Chemical Bond – a force that holds two atoms together, the bond
... Chemical Bond – a force that holds two atoms together, the bond could be between two elements that are the same element or different elements. Ionic Bond – an electrostatic force between two different atomic elements (atomic nonmetal and an atomic metal) in which the atomic nonmetal steals the avail ...
... Chemical Bond – a force that holds two atoms together, the bond could be between two elements that are the same element or different elements. Ionic Bond – an electrostatic force between two different atomic elements (atomic nonmetal and an atomic metal) in which the atomic nonmetal steals the avail ...
2-1 Checkpoint - Jordan High School
... – Glycogen—animal starch; chain of glucose molecules used as energy ...
... – Glycogen—animal starch; chain of glucose molecules used as energy ...
112- Unit I -Electrochem -pdf
... becomes more positive. c) The more negative E° red , the weaker the tendency for the substance to be reduced. Actually, the greater the tendency of the substance to be oxidized and it is considered to be a strong reducing agent. 2) The half reactions are reversible. Depending on the conditions, a gi ...
... becomes more positive. c) The more negative E° red , the weaker the tendency for the substance to be reduced. Actually, the greater the tendency of the substance to be oxidized and it is considered to be a strong reducing agent. 2) The half reactions are reversible. Depending on the conditions, a gi ...
Document
... becomes negative by default because the metal must be oxidized and thus be positive. Remember …it has to do with electronegativity values and thus effective nuclear charge …. Hydrogen has a lesser electronegativity value than virtually any other nonmetal … BUT, its value of 2.2 is greater than any m ...
... becomes negative by default because the metal must be oxidized and thus be positive. Remember …it has to do with electronegativity values and thus effective nuclear charge …. Hydrogen has a lesser electronegativity value than virtually any other nonmetal … BUT, its value of 2.2 is greater than any m ...
Name___________________________________ Physical
... CHEMICAL REACTIONS - P. 582 12) The process in which the atoms of one or more substance are rearranged to form different substances is called a(n) ____________________. A) nuclear reaction B) substantiation C) chemical reaction D) physical reaction ...
... CHEMICAL REACTIONS - P. 582 12) The process in which the atoms of one or more substance are rearranged to form different substances is called a(n) ____________________. A) nuclear reaction B) substantiation C) chemical reaction D) physical reaction ...
Chemistry - StudyTime NZ
... Neither Oxygen nor Magnesium have full valence electron shells. Because of this, they must each lose or gain electrons in order to become stable. Oxygen has 8 electrons and hence an electron arrangement ...
... Neither Oxygen nor Magnesium have full valence electron shells. Because of this, they must each lose or gain electrons in order to become stable. Oxygen has 8 electrons and hence an electron arrangement ...
Chapter 4 Notes - Atomic Theory
... A skeleton equation shows only the formulas of the elements/compounds Shows atoms, but is not balanced K(s) + O2 (g) K2O(s) A balanced chemical equation shows the correct number of each atom Balancing ensures that the number of each atom is the same on both sides of the reaction arrow ...
... A skeleton equation shows only the formulas of the elements/compounds Shows atoms, but is not balanced K(s) + O2 (g) K2O(s) A balanced chemical equation shows the correct number of each atom Balancing ensures that the number of each atom is the same on both sides of the reaction arrow ...
CBSE/12th Class/2010/CHEMISTRY
... (ii)The E0 value for the Mn3/Mn2+ couple is much more positive than that for Cr3/Cr2+ couple or Fe3+/Fe2+ couple.Because Mn3+ has the outer electronic configuration of 3d4 and Mn2+ has the outer electronic configuration of 3d5. Thus, the conversion of Mn3+ to Mn2+ will be a favourable reaction since ...
... (ii)The E0 value for the Mn3/Mn2+ couple is much more positive than that for Cr3/Cr2+ couple or Fe3+/Fe2+ couple.Because Mn3+ has the outer electronic configuration of 3d4 and Mn2+ has the outer electronic configuration of 3d5. Thus, the conversion of Mn3+ to Mn2+ will be a favourable reaction since ...
