AQA GCSE Chemistry My Revision Notes
... (a) Because the products are at a lower energy level than the reactants. This shows that heat is being released to the surroundings. (b) CH4(g) + 2O2(g) CO2(g) + 2H2O(g) 1 mark is for correct formulae, 1 for balancing and 1 for correct state symbols. As with all formulae and equations, jus ...
... (a) Because the products are at a lower energy level than the reactants. This shows that heat is being released to the surroundings. (b) CH4(g) + 2O2(g) CO2(g) + 2H2O(g) 1 mark is for correct formulae, 1 for balancing and 1 for correct state symbols. As with all formulae and equations, jus ...
2002 AP Chemistry Free-Response Questions
... 4. Write the formulas to show the reactants and the products for any FIVE of the laboratory situations described below. Answers to more than five choices will not be graded. In all cases, a reaction occurs. Assume that solutions are aqueous unless otherwise indicated. Represent substances in solutio ...
... 4. Write the formulas to show the reactants and the products for any FIVE of the laboratory situations described below. Answers to more than five choices will not be graded. In all cases, a reaction occurs. Assume that solutions are aqueous unless otherwise indicated. Represent substances in solutio ...
FINAL EXAM REVIEW PROBLEMS
... 45. Consider two sample of nitrogen gas. Sample 1 contains 1.5 mol of N2 and has a volume of 36.7 L at 25C and 1 atm. Sample 2 has a volume of 16.5 L at 25C and 1 atm. Calculate the number of moles of N2 in sample 2. 46. A weather balloon contains 1.10 x 103 mol of helium and has a volume of 2.70 ...
... 45. Consider two sample of nitrogen gas. Sample 1 contains 1.5 mol of N2 and has a volume of 36.7 L at 25C and 1 atm. Sample 2 has a volume of 16.5 L at 25C and 1 atm. Calculate the number of moles of N2 in sample 2. 46. A weather balloon contains 1.10 x 103 mol of helium and has a volume of 2.70 ...
C6 Revision Guide - West Derby School
... When chlorofluorocarbons (CFCs) were first discovered, it was thought that they were safe to use. However, since then scientists have discovered that CFC molecules slowly move upwards into the stratosphere where they attack the ozone. Society agreed with the view of the scientists’ that CFCs had bee ...
... When chlorofluorocarbons (CFCs) were first discovered, it was thought that they were safe to use. However, since then scientists have discovered that CFC molecules slowly move upwards into the stratosphere where they attack the ozone. Society agreed with the view of the scientists’ that CFCs had bee ...
Unit 8 Homework Packet
... 27. One process for the commercial production of baking soda (sodium hydrogen carbonate) involves the following reaction, in which the carbon dioxide is used in its solid form ("dry ice") both to serve as a source of reactant and to cool the reaction system to a temperature low enough for the sodium ...
... 27. One process for the commercial production of baking soda (sodium hydrogen carbonate) involves the following reaction, in which the carbon dioxide is used in its solid form ("dry ice") both to serve as a source of reactant and to cool the reaction system to a temperature low enough for the sodium ...
Reporting Category 3: Bonding and Chemical Reactions
... Add the total number of valence electrons for every atom in the molecule. If it is an ion, add or subtract electrons to produce the correct charge. 2. Write the structure of the skeleton. The more electronegative atom usually belongs in the center. Connect atoms with lines (or pairs of dots). 3. Dis ...
... Add the total number of valence electrons for every atom in the molecule. If it is an ion, add or subtract electrons to produce the correct charge. 2. Write the structure of the skeleton. The more electronegative atom usually belongs in the center. Connect atoms with lines (or pairs of dots). 3. Dis ...
3 chemical foundations: elements, atoms and ions
... Matter almost always is found as mixtures of different chemical compounds. Elements are rarely found in their pure forms. However, some elements are relatively unreactive and, on occasion, may be found in pure form. Examples include: the noble metals such as gold (Au), silver (Ag) and platinum (Pt) ...
... Matter almost always is found as mixtures of different chemical compounds. Elements are rarely found in their pure forms. However, some elements are relatively unreactive and, on occasion, may be found in pure form. Examples include: the noble metals such as gold (Au), silver (Ag) and platinum (Pt) ...
School of Chemistry and Physics Westville Campus, Durban
... What is the volume of 30.0 g of methanol (CH3OH(l), density = 0.791 g mL-1)? A ...
... What is the volume of 30.0 g of methanol (CH3OH(l), density = 0.791 g mL-1)? A ...
- Catalyst
... 4. The ______molar mass______________________ of an element has the units g/mole. 5. Smallest unit of an element is a(n) ____atom_____________________________. 6. The ___chemical symbol_________ of an element is one or two letters found on the periodic table. 7. Isotopes have the same number ...
