Instructor`s Guide - Ventura Educational Systems
... Lesson 4: Making Molecules 1… Getting Started Teacher Sheet Background Atoms are nature’s building blocks, but we very seldom see atoms by themselves in nature. More often than not, atoms bond with other atoms to form molecules and compounds. Covalent Molecules, like ...
... Lesson 4: Making Molecules 1… Getting Started Teacher Sheet Background Atoms are nature’s building blocks, but we very seldom see atoms by themselves in nature. More often than not, atoms bond with other atoms to form molecules and compounds. Covalent Molecules, like ...
English Medium - sakshieducation.com
... 1 Mark 1. State important uses of decomposition reaction? A. Extract metals from their compounds & digestion of food. 2. What happens when silver chloride exposed to sunlight? A. 2 AgCl →2Ag+Cl2↑ 3. Why photosynthesis reaction considered as endothermic reaction? A. During photosynthesis plants absor ...
... 1 Mark 1. State important uses of decomposition reaction? A. Extract metals from their compounds & digestion of food. 2. What happens when silver chloride exposed to sunlight? A. 2 AgCl →2Ag+Cl2↑ 3. Why photosynthesis reaction considered as endothermic reaction? A. During photosynthesis plants absor ...
Module-2-s-and-d-elements - Львівський національний медичний
... Chemical elements are substances that cannot be decomposed or broken into more elementary substances by ordinary chemical means. Elements were at one time believed to be the fundamental substances but they are known to consist of a number of different elementary particles now. More than 100 chemical ...
... Chemical elements are substances that cannot be decomposed or broken into more elementary substances by ordinary chemical means. Elements were at one time believed to be the fundamental substances but they are known to consist of a number of different elementary particles now. More than 100 chemical ...
Chapter 3 Powerpoint
... • Stoichiometry is the area of study that examines the quantities of substances consumed and produced in chemical reactions. • Remember: Atoms are neither created nor destroyed during any chemical reaction or physical process. ...
... • Stoichiometry is the area of study that examines the quantities of substances consumed and produced in chemical reactions. • Remember: Atoms are neither created nor destroyed during any chemical reaction or physical process. ...
Subject Area Assessment Guides
... or inter-atomic forces. When enough energy is added to the solid, the kinetic energy of the atoms and molecules increases sufficiently to overcome the attractive forces between the particles, and they break free of their fixed lattice positions. This ...
... or inter-atomic forces. When enough energy is added to the solid, the kinetic energy of the atoms and molecules increases sufficiently to overcome the attractive forces between the particles, and they break free of their fixed lattice positions. This ...
CHAPTER 4: CHEMICAL QUANTITIES and AQUEOUS REACTIONS
... Solute (solid, liquid or gas) + Water (solvent) → Aqueous solution. If the aqueous solution conducts electric current, the solute is called as electrolytes. Electrolytes are classified into 3 types. ...
... Solute (solid, liquid or gas) + Water (solvent) → Aqueous solution. If the aqueous solution conducts electric current, the solute is called as electrolytes. Electrolytes are classified into 3 types. ...
Chapter 3 - Whitwell High School
... amounts of substances • Within chemical compounds, moles of atoms always combine in the same ratio as the individual atoms themselves. ...
... amounts of substances • Within chemical compounds, moles of atoms always combine in the same ratio as the individual atoms themselves. ...
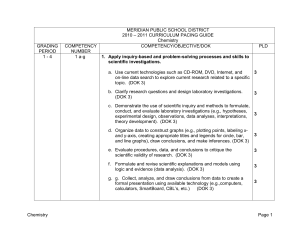
MERIDIAN PUBLIC SCHOOL DISTRICT
... Atomic number, atomic mass, mass number, and number of protons, electrons, and neutrons in isotopes of elements Chemical characteristics of each region Periodic properties (e.g., metal/nonmetal/metalloid behavior, electrical/heat conductivity, electronegativity, electron affinity, ionization energy, ...
... Atomic number, atomic mass, mass number, and number of protons, electrons, and neutrons in isotopes of elements Chemical characteristics of each region Periodic properties (e.g., metal/nonmetal/metalloid behavior, electrical/heat conductivity, electronegativity, electron affinity, ionization energy, ...
www.xtremepapers.net
... The alternating oppositely charged ions in 3 dimensions in ionic solids allows a strong attraction between them. The continuous, 3dimensional, tetrahedral, strongly-bonded covalent structures of diamond and silicon(IV) oxide also result in strong attractions and hence high m.pts. The layer nature of ...
... The alternating oppositely charged ions in 3 dimensions in ionic solids allows a strong attraction between them. The continuous, 3dimensional, tetrahedral, strongly-bonded covalent structures of diamond and silicon(IV) oxide also result in strong attractions and hence high m.pts. The layer nature of ...
