MATERIALS FOR ELECTRICAL ENGINEERING
... Material properties are determined by the structure of the outer electron shell of the elements which the material consists of. If the atoms are close enough, their electrons from outer shell interact and this constitutes the atomic bond. Character and intensity of this bond is determined by the dis ...
... Material properties are determined by the structure of the outer electron shell of the elements which the material consists of. If the atoms are close enough, their electrons from outer shell interact and this constitutes the atomic bond. Character and intensity of this bond is determined by the dis ...
Hidden Scale Invariance in Condensed Matter
... C(ρ) gives the energy-independent contribution to the pressure, which is responsible for a sizable fraction of the socalled cold pressure.4 The liquid state is complex. Like a solid, a liquid consists of strongly interacting atoms or molecules, but at the same time a liquid has the disorder and tran ...
... C(ρ) gives the energy-independent contribution to the pressure, which is responsible for a sizable fraction of the socalled cold pressure.4 The liquid state is complex. Like a solid, a liquid consists of strongly interacting atoms or molecules, but at the same time a liquid has the disorder and tran ...
Development of Tire Side Force Model Based on "Magic Formula
... We should note that Ky(T ) indicates the cornering power, as described in the previous section. In this paper, we regard the tire side force at α = 1[degree] or at α = −1[degree] to be the cornering power. The gradient of change of the cornering power with an increase/decrease in the tire surface te ...
... We should note that Ky(T ) indicates the cornering power, as described in the previous section. In this paper, we regard the tire side force at α = 1[degree] or at α = −1[degree] to be the cornering power. The gradient of change of the cornering power with an increase/decrease in the tire surface te ...
Igneous Petrology 2001
... Thermodynamics: absolute basics – Gibbs free energy, heat capacity, entropy, enthalpy, work etc. (Thermodynamics is desirable but not essential) Mathematics: basic algebra and elementary calculus (Basic Math, Introduction to Calculus). MP-SKM, slide 3 ...
... Thermodynamics: absolute basics – Gibbs free energy, heat capacity, entropy, enthalpy, work etc. (Thermodynamics is desirable but not essential) Mathematics: basic algebra and elementary calculus (Basic Math, Introduction to Calculus). MP-SKM, slide 3 ...
European Journal of Biochemistry
... cells contain increased amounts of phospholipids which are at least partly present in the outer leaflet [2]. The characteristics of the Arrhenius plot differ considerably from the ones observed with cells of strain JC7620. Some phage adsorption occurs below the inflection point temperature at 18 "C ...
... cells contain increased amounts of phospholipids which are at least partly present in the outer leaflet [2]. The characteristics of the Arrhenius plot differ considerably from the ones observed with cells of strain JC7620. Some phage adsorption occurs below the inflection point temperature at 18 "C ...
Parent Anions of Iron, Manganese, and Nickel Tetraphenyl
... used 20 single Gaussians, with decay parameters ranging between 0.0452 and 3.87 × 106, to construct a set of 11 s-type contracted Gaussians, 4 of which have r2 prefactors, 5 p-type contracted Gaussians, and 4 d-type contracted Gaussians. Similar size basis sets were used for the Ni and Mn calculatio ...
... used 20 single Gaussians, with decay parameters ranging between 0.0452 and 3.87 × 106, to construct a set of 11 s-type contracted Gaussians, 4 of which have r2 prefactors, 5 p-type contracted Gaussians, and 4 d-type contracted Gaussians. Similar size basis sets were used for the Ni and Mn calculatio ...
The study of the pre-exponential factor and activation energy in
... Sherman [6] stated that between two conducting particles in a disordered system whose separation d is large (≥10nm), the resistance is controlled by the bulk resistivity of the matrix itself. However, when d is smaller (≤10nm), electrons may tunnel between conductive elements, which leads to a lower ...
... Sherman [6] stated that between two conducting particles in a disordered system whose separation d is large (≥10nm), the resistance is controlled by the bulk resistivity of the matrix itself. However, when d is smaller (≤10nm), electrons may tunnel between conductive elements, which leads to a lower ...
Stress relaxation behaviour in compression and some other
... Thermoplastic elastomer shows two Ltransition temperatures: a lower one corresponding to the freezing-in or appearance of molecular motion and upper one corresponding to the break down or restoration of the secondary valence cross-linking. Softening takes place within two temperature regions which m ...
... Thermoplastic elastomer shows two Ltransition temperatures: a lower one corresponding to the freezing-in or appearance of molecular motion and upper one corresponding to the break down or restoration of the secondary valence cross-linking. Softening takes place within two temperature regions which m ...