... 4. The ______molar mass______________________ of an element has the units g/mole. 5. Smallest unit of an element is a(n) ____atom_____________________________. 6. The ___chemical symbol_________ of an element is one or two letters found on the periodic table. 7. Isotopes have the same number ...
Lecture syllabus - Linfield College
... organic compounds are studied. During second semester (CHEM 322), aromatics, organometallics, alcohols, ethers, aldehydes, ketones, carboxylic acids, acid derivatives, and amines are the classes stressed. The course is designed to provide a fundamental knowledge of organic chemistry - the study of c ...
... organic compounds are studied. During second semester (CHEM 322), aromatics, organometallics, alcohols, ethers, aldehydes, ketones, carboxylic acids, acid derivatives, and amines are the classes stressed. The course is designed to provide a fundamental knowledge of organic chemistry - the study of c ...
Syllabus of Medical / Dental Colleges Entrance Test 2016
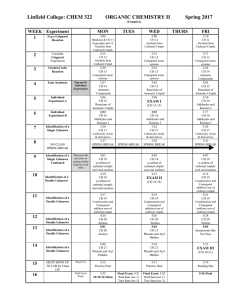
... Describe the chemistry of Alkanes with emphasis on a) Combustion b) Free radical substitution including mechanism Discuss the chemistry of Alkenes with emphasis on a) Preparation of alkenes by elimination reactions i) Dehydration of alcohols ii) Dehydrohalogenation of Alkyl halide b) Reaction of Alk ...
... Describe the chemistry of Alkanes with emphasis on a) Combustion b) Free radical substitution including mechanism Discuss the chemistry of Alkenes with emphasis on a) Preparation of alkenes by elimination reactions i) Dehydration of alcohols ii) Dehydrohalogenation of Alkyl halide b) Reaction of Alk ...
Ch. 3 Sections 3.9-3.10 Notes
... Suppose a chemist mixed 1.00 mol of N2 with 5.00 mol of H2. What is the maximum number of moles of product that could form? Note the coefficients tell us that 1 mol of N2 consumes 3 mol of H2. 1 mol N2 ↔ 3 mol H2 But 5 mol of H2 was used, not 3, so there will be 2 mol of H2 left over. Once the 1 mol ...
... Suppose a chemist mixed 1.00 mol of N2 with 5.00 mol of H2. What is the maximum number of moles of product that could form? Note the coefficients tell us that 1 mol of N2 consumes 3 mol of H2. 1 mol N2 ↔ 3 mol H2 But 5 mol of H2 was used, not 3, so there will be 2 mol of H2 left over. Once the 1 mol ...
Effect Of Convection For Gaseous Hydrochloride
... feedstock consists of about 40% of virgin pig iron consisting mostly of Fe, C, Si, P Mn and S. and 60% of scrap, containing number of different metals. Some of them, Ni, Cr, V make steel better etc., however metals like Zn, Cd, Pb are unwelcome. In modern steelmaking equipment using oxygen atmospher ...
... feedstock consists of about 40% of virgin pig iron consisting mostly of Fe, C, Si, P Mn and S. and 60% of scrap, containing number of different metals. Some of them, Ni, Cr, V make steel better etc., however metals like Zn, Cd, Pb are unwelcome. In modern steelmaking equipment using oxygen atmospher ...
The Chemistry of Aqueous Systems
... The Chemistry of Aqueous Systems: Gases, Liquids, Acids, Bases and Electrochemistry Objectives for The Chemistry of Aqueous Systems Properly prepared students, in random order, will 1) Be able to explain, apply and demonstrate the use (verbally, written and arithmetically) of the following gas laws ...
... The Chemistry of Aqueous Systems: Gases, Liquids, Acids, Bases and Electrochemistry Objectives for The Chemistry of Aqueous Systems Properly prepared students, in random order, will 1) Be able to explain, apply and demonstrate the use (verbally, written and arithmetically) of the following gas laws ...
RxnTypesPrednotesIIAP
... and [AgCl2] are two important complexes that form. The diammine silver complex results when a solution containing the silver ion is reacted with an aqueous ammonium hydroxide solution. The dichloroargentate complex only forms when there is a strong excess of the chloride ion. For example, if 6 M hyd ...
... and [AgCl2] are two important complexes that form. The diammine silver complex results when a solution containing the silver ion is reacted with an aqueous ammonium hydroxide solution. The dichloroargentate complex only forms when there is a strong excess of the chloride ion. For example, if 6 M hyd ...
Net ionic equation
... Why Ionic Compounds Dissolve in Water The forces holding an ionic compound together are the strong electrical attraction that exists between cations and anions. It is therefore somewhat surprising that ionic compounds will dissolve in water. The reason some ionic compounds will dissolve in water is ...
... Why Ionic Compounds Dissolve in Water The forces holding an ionic compound together are the strong electrical attraction that exists between cations and anions. It is therefore somewhat surprising that ionic compounds will dissolve in water. The reason some ionic compounds will dissolve in water is ...
ppt - UCLA Chemistry and Biochemistry
... 1) Solve exactly using quadratic equation 2) Solve by approximation 3) Solve by successive approximation if 2 doesn’t work ...
