Intra-European Fellowships (IEF)
... inhomogeneous, forming clumps of very different shape and sizes. Indeed, the CDW order extends from many small puddles like the small ice crystals, to very large one (like an iceberg). We discovered that the distribution of the puddles size and puddle density follows a power-law distribution over mo ...
... inhomogeneous, forming clumps of very different shape and sizes. Indeed, the CDW order extends from many small puddles like the small ice crystals, to very large one (like an iceberg). We discovered that the distribution of the puddles size and puddle density follows a power-law distribution over mo ...
Unit 4: Physical Properties and Changes
... Brittle – likely to break, snap, or crack when subjected to pressure Conductivity – a property that describes if heat or electrical charges pass through the material easily Density – the amount of matter in a given space or volume, a relationship between mass and volume Displacement – a way to measu ...
... Brittle – likely to break, snap, or crack when subjected to pressure Conductivity – a property that describes if heat or electrical charges pass through the material easily Density – the amount of matter in a given space or volume, a relationship between mass and volume Displacement – a way to measu ...
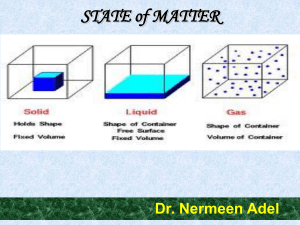
Changes of State
... WHY IS SALT SO STABLE? When salt is made, it forms a lattice, or a strong cube structure. Positive Sodium is attracted to negative Chlorine ...
... WHY IS SALT SO STABLE? When salt is made, it forms a lattice, or a strong cube structure. Positive Sodium is attracted to negative Chlorine ...
Key - Sardis Secondary
... 15. What is the relationship between the kinetic energy of molecules and their physical state? ...
... 15. What is the relationship between the kinetic energy of molecules and their physical state? ...
Slide 1
... radical flux at low gas temperatures for material processing applications. In this work, a short-pulse (200 ns FWHM applied voltage) excited argon DBD was operated between 5-500 Torr, 1-4 kV, and 5 kHz repetition rate. Diode-Laser Absorption Spectroscopy was used to obtain Ar 1s3*2p2 transition prof ...
... radical flux at low gas temperatures for material processing applications. In this work, a short-pulse (200 ns FWHM applied voltage) excited argon DBD was operated between 5-500 Torr, 1-4 kV, and 5 kHz repetition rate. Diode-Laser Absorption Spectroscopy was used to obtain Ar 1s3*2p2 transition prof ...
Chemistry II Demonstration Assessment
... elements from a compound. To use electrical energy to break the chemical bonds of a compound. Background Information: Chemical reactions occur when atoms are separated, rearranged, and/or joined in a new way. In this experiment you will decompose the chemical compound you synthesized in the laborato ...
... elements from a compound. To use electrical energy to break the chemical bonds of a compound. Background Information: Chemical reactions occur when atoms are separated, rearranged, and/or joined in a new way. In this experiment you will decompose the chemical compound you synthesized in the laborato ...
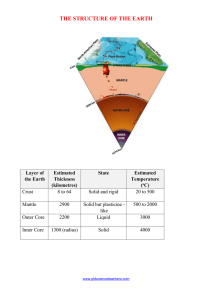
Structure of the Earth Worksheet
... (b) Predict the temperature at the following depths – 8 km and 20 km. (c) The deepest mine is less than 10 kilometres deep. How do you think geologists (scientists who study earth movements and rocks) estimate the thickness and temperature of the layers of the earth? ...
... (b) Predict the temperature at the following depths – 8 km and 20 km. (c) The deepest mine is less than 10 kilometres deep. How do you think geologists (scientists who study earth movements and rocks) estimate the thickness and temperature of the layers of the earth? ...
CHM 111: General Physical Chemistry 3 Units
... masses. Fundamental particles of the atom and atomic structure. Modern electronic theory of atoms; electronic configuration of the elements. Periodicity of the elements. Radioactivity: Stoichiometry: mole concept, chemical formulas, equations and calculations. States of matter: gas empirical gas law ...
... masses. Fundamental particles of the atom and atomic structure. Modern electronic theory of atoms; electronic configuration of the elements. Periodicity of the elements. Radioactivity: Stoichiometry: mole concept, chemical formulas, equations and calculations. States of matter: gas empirical gas law ...
