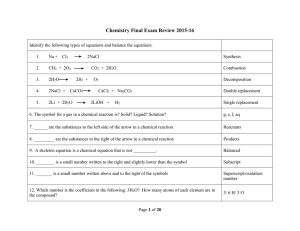
Semester II Review
... NaCl + AgNO3 AgCl + NaNO3 D.R. C3H8 + 5O2 3CO2 + 4H2O Combustion 2H2O 2H2 + O2 Decomposition 2Li + Na2CO3 2Na + Li2CO3 S.R. In every chemical reaction, what item(s) is/are conserved? Mass and Atoms ...
... NaCl + AgNO3 AgCl + NaNO3 D.R. C3H8 + 5O2 3CO2 + 4H2O Combustion 2H2O 2H2 + O2 Decomposition 2Li + Na2CO3 2Na + Li2CO3 S.R. In every chemical reaction, what item(s) is/are conserved? Mass and Atoms ...
Lecture 35 (Slides) November 7
... atmospheric pressure at sea level). If we heat water in a sealed container all of the steam that is formed is trapped above the liquid water. The additional steam formed increases the pressure of the gas above the liquid and “naturally” the boiling point rises. ...
... atmospheric pressure at sea level). If we heat water in a sealed container all of the steam that is formed is trapped above the liquid water. The additional steam formed increases the pressure of the gas above the liquid and “naturally” the boiling point rises. ...
Vapor Pressure of a Pure Liquid
... Since the change in state is isothermal and ΔG is zero (because the system is at equilibrium), ΔS may be replaced by ΔH/T: ...
... Since the change in state is isothermal and ΔG is zero (because the system is at equilibrium), ΔS may be replaced by ΔH/T: ...
Chem Unit 3 Vocabulary
... 16 dynamic condition in which 2 opposing changes occur at equal rates in a closed system 17 pressure exerted by a vapor in equilibrium with its corresponding liquid at a given temperature 18 process by which particles escape from the surface of a non-boiling liquid & enter the gas state 19 substance ...
... 16 dynamic condition in which 2 opposing changes occur at equal rates in a closed system 17 pressure exerted by a vapor in equilibrium with its corresponding liquid at a given temperature 18 process by which particles escape from the surface of a non-boiling liquid & enter the gas state 19 substance ...
Real Gases
... At higher pressures and temperatures, the gas is, again, behaving more as a super-critical fluid and so will not "condense" into a liquid. It is already extremely dense and behaving somewhat as a liquid already. The density of a supercritical fluid is more like that of a liquid than a gas, but is si ...
... At higher pressures and temperatures, the gas is, again, behaving more as a super-critical fluid and so will not "condense" into a liquid. It is already extremely dense and behaving somewhat as a liquid already. The density of a supercritical fluid is more like that of a liquid than a gas, but is si ...
Shielding Property of Closed-Cell AlSi7 Foams and AlSi7 Bulk
... energies are shown in Figure 3. As shown in the graphs, the transmission values decreased with increase in sample thickness. Transmission in the same kind of samples under all sources (energies) is similar slope. It is clear that the bulk materials have better absorbing and radiation attenuation cha ...
... energies are shown in Figure 3. As shown in the graphs, the transmission values decreased with increase in sample thickness. Transmission in the same kind of samples under all sources (energies) is similar slope. It is clear that the bulk materials have better absorbing and radiation attenuation cha ...
Answers - Shelton State
... Melting point and freezing point are the same temperature. What is vapor pressure? The pressure of vapor in equilibrium with its liquid state. The pressure of a vapor evaporating from the liquid state. What is boiling? The especially rapid evaporation that occurs when the vapor pressure of a liquid ...
... Melting point and freezing point are the same temperature. What is vapor pressure? The pressure of vapor in equilibrium with its liquid state. The pressure of a vapor evaporating from the liquid state. What is boiling? The especially rapid evaporation that occurs when the vapor pressure of a liquid ...
ln2_storage_pre
... atmospheric pressure is only about 12.5 psia because of the altitude, so the boiling point (temperature needed to exert that much vapor pressure) is only about 96 C. Similarly, what a pressure cooker does is raise the boiling temperature by raising the pressure. So for any substance, the border betw ...