... 1) Solve exactly using quadratic equation 2) Solve by approximation 3) Solve by successive approximation if 2 doesn’t work ...
Chemistry Revision Checklist F4 2017 (inc F3)
... Describe the formation of ionic bonds between metallic and non-metallic elements Describe the formation of ionic bonds between elements from Groups I and VII Use the symbols of the elements and write the formulae of simple compounds Deduce the formula of a simple compound from the relative numbers o ...
... Describe the formation of ionic bonds between metallic and non-metallic elements Describe the formation of ionic bonds between elements from Groups I and VII Use the symbols of the elements and write the formulae of simple compounds Deduce the formula of a simple compound from the relative numbers o ...
AP Chemistry
... Part II. Atoms, Molecules and Ions 2.1 Fundamental Chemical Laws The Law of Conservation of Mass: mass is neither lost nor gained during an ordinary chemical reaction. In other words, the products of a reaction must have the same number of type of atoms as the reactants. Law of Definite Proportion: ...
... Part II. Atoms, Molecules and Ions 2.1 Fundamental Chemical Laws The Law of Conservation of Mass: mass is neither lost nor gained during an ordinary chemical reaction. In other words, the products of a reaction must have the same number of type of atoms as the reactants. Law of Definite Proportion: ...
From (2)
... Why is it important to investigate the rates of this reaction for metallurgical engineering? Disposal problems of radioactive wastes generated from treatment of uranium and thorium ores. Traces of uranium and thorium in other ores. Example: the slag resulted from the production of ferro-noibium is r ...
... Why is it important to investigate the rates of this reaction for metallurgical engineering? Disposal problems of radioactive wastes generated from treatment of uranium and thorium ores. Traces of uranium and thorium in other ores. Example: the slag resulted from the production of ferro-noibium is r ...
inorganic chemistry
... organisms in sufficient quantities. This high reactivity is due to the atoms being highly electronegative due to their high effective nuclear charge. They can gain an electron by reacting with atoms of other elements. Fluorine is one of the most reactive elements in existence, attacking otherwise in ...
... organisms in sufficient quantities. This high reactivity is due to the atoms being highly electronegative due to their high effective nuclear charge. They can gain an electron by reacting with atoms of other elements. Fluorine is one of the most reactive elements in existence, attacking otherwise in ...
7 Periodic Properties of the Elements
... A billiard ball is an imperfect model for an atom. The ball has a definite “hard” boundary, while an atom has no definite edge and can be reshaped by interactions with other atoms. That said, the billiard ball is a more appropriate analogy for the nonbonding radius of a fluorine atom. The ball’s rad ...
... A billiard ball is an imperfect model for an atom. The ball has a definite “hard” boundary, while an atom has no definite edge and can be reshaped by interactions with other atoms. That said, the billiard ball is a more appropriate analogy for the nonbonding radius of a fluorine atom. The ball’s rad ...
Redox

Redox reactions include all chemical reactions in which atoms have their oxidation state changed; in general, redox reactions involve the transfer of electrons between species. The term ""redox"" comes from two concepts involved with electron transfer: reduction and oxidation. It can be explained in simple terms: Oxidation is the loss of electrons or an increase in oxidation state by a molecule, atom, or ion. Reduction is the gain of electrons or a decrease in oxidation state by a molecule, atom, or ion.Although oxidation reactions are commonly associated with the formation of oxides from oxygen molecules, these are only specific examples of a more general concept of reactions involving electron transfer.Redox reactions, or oxidation-reduction reactions, have a number of similarities to acid–base reactions. Like acid–base reactions, redox reactions are a matched set, that is, there cannot be an oxidation reaction without a reduction reaction happening simultaneously. The oxidation alone and the reduction alone are each called a half-reaction, because two half-reactions always occur together to form a whole reaction. When writing half-reactions, the gained or lost electrons are typically included explicitly in order that the half-reaction be balanced with respect to electric charge.Though sufficient for many purposes, these descriptions are not precisely correct. Oxidation and reduction properly refer to a change in oxidation state — the actual transfer of electrons may never occur. The oxidation state of an atom is the fictitious charge that an atom would have if all bonds between atoms of different elements were 100% ionic. Thus, oxidation is better defined as an increase in oxidation state, and reduction as a decrease in oxidation state. In practice, the transfer of electrons will always cause a change in oxidation state, but there are many reactions that are classed as ""redox"" even though no electron transfer occurs (such as those involving covalent bonds).There are simple redox processes, such as the oxidation of carbon to yield carbon dioxide (CO2) or the reduction of carbon by hydrogen to yield methane (CH4), and more complex processes such as the oxidation of glucose (C6H12O6) in the human body through a series of complex electron transfer processes.