Answer
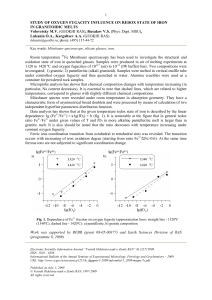
... Visible light contains radiation in the wavelength range 4000Å (violet)7000Å (deep red). A material that appears red in transmission light absorbs all the wavelengths of the visible light in the range 4000-6500Å and transmits wavelengths >6500Å. This material must belong to semiconducting class of ...
... Visible light contains radiation in the wavelength range 4000Å (violet)7000Å (deep red). A material that appears red in transmission light absorbs all the wavelengths of the visible light in the range 4000-6500Å and transmits wavelengths >6500Å. This material must belong to semiconducting class of ...
NOTES – CHAPTER 4 PYSICAL PROPERTIES – GLASS AND SOIL
... b. Glass is a hard, brittle, amorphous substance that is composed of silicon oxides mixed with various metal oxides. c. Tempered glass = glass to which strength is added by introducing stress through the rapid heating and cooling of the glass surface d. Laminated glass = two sheets of ordinary glass ...
... b. Glass is a hard, brittle, amorphous substance that is composed of silicon oxides mixed with various metal oxides. c. Tempered glass = glass to which strength is added by introducing stress through the rapid heating and cooling of the glass surface d. Laminated glass = two sheets of ordinary glass ...
kauzmann temperature and the glass transition
... kinetic glass transition prevents equilibration of supercooled liquid near TK. Because kinetic glass transition is higher than Kauzmann temperature, it intervenes before the entropy crisis occurs. From it follows that the estimation of TK can be obtained by extrapolation of supercooled liquid entrop ...
... kinetic glass transition prevents equilibration of supercooled liquid near TK. Because kinetic glass transition is higher than Kauzmann temperature, it intervenes before the entropy crisis occurs. From it follows that the estimation of TK can be obtained by extrapolation of supercooled liquid entrop ...
PHYSICAL PROPERTIES * GLASS AND SOIL
... Glass is a hard, brittle, amorphous substance that is composed of silicon oxides mixed with various metal oxides ...
... Glass is a hard, brittle, amorphous substance that is composed of silicon oxides mixed with various metal oxides ...
Relaxations of PVC and Calculation of the Activation Energy of the
... The Arrhenius plot with a line of best fit and the equation of that line is highlighted in Figure 4. The slope is equal to the negative of the activation energy divided by the gas constant. The activation energy for the β relaxation of PVC is 77 kJmol-1. This is effectively the energy barrier the ma ...
... The Arrhenius plot with a line of best fit and the equation of that line is highlighted in Figure 4. The slope is equal to the negative of the activation energy divided by the gas constant. The activation energy for the β relaxation of PVC is 77 kJmol-1. This is effectively the energy barrier the ma ...
The Viscoelastic phenomena Viscoelasticity is a general property of
... motion is possible before the onset of the liquid-like Brownian motion at the glass transition. The viscoelastic properties are highly temperature-dependent so that the maximum temperature must be clearly specified, and taken into consideration. Viscoelastic behavior is a relationship between stress ...
... motion is possible before the onset of the liquid-like Brownian motion at the glass transition. The viscoelastic properties are highly temperature-dependent so that the maximum temperature must be clearly specified, and taken into consideration. Viscoelastic behavior is a relationship between stress ...
Glass transition
The glass–liquid transition or glass transition for short is the reversible transition in amorphous materials (or in amorphous regions within semicrystalline materials) from a hard and relatively brittle state into a molten or rubber-like state. An amorphous solid that exhibits a glass transition is called a glass. Supercooling a viscous liquid into the glass state is called vitrification, from the Latin vitreum, ""glass"" via French vitrifier.Despite the massive change in the physical properties of a material through its glass transition, the transition is not itself a phase transition of any kind; rather it is a laboratory phenomenon extending over a range of temperature and defined by one of several conventions. Such conventions include a constant cooling rate (20 K/min) and a viscosity threshold of 1012 Pa·s, among others. Upon cooling or heating through this glass-transition range, the material also exhibits a smooth step in the thermal-expansion coefficient and in the specific heat, with the location of these effects again being dependent on the history of the material. However, the question of whether some phase transition underlies the glass transition is a matter of continuing research.The glass-transition temperature Tg is always lower than the melting temperature, Tm, of the crystalline state of the material, if one exists.