Slide 1 - Mrs. Reed Science Classes
... For the reaction represented by the equation Mg + 2HCl H2 + MgCl2, calculate the percentage yield of magnesium chloride if 100. g of magnesium react with excess hydrochloric acid to yield 330. g of magnesium chloride. a. 71.8% c. 81.6% b. 74.3% d. 84.2% ...
... For the reaction represented by the equation Mg + 2HCl H2 + MgCl2, calculate the percentage yield of magnesium chloride if 100. g of magnesium react with excess hydrochloric acid to yield 330. g of magnesium chloride. a. 71.8% c. 81.6% b. 74.3% d. 84.2% ...
Unit 16 Worksheet - Jensen Chemistry
... 1. When do electrons release photons(packets of energy)? When the electrons: a. move to higher levels of energy b. return to their original energy level c increase orbital speed around the nucleus d. are released by the atom 2. Helium was discovered on the sun in 1868, almost 30 years before it was ...
... 1. When do electrons release photons(packets of energy)? When the electrons: a. move to higher levels of energy b. return to their original energy level c increase orbital speed around the nucleus d. are released by the atom 2. Helium was discovered on the sun in 1868, almost 30 years before it was ...
File
... The compound always burns in oxygen gas and always releases carbon dioxide and water. During incomplete combustion (a limited amt. of O2), carbon monoxide (CO) is also produced. Example: 2 C3H6 + 9 O2 6 CO2 + 6 H2O ...
... The compound always burns in oxygen gas and always releases carbon dioxide and water. During incomplete combustion (a limited amt. of O2), carbon monoxide (CO) is also produced. Example: 2 C3H6 + 9 O2 6 CO2 + 6 H2O ...
ch22_lecture_6e_final
... Elements are not equally abundant in all regions – abundances differ due to the differences in physical and chemical behavior of the elements. - The core of the Earth is rich in dense Group 8B(8) to 8B(10) metals. - The Earth’s crust has the largest share of nonmetals, metalloids, and light active m ...
... Elements are not equally abundant in all regions – abundances differ due to the differences in physical and chemical behavior of the elements. - The core of the Earth is rich in dense Group 8B(8) to 8B(10) metals. - The Earth’s crust has the largest share of nonmetals, metalloids, and light active m ...
2009-10 Chemistry 1st Semester Final Exam Topics and Review
... e. One of the problems with space travel is the building up of carbon dioxide produced by the astronauts. The typical procedure is to react the carbon dioxide with lithium hydroxide to form lithium carbonate and liquid water. 36. Write and balance a chemical equation for this reaction. a. Aluminum m ...
... e. One of the problems with space travel is the building up of carbon dioxide produced by the astronauts. The typical procedure is to react the carbon dioxide with lithium hydroxide to form lithium carbonate and liquid water. 36. Write and balance a chemical equation for this reaction. a. Aluminum m ...
STUDY GUIDE for DIGESTION and NUTRITION
... Determine how many electrons an element will gain or lose Draw electron transfer diagrams of ionic compounds Use combining capacities to write formulas of ionic compounds Write formulae and names of simple ionic compounds, multivalent ionic compounds, and compounds with polyatomic ions. Wr ...
... Determine how many electrons an element will gain or lose Draw electron transfer diagrams of ionic compounds Use combining capacities to write formulas of ionic compounds Write formulae and names of simple ionic compounds, multivalent ionic compounds, and compounds with polyatomic ions. Wr ...
File
... type of atoms in a molecule Simplest Formula gives the smallest whole-number ratios for the atoms present (NH2 – 1:2 ratio of nitrogen to hydrogen) Molecular Formula indicates the actual numbers of each type of atom per molecule (N2H4 – each molecule of hydrazine consists of two molecules of nit ...
... type of atoms in a molecule Simplest Formula gives the smallest whole-number ratios for the atoms present (NH2 – 1:2 ratio of nitrogen to hydrogen) Molecular Formula indicates the actual numbers of each type of atom per molecule (N2H4 – each molecule of hydrazine consists of two molecules of nit ...
Classifying Chemical Reactions 9-3
... We need one more oxygen in the products. Can’t change the formula, because it describes what it is (carbon monoxide in this example) ...
... We need one more oxygen in the products. Can’t change the formula, because it describes what it is (carbon monoxide in this example) ...
Chap 11 Sect 1 Notes Atomic Theory
... •Atom is composed of electrons surrounded by a “soup” of positive charge to balance the electron’s negative charge, like negativelycharged “plums” surrounded by ...