Thermochemistry
... In the first equation, we formed 2 moles of H2O and have a H value of 571.7 kJ. In the second equation, we formed half as much water using half as much reactant material and t he H value was half the size. What if we wanted to know the H value for the formation of 10 moles of H2O. This means that ...
... In the first equation, we formed 2 moles of H2O and have a H value of 571.7 kJ. In the second equation, we formed half as much water using half as much reactant material and t he H value was half the size. What if we wanted to know the H value for the formation of 10 moles of H2O. This means that ...
In situ Raman Spectroscopic Study of Supported Molten Salt
... The catalytic oxidation of SO2 to SO3 plays a key role in a number of industrial processes, which due to the associated sulfur oxide emissions have significant environmental impact. Although the main source of SO2 emissions to the atmosphere is the coal-fired power generation, large amounts of SO2 a ...
... The catalytic oxidation of SO2 to SO3 plays a key role in a number of industrial processes, which due to the associated sulfur oxide emissions have significant environmental impact. Although the main source of SO2 emissions to the atmosphere is the coal-fired power generation, large amounts of SO2 a ...
1 Unit 11-12: Equilibrium and Acid/Bases Notes Colligative
... ‐ a direct relationship between kinetic energy and temperature exists ‐ the higher the temp, the faster the molecules will go ‐ more chaotic motion will lead to more collisions Increase surface area of reactants ‐ the more sites exposed to react, the more collisions can occur ‐ a large piece ...
... ‐ a direct relationship between kinetic energy and temperature exists ‐ the higher the temp, the faster the molecules will go ‐ more chaotic motion will lead to more collisions Increase surface area of reactants ‐ the more sites exposed to react, the more collisions can occur ‐ a large piece ...
Additional Review
... “ite” has fewer atoms of an atom “ate” has more atoms of an element Sulfite: ___________ Sulfate: ___________ o Compounds containing polyatomic ions follow the same balancing rules as all other ionic compounds. o Naming polyatomic compounds: {metal} + {polyatomic ion} the name of the pol ...
... “ite” has fewer atoms of an atom “ate” has more atoms of an element Sulfite: ___________ Sulfate: ___________ o Compounds containing polyatomic ions follow the same balancing rules as all other ionic compounds. o Naming polyatomic compounds: {metal} + {polyatomic ion} the name of the pol ...
Document
... piece of tin metal, Sn(s), was added. To the second beaker a piece of magnesium metal, Mg(s), was added. In one beaker, a reaction took place, while in the other beaker no reaction was ...
... piece of tin metal, Sn(s), was added. To the second beaker a piece of magnesium metal, Mg(s), was added. In one beaker, a reaction took place, while in the other beaker no reaction was ...
Complete the following equations
... (Nitrogen cannot form NCl5 because nitrogen atom is a second period element, which an outermost shell (n = 2) cannot accommodate more than 8 electrons. P, As, and Sb have the outermost-shell with n > 2; each can form 5 covalent bonds and form compounds, such as MX5.) ...
... (Nitrogen cannot form NCl5 because nitrogen atom is a second period element, which an outermost shell (n = 2) cannot accommodate more than 8 electrons. P, As, and Sb have the outermost-shell with n > 2; each can form 5 covalent bonds and form compounds, such as MX5.) ...
PREPARATORY PROBLEMS (Theoretical)
... The natural tendency of any chemical reaction to proceed in a certain direction at constant temperature and pressure is determined by the sign of the Gibbs energy of the reaction, DG. This is the universal principle. If DG < 0, the reaction can proceed predominantly in the forward direction (a produ ...
... The natural tendency of any chemical reaction to proceed in a certain direction at constant temperature and pressure is determined by the sign of the Gibbs energy of the reaction, DG. This is the universal principle. If DG < 0, the reaction can proceed predominantly in the forward direction (a produ ...
PREPARATORY PROBLEMS
... The natural tendency of any chemical reaction to proceed in a certain direction at constant temperature and pressure is determined by the sign of the Gibbs energy of the reaction, DG. This is the universal principle. If DG < 0, the reaction can proceed predominantly in the forward direction (a produ ...
... The natural tendency of any chemical reaction to proceed in a certain direction at constant temperature and pressure is determined by the sign of the Gibbs energy of the reaction, DG. This is the universal principle. If DG < 0, the reaction can proceed predominantly in the forward direction (a produ ...
Environmental Chemistry - Robert Morris University
... Metal Ions and Calcium in Water Calcium and Harness Hard water contains high concentrations of dissolved calcium and magnesium ions Soft water contains few of these dissolved ions. Counter ions of ...