Chemistry Module 1- Basic Revision Notes 1.1a Atomic Structure 1.1
... reactivity down the group, they also, are known as soft metals(i.e. can be cut easily by a knife) are low in density(i.e. they even float on water) are stored under paraffin (oil) due to their high reactivity with water react quickly with oxygen (in the air)and tarnish are shiny when newly ...
... reactivity down the group, they also, are known as soft metals(i.e. can be cut easily by a knife) are low in density(i.e. they even float on water) are stored under paraffin (oil) due to their high reactivity with water react quickly with oxygen (in the air)and tarnish are shiny when newly ...
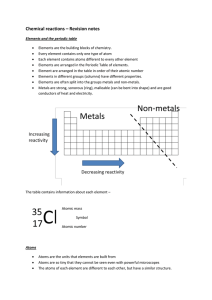
Chemical reactions revision
... Elements are the building blocks of chemistry. Every element contains only one type of atom Each element contains atoms different to every other element Elements are arranged in the Periodic Table of elements. Element are arranged in the table in order of their atomic number Elements in different gr ...
... Elements are the building blocks of chemistry. Every element contains only one type of atom Each element contains atoms different to every other element Elements are arranged in the Periodic Table of elements. Element are arranged in the table in order of their atomic number Elements in different gr ...
Cluster Fragmentation and Catalysis
... interest in the use of lanthanide ions for biochemical applications. Many organolanthanide complexes have found their way into mainstream science with varied uses. For example, many lanthanide-chelate complexes have been used as contrast agents for MRI, as probes in timefluorescence spectroscopy, ma ...
... interest in the use of lanthanide ions for biochemical applications. Many organolanthanide complexes have found their way into mainstream science with varied uses. For example, many lanthanide-chelate complexes have been used as contrast agents for MRI, as probes in timefluorescence spectroscopy, ma ...
Chemistry Test Review - Greenslime Home Page
... amu – charge is negative (-) 9. Name 3 elements that have the most similar chemical properties to Cesium. a. Any three of the hydrogen family (hydrogen, lithium, sodium, potassium, rubidium, francium) 10. Explain why chemical equation must be balanced. a. Due to the Law of Conservation of Mass; matt ...
... amu – charge is negative (-) 9. Name 3 elements that have the most similar chemical properties to Cesium. a. Any three of the hydrogen family (hydrogen, lithium, sodium, potassium, rubidium, francium) 10. Explain why chemical equation must be balanced. a. Due to the Law of Conservation of Mass; matt ...
Labs - newtunings.com
... 5.2i When a bond is broken, energy is absorbed. When a bond is formed, energy is released. 5.2j Electronegativity indicates how strongly an atom of an element attracts electrons in a chemical bond. Electronegativity values are assigned according to arbitrary scales. 5.2k The electronegativity differ ...
... 5.2i When a bond is broken, energy is absorbed. When a bond is formed, energy is released. 5.2j Electronegativity indicates how strongly an atom of an element attracts electrons in a chemical bond. Electronegativity values are assigned according to arbitrary scales. 5.2k The electronegativity differ ...
Title - Iowa State University
... 3. Which of the following statements about catalysts is false? a. A catalyst will speed up the rate of a reaction. b. Catalysts are used in very many commercially important chemical reactions. c. Catalytic converters are examples of heterogeneous catalysts. d. A catalyst can cause a nonspontaneous r ...
... 3. Which of the following statements about catalysts is false? a. A catalyst will speed up the rate of a reaction. b. Catalysts are used in very many commercially important chemical reactions. c. Catalytic converters are examples of heterogeneous catalysts. d. A catalyst can cause a nonspontaneous r ...