... •Atom is composed of electrons surrounded by a “soup” of positive charge to balance the electron’s negative charge, like negativelycharged “plums” surrounded by ...
File - Mr. Gittermann
... (lost the electron) and the other is negatively charged (gained the electron), the opposite charges attract forming an ionic bond • Ion: An atom or molecule that has gained or lost one or more electrons and has a negative or positive charge ...
... (lost the electron) and the other is negatively charged (gained the electron), the opposite charges attract forming an ionic bond • Ion: An atom or molecule that has gained or lost one or more electrons and has a negative or positive charge ...
23. Oxidation and Reduction
... equation for the reaction between Cu atoms and N3+ ions. The two half-reactions are: Cu ---> Cu2+ and 2 N3+ ---> N2 The 2 is needed in front of N 3+ in the second half-reaction because nitrogen is diatomic as a free element, and the equation must be balanced by mass. While the two half-equations are ...
... equation for the reaction between Cu atoms and N3+ ions. The two half-reactions are: Cu ---> Cu2+ and 2 N3+ ---> N2 The 2 is needed in front of N 3+ in the second half-reaction because nitrogen is diatomic as a free element, and the equation must be balanced by mass. While the two half-equations are ...
Honors Chemistry Semester 1 Exam Review
... 4. What type of bond is formed by the transferring of electrons? ______________ Sharing electrons? ___________ 5. How does an element form a cation?___________________________ Anion? __________________________ 6. Why does Magnesium form a +2 charge? __________________________________________________ ...
... 4. What type of bond is formed by the transferring of electrons? ______________ Sharing electrons? ___________ 5. How does an element form a cation?___________________________ Anion? __________________________ 6. Why does Magnesium form a +2 charge? __________________________________________________ ...
PRACTICE * Naming and Writing Ionic Compounds
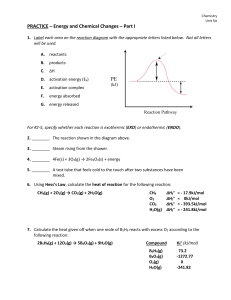
... 2. ________ The reaction shown in the diagram above. 3. ________ Steam rising from the shower. 4. ________ 4Fe(s) + 3O2(g) → 2Fe2O3(s) + energy 5. ________ A test tube that feels cold to the touch after two substances have been mixed. 6. Using Hess’s Law, calculate the heat of reaction for the follo ...
... 2. ________ The reaction shown in the diagram above. 3. ________ Steam rising from the shower. 4. ________ 4Fe(s) + 3O2(g) → 2Fe2O3(s) + energy 5. ________ A test tube that feels cold to the touch after two substances have been mixed. 6. Using Hess’s Law, calculate the heat of reaction for the follo ...
Sugárkémiai áttekintés Schiller Róbert
... H· + R-CH2-CH2-R’ H2+ R-C·H-CH2-R’ Generally speaking: bond cleavage and bond formation. Main product usually H2 The failure of the organic moderated reactors. But: chemistry of the nuclear reactors!! ...
... H· + R-CH2-CH2-R’ H2+ R-C·H-CH2-R’ Generally speaking: bond cleavage and bond formation. Main product usually H2 The failure of the organic moderated reactors. But: chemistry of the nuclear reactors!! ...
Metals and non-metals III IMPORTANT POINTS Non-metals
... 1. a. Magnesium, chromium and sodium are all metals, hence, they react with oxygen to form basic oxides b. Chromium, as it is a transition metal. Metals have high density and coloured compounds are formed by transition metals. c. Bromine - the formula is Br2, that is, two atoms of bromine. d. Bromin ...
... 1. a. Magnesium, chromium and sodium are all metals, hence, they react with oxygen to form basic oxides b. Chromium, as it is a transition metal. Metals have high density and coloured compounds are formed by transition metals. c. Bromine - the formula is Br2, that is, two atoms of bromine. d. Bromin ...
File
... • Formed by the attraction of oppositely charged ions • Gain or loss of electrons forms ions – Na atom loses an electron to become Na+ – Cl atom gains an electron to become Cl– – Opposite charges attract so that Na+ and Cl– remain associated as an ionic compound ...
... • Formed by the attraction of oppositely charged ions • Gain or loss of electrons forms ions – Na atom loses an electron to become Na+ – Cl atom gains an electron to become Cl– – Opposite charges attract so that Na+ and Cl– remain associated as an ionic compound ...