Adhesion strength and contact resistance of flip chip on flex packages
... are capable of moving relatively easily and exhibit less adhesion strength. Again, as the bonding temperature increases the degree of cross-linking as well as adhesion also increases. Because, the polymer chains locked together and their movement become consequently somewhat restricted. Cross-linked ...
... are capable of moving relatively easily and exhibit less adhesion strength. Again, as the bonding temperature increases the degree of cross-linking as well as adhesion also increases. Because, the polymer chains locked together and their movement become consequently somewhat restricted. Cross-linked ...
Absorption Spectra and Photolysis of Methyl Peroxide in Liquid and
... Synthesis and Identification of Methyl Peroxide. Methyl peroxide is not available commercially and, therefore, was synthesized by methylation of hydrogen peroxide as described by Vaghjiani and Ravishankara13 with minor modifications. A 12 M solution of KOH (Fisher Scientific >85%) was slowly added t ...
... Synthesis and Identification of Methyl Peroxide. Methyl peroxide is not available commercially and, therefore, was synthesized by methylation of hydrogen peroxide as described by Vaghjiani and Ravishankara13 with minor modifications. A 12 M solution of KOH (Fisher Scientific >85%) was slowly added t ...
synthesis and characterization of complex
... The author, whose copyright is declared on the title page of this work, has granted to Simon Fraser University the right to lend this thesis, project or extended essay to users of the Simon Fraser University Library, and to make partial or single copies only for such users or in response to a reques ...
... The author, whose copyright is declared on the title page of this work, has granted to Simon Fraser University the right to lend this thesis, project or extended essay to users of the Simon Fraser University Library, and to make partial or single copies only for such users or in response to a reques ...
Chapter 16
... In the last section we developed three equivalent expressions for the equilibrium constant KP of reacting ideal-gas mixtures: Eq. 16–13, which expresses KP in terms of partial pressures; Eq. 16–14, which expresses KP in terms of the standard-state Gibbs function change ΔG*(T); and Eq. 16–15, which e ...
... In the last section we developed three equivalent expressions for the equilibrium constant KP of reacting ideal-gas mixtures: Eq. 16–13, which expresses KP in terms of partial pressures; Eq. 16–14, which expresses KP in terms of the standard-state Gibbs function change ΔG*(T); and Eq. 16–15, which e ...
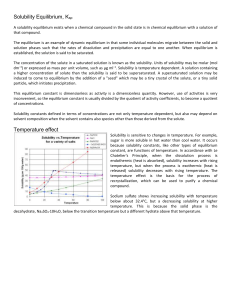
ksp - lozon.ca
... The solubility of a sparingly soluble salt is reduced in a solution that contains an ion in common with that salt. For instance, the solubility of silver chloride in water is reduced if a solution of sodium chloride is added to a suspension of silver chloride in water. A practical example used very ...
... The solubility of a sparingly soluble salt is reduced in a solution that contains an ion in common with that salt. For instance, the solubility of silver chloride in water is reduced if a solution of sodium chloride is added to a suspension of silver chloride in water. A practical example used very ...
In Situ Soft X‑ray Absorption Spectroscopy Applied to Solid
... mixture of substrates, reactants, products, solvents, and solid catalysts. Element-specific soft X-ray absorption spectroscopy (XAS) is a promising method to detect target substrate and product separately from the other components using chemically different inner shell excitation energies. In the pres ...
... mixture of substrates, reactants, products, solvents, and solid catalysts. Element-specific soft X-ray absorption spectroscopy (XAS) is a promising method to detect target substrate and product separately from the other components using chemically different inner shell excitation energies. In the pres ...
Glass transition
The glass–liquid transition or glass transition for short is the reversible transition in amorphous materials (or in amorphous regions within semicrystalline materials) from a hard and relatively brittle state into a molten or rubber-like state. An amorphous solid that exhibits a glass transition is called a glass. Supercooling a viscous liquid into the glass state is called vitrification, from the Latin vitreum, ""glass"" via French vitrifier.Despite the massive change in the physical properties of a material through its glass transition, the transition is not itself a phase transition of any kind; rather it is a laboratory phenomenon extending over a range of temperature and defined by one of several conventions. Such conventions include a constant cooling rate (20 K/min) and a viscosity threshold of 1012 Pa·s, among others. Upon cooling or heating through this glass-transition range, the material also exhibits a smooth step in the thermal-expansion coefficient and in the specific heat, with the location of these effects again being dependent on the history of the material. However, the question of whether some phase transition underlies the glass transition is a matter of continuing research.The glass-transition temperature Tg is always lower than the melting temperature, Tm, of the crystalline state of the material, if one exists.