NYS Regents Chemistry June 21, 2002
... 1: II. PERIODIC TABLE\1. Properties of Elements\A. Metals\1. Metals - (32) 2: II. PERIODIC TABLE\2. Valence Electrons\A. Electron / Ionic Configuration\2. Ionic Configuration - (10, 30) 2: II. PERIODIC TABLE\4. Properties of Periods\C. Electronegativity\1. Electronegativity - (11, 13) 1: II. PERIODI ...
... 1: II. PERIODIC TABLE\1. Properties of Elements\A. Metals\1. Metals - (32) 2: II. PERIODIC TABLE\2. Valence Electrons\A. Electron / Ionic Configuration\2. Ionic Configuration - (10, 30) 2: II. PERIODIC TABLE\4. Properties of Periods\C. Electronegativity\1. Electronegativity - (11, 13) 1: II. PERIODI ...
+ H 2 O(g)
... Info on Decomp Reactions • Energy is usually need to make these reactions happen • Often hard to predict products unless the substance breaks into its ...
... Info on Decomp Reactions • Energy is usually need to make these reactions happen • Often hard to predict products unless the substance breaks into its ...
Periodic Table Jeopardy
... A substance that cannot be separated or broken down into simpler substances by chemical means. All atoms in this substance have the same atomic #. ...
... A substance that cannot be separated or broken down into simpler substances by chemical means. All atoms in this substance have the same atomic #. ...
Ch. 2-1 Nature of Matter
... constant rate over time, releasing energy – Although the radiation these isotopes give off can be dangerous, they have important scientific and practical uses. ...
... constant rate over time, releasing energy – Although the radiation these isotopes give off can be dangerous, they have important scientific and practical uses. ...
Reactions of common metals and properties of
... metals form salt-like hydrides, such as NaH. Such hydrides have similar crystal structures to alkali halides. However, they are extremely reactive towards water, forming the corresponding metal hydroxide and liberating hydrogen gas. These salt-like hydrides find some use as reducing agents in organi ...
... metals form salt-like hydrides, such as NaH. Such hydrides have similar crystal structures to alkali halides. However, they are extremely reactive towards water, forming the corresponding metal hydroxide and liberating hydrogen gas. These salt-like hydrides find some use as reducing agents in organi ...
Redox

Redox reactions include all chemical reactions in which atoms have their oxidation state changed; in general, redox reactions involve the transfer of electrons between species. The term ""redox"" comes from two concepts involved with electron transfer: reduction and oxidation. It can be explained in simple terms: Oxidation is the loss of electrons or an increase in oxidation state by a molecule, atom, or ion. Reduction is the gain of electrons or a decrease in oxidation state by a molecule, atom, or ion.Although oxidation reactions are commonly associated with the formation of oxides from oxygen molecules, these are only specific examples of a more general concept of reactions involving electron transfer.Redox reactions, or oxidation-reduction reactions, have a number of similarities to acid–base reactions. Like acid–base reactions, redox reactions are a matched set, that is, there cannot be an oxidation reaction without a reduction reaction happening simultaneously. The oxidation alone and the reduction alone are each called a half-reaction, because two half-reactions always occur together to form a whole reaction. When writing half-reactions, the gained or lost electrons are typically included explicitly in order that the half-reaction be balanced with respect to electric charge.Though sufficient for many purposes, these descriptions are not precisely correct. Oxidation and reduction properly refer to a change in oxidation state — the actual transfer of electrons may never occur. The oxidation state of an atom is the fictitious charge that an atom would have if all bonds between atoms of different elements were 100% ionic. Thus, oxidation is better defined as an increase in oxidation state, and reduction as a decrease in oxidation state. In practice, the transfer of electrons will always cause a change in oxidation state, but there are many reactions that are classed as ""redox"" even though no electron transfer occurs (such as those involving covalent bonds).There are simple redox processes, such as the oxidation of carbon to yield carbon dioxide (CO2) or the reduction of carbon by hydrogen to yield methane (CH4), and more complex processes such as the oxidation of glucose (C6H12O6) in the human body through a series of complex electron transfer processes.