Chem Reactions (and Balancing Equations)
... molecule (O2) to form H2O. Then, the remaining oxygen atom combines with two more hydrogen atoms (from another H2 molecule) to make a second H2O molecule. ...
... molecule (O2) to form H2O. Then, the remaining oxygen atom combines with two more hydrogen atoms (from another H2 molecule) to make a second H2O molecule. ...
Primary electrons make random elastic and inelastic collision either
... effect…. i.e. as to pass though the stronger electric filed, close to nuclei, it will suffer a “quantum jump” to a low energy state, which will make emission of X-ray photon, and it would be all possible energy up to E0… Secondary electron, (<50 eV, normally around 2-6 eV, larger than sample’s work ...
... effect…. i.e. as to pass though the stronger electric filed, close to nuclei, it will suffer a “quantum jump” to a low energy state, which will make emission of X-ray photon, and it would be all possible energy up to E0… Secondary electron, (<50 eV, normally around 2-6 eV, larger than sample’s work ...
Chapter 2
... • Formed by the attraction of oppositely charged ions • Gain or loss of electrons forms ions – Na atom loses an electron to become Na+ – Cl atom gains an electron to become Cl– – Opposite charges attract so that Na+ and Cl– remain associated as an ionic compound ...
... • Formed by the attraction of oppositely charged ions • Gain or loss of electrons forms ions – Na atom loses an electron to become Na+ – Cl atom gains an electron to become Cl– – Opposite charges attract so that Na+ and Cl– remain associated as an ionic compound ...
key - Greenslime.info
... is sodium, followed by magnesium, boron and then carbon. Why? Sodium only has one valence electron to lose in order to react. Magnesium has two valance electrons, boron has three, and carbon has four. The fewer the valance electrons that have to be either gained or lost to reach a complete valance e ...
... is sodium, followed by magnesium, boron and then carbon. Why? Sodium only has one valence electron to lose in order to react. Magnesium has two valance electrons, boron has three, and carbon has four. The fewer the valance electrons that have to be either gained or lost to reach a complete valance e ...
Document
... dry-cells (batteries) are such that the potential is always about 1.5 V. This has become a standard and is now a limiting factor in deciding which chemicals can be used to create a battery. The cell potential is given a symbol of Ecell. If all chemicals are at activity of 1 (conc. = 1 M, p = 1 bar) ...
... dry-cells (batteries) are such that the potential is always about 1.5 V. This has become a standard and is now a limiting factor in deciding which chemicals can be used to create a battery. The cell potential is given a symbol of Ecell. If all chemicals are at activity of 1 (conc. = 1 M, p = 1 bar) ...
Redox

Redox reactions include all chemical reactions in which atoms have their oxidation state changed; in general, redox reactions involve the transfer of electrons between species. The term ""redox"" comes from two concepts involved with electron transfer: reduction and oxidation. It can be explained in simple terms: Oxidation is the loss of electrons or an increase in oxidation state by a molecule, atom, or ion. Reduction is the gain of electrons or a decrease in oxidation state by a molecule, atom, or ion.Although oxidation reactions are commonly associated with the formation of oxides from oxygen molecules, these are only specific examples of a more general concept of reactions involving electron transfer.Redox reactions, or oxidation-reduction reactions, have a number of similarities to acid–base reactions. Like acid–base reactions, redox reactions are a matched set, that is, there cannot be an oxidation reaction without a reduction reaction happening simultaneously. The oxidation alone and the reduction alone are each called a half-reaction, because two half-reactions always occur together to form a whole reaction. When writing half-reactions, the gained or lost electrons are typically included explicitly in order that the half-reaction be balanced with respect to electric charge.Though sufficient for many purposes, these descriptions are not precisely correct. Oxidation and reduction properly refer to a change in oxidation state — the actual transfer of electrons may never occur. The oxidation state of an atom is the fictitious charge that an atom would have if all bonds between atoms of different elements were 100% ionic. Thus, oxidation is better defined as an increase in oxidation state, and reduction as a decrease in oxidation state. In practice, the transfer of electrons will always cause a change in oxidation state, but there are many reactions that are classed as ""redox"" even though no electron transfer occurs (such as those involving covalent bonds).There are simple redox processes, such as the oxidation of carbon to yield carbon dioxide (CO2) or the reduction of carbon by hydrogen to yield methane (CH4), and more complex processes such as the oxidation of glucose (C6H12O6) in the human body through a series of complex electron transfer processes.