... atmospheric pressure is only about 12.5 psia because of the altitude, so the boiling point (temperature needed to exert that much vapor pressure) is only about 96 C. Similarly, what a pressure cooker does is raise the boiling temperature by raising the pressure. So for any substance, the border betw ...
Chemistry 111 Study Sheet - Answers
... Melting point and freezing point are the same temperature. What is vapor pressure? The pressure of vapor in equilibrium with its liquid state. The pressure of a vapor evaporating from the liquid state. What is boiling? The especially rapid evaporation that occurs when the vapor pressure of a liquid ...
... Melting point and freezing point are the same temperature. What is vapor pressure? The pressure of vapor in equilibrium with its liquid state. The pressure of a vapor evaporating from the liquid state. What is boiling? The especially rapid evaporation that occurs when the vapor pressure of a liquid ...
practice test2(Answers)
... C) The temperature of liquid water increases linearly as it is heated D) The temperature of liquid water remains at 100°C as it boils E) Both liquid water and ice are present at 0°C. ...
... C) The temperature of liquid water increases linearly as it is heated D) The temperature of liquid water remains at 100°C as it boils E) Both liquid water and ice are present at 0°C. ...
Find your NEW seats Bellringer: Please complete Ms - Parkway C-2
... Evaporation • Liquids may change to a vapor at temperatures below their boiling points through the process of evaporation. Evaporation is a surface phenomenon in which molecules located near the liquid's edge, escape into the surroundings as vapor (a gas). On the other hand, boiling is a process in ...
... Evaporation • Liquids may change to a vapor at temperatures below their boiling points through the process of evaporation. Evaporation is a surface phenomenon in which molecules located near the liquid's edge, escape into the surroundings as vapor (a gas). On the other hand, boiling is a process in ...
Anomalies in mid-high-temperature linear thermal expansion
... The average LTECs over the temperature range of 20–500 °C are plotted in Fig. 1. The results of the pure aluminum during the third and fourth tests are the same, thus presented with one shared curve. As the tests are repeated, average LTECs of both the foams and the pure aluminum increase to their s ...
... The average LTECs over the temperature range of 20–500 °C are plotted in Fig. 1. The results of the pure aluminum during the third and fourth tests are the same, thus presented with one shared curve. As the tests are repeated, average LTECs of both the foams and the pure aluminum increase to their s ...
practice test2
... C) The temperature of liquid water increases linearly as it is heated D) The temperature of liquid water remains at 100°C as it boils E) Both liquid water and ice are present at 0°C. ...
... C) The temperature of liquid water increases linearly as it is heated D) The temperature of liquid water remains at 100°C as it boils E) Both liquid water and ice are present at 0°C. ...
Foam

A foam is a substance that is formed by trapping pockets of gas in a liquid or solid. A bath sponge and the head on a glass of beer are examples of foams. In most foams, the volume of gas is large, with thin films of liquid or solid separating the regions of gas.An important division of solid foams is into closed-cell foams and open-cell foams. In a closed-cell foam, the gas forms discrete pockets, each completely surrounded by the solid material. In an open-cell foam, the gas pockets connect with each other. A bath sponge is an example of an open-cell foam: water can easily flow through the entire structure, displacing the air. A camping mat is an example of a closed-cell foam: the gas pockets are sealed from each other so the mat cannot soak up water.Foams are examples of dispersed media. In general, gas is present in large amount so it will be divided into gas bubbles of many different sizes (the material is polydisperse) separated by liquid regions which may form films, thinner and thinner when the liquid phase is drained out of the system films. When the principal scale is small, i.e. for a very fine foam, this dispersed medium can be considered as a type of colloid.The term foam may also refer to anything that is analogous to such a foam, such as quantum foam, polyurethane foam (foam rubber), XPS foam, polystyrene, phenolic, or many other manufactured foams.