... Metal Ions and Calcium in Water Calcium and Harness Hard water contains high concentrations of dissolved calcium and magnesium ions Soft water contains few of these dissolved ions. Counter ions of ...
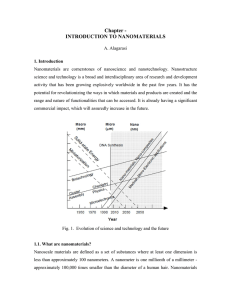
Chapter - INTRODUCTION TO NANOMATERIALS
... Today nanophase engineering expands in a rapidly growing number of structural and functional materials, both inorganic and organic, allowing to manipulate mechanical, catalytic, electric, magnetic, optical and electronic functions. The production of nanophase or cluster-assembled materials is usuall ...
... Today nanophase engineering expands in a rapidly growing number of structural and functional materials, both inorganic and organic, allowing to manipulate mechanical, catalytic, electric, magnetic, optical and electronic functions. The production of nanophase or cluster-assembled materials is usuall ...
Classification of
... a) Name two acids. _____lemon juice_____________ & ________vinegar____________ b) Name two bases. ______ammonia_______________ & _________baking soda________ c) What is the pH range of acids? ____1____ - _____6____ bases? ___8___ - ____14___ d) What is the pH of a neutral solution? ___7______ 14. Ex ...
... a) Name two acids. _____lemon juice_____________ & ________vinegar____________ b) Name two bases. ______ammonia_______________ & _________baking soda________ c) What is the pH range of acids? ____1____ - _____6____ bases? ___8___ - ____14___ d) What is the pH of a neutral solution? ___7______ 14. Ex ...
Document
... of a solid at its melting point into Constant of a liquid at its boiling point into liquid is heat of fusion. temperature! vapor is heat of vaporization. Qfusion = (mass) (spec.heat of fusion) Qvaporization = (mass) (spec.heat of vaporization) Example 1: How many joules is needed to change 20.0 g of ...
... of a solid at its melting point into Constant of a liquid at its boiling point into liquid is heat of fusion. temperature! vapor is heat of vaporization. Qfusion = (mass) (spec.heat of fusion) Qvaporization = (mass) (spec.heat of vaporization) Example 1: How many joules is needed to change 20.0 g of ...
Unit 8: Reactions
... 3. Double Replacement: A solution reaction in which the positive ion of one compound combines with the negative ion of the other compound to form a precipitate, and the other ions remain dissolved in solution. 4. Law of Conservation of Charge: Charge may not be created or destroyed by physical or ch ...
... 3. Double Replacement: A solution reaction in which the positive ion of one compound combines with the negative ion of the other compound to form a precipitate, and the other ions remain dissolved in solution. 4. Law of Conservation of Charge: Charge may not be created or destroyed by physical or ch ...
Redox

Redox reactions include all chemical reactions in which atoms have their oxidation state changed; in general, redox reactions involve the transfer of electrons between species. The term ""redox"" comes from two concepts involved with electron transfer: reduction and oxidation. It can be explained in simple terms: Oxidation is the loss of electrons or an increase in oxidation state by a molecule, atom, or ion. Reduction is the gain of electrons or a decrease in oxidation state by a molecule, atom, or ion.Although oxidation reactions are commonly associated with the formation of oxides from oxygen molecules, these are only specific examples of a more general concept of reactions involving electron transfer.Redox reactions, or oxidation-reduction reactions, have a number of similarities to acid–base reactions. Like acid–base reactions, redox reactions are a matched set, that is, there cannot be an oxidation reaction without a reduction reaction happening simultaneously. The oxidation alone and the reduction alone are each called a half-reaction, because two half-reactions always occur together to form a whole reaction. When writing half-reactions, the gained or lost electrons are typically included explicitly in order that the half-reaction be balanced with respect to electric charge.Though sufficient for many purposes, these descriptions are not precisely correct. Oxidation and reduction properly refer to a change in oxidation state — the actual transfer of electrons may never occur. The oxidation state of an atom is the fictitious charge that an atom would have if all bonds between atoms of different elements were 100% ionic. Thus, oxidation is better defined as an increase in oxidation state, and reduction as a decrease in oxidation state. In practice, the transfer of electrons will always cause a change in oxidation state, but there are many reactions that are classed as ""redox"" even though no electron transfer occurs (such as those involving covalent bonds).There are simple redox processes, such as the oxidation of carbon to yield carbon dioxide (CO2) or the reduction of carbon by hydrogen to yield methane (CH4), and more complex processes such as the oxidation of glucose (C6H12O6) in the human body through a series of complex electron transfer processes.