Introduction to Chemistry for Coach Keith`s Biology
... electrons spinning in energy levels around the center The nucleus is the center of an atom where most of the mass is concentrated Protons are positively charged ( p+ ), have a mass of 1 amu (atomic mass unit) , are found in the nucleus, and determine the atomic number of the element Example: Carbon ...
... electrons spinning in energy levels around the center The nucleus is the center of an atom where most of the mass is concentrated Protons are positively charged ( p+ ), have a mass of 1 amu (atomic mass unit) , are found in the nucleus, and determine the atomic number of the element Example: Carbon ...
Exam 3 - Canvas by Instructure
... 4. Over 60 MILLION TONS of nitric acid is produced across the world annually, most of which goes towards the production of synthetic fertilizers and explosives. The first step in synthesizing nitric acid starts with ammonia reacting with oxygen gas according to the equation below. When properly bala ...
... 4. Over 60 MILLION TONS of nitric acid is produced across the world annually, most of which goes towards the production of synthetic fertilizers and explosives. The first step in synthesizing nitric acid starts with ammonia reacting with oxygen gas according to the equation below. When properly bala ...
Dr. Ali Ebneshahidi
... Organic chemistry – chemistry that deals with organic substances (those that contain carbon and hydrogen). Biochemistry - chemistry of living organisms; essential for understanding physiology because body functions involve chemical changes that occur within cells. Matter – anything that has we ...
... Organic chemistry – chemistry that deals with organic substances (those that contain carbon and hydrogen). Biochemistry - chemistry of living organisms; essential for understanding physiology because body functions involve chemical changes that occur within cells. Matter – anything that has we ...
Topic 9 Reduction and Oxidation File
... Electrolytic cell: Used to make non-spontaneous redox reactions occur by providing energy in the form of electricity from an external source. Electroplating: A process of coating one metal with a thin layer of another metal, by electrolysis. Half cell: A metal in contact with an aqueous solution of ...
... Electrolytic cell: Used to make non-spontaneous redox reactions occur by providing energy in the form of electricity from an external source. Electroplating: A process of coating one metal with a thin layer of another metal, by electrolysis. Half cell: A metal in contact with an aqueous solution of ...
Types of Chemical Reactions
... • For example, when ammonia and hydrogen chloride vapours combine, they form a white smoke as solid particles of ammonium chloride are formed. ...
... • For example, when ammonia and hydrogen chloride vapours combine, they form a white smoke as solid particles of ammonium chloride are formed. ...
Chapter 7 Chemical Reactions
... 3. Iron (III) oxide is formed when Iron is oxidized. 4. pure copper and sulfur dioxide are produced by heating copper( II) sulfide in the presence of oxygen 5, Water is formed by the explosive reaction between hydrogen and oxygen 7.2 Writing Chemical Equations In chemistry to communicate more effe ...
... 3. Iron (III) oxide is formed when Iron is oxidized. 4. pure copper and sulfur dioxide are produced by heating copper( II) sulfide in the presence of oxygen 5, Water is formed by the explosive reaction between hydrogen and oxygen 7.2 Writing Chemical Equations In chemistry to communicate more effe ...
Chemical Compounds
... S Take your ion and find someone you can bond with S Attempt to create the compound H2O, MgCl2..and so on S We will come together as a class and try to figure out if you ...
... S Take your ion and find someone you can bond with S Attempt to create the compound H2O, MgCl2..and so on S We will come together as a class and try to figure out if you ...
CHEM 13 NEWS EXAM 1998 - University of Waterloo
... Fe loses electrons less readily than Mg, making Mg the anode. ...
... Fe loses electrons less readily than Mg, making Mg the anode. ...
chemisty_ass_2
... They do not dissociate into ions when dissolved in water They have low melting and boiling point ...
... They do not dissociate into ions when dissolved in water They have low melting and boiling point ...
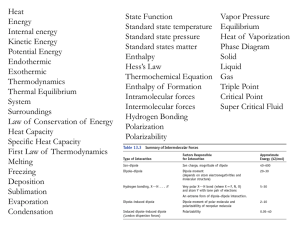
Chemistry Cram Sheet
... After studying about recycling, members of John’s biology class investigated the effect of various recycled products on plant growth. John’s lab group compared the effect of different aged grass compost on bean plants. Because decomposition is necessary for release of nutrients, the group hypothesiz ...
... After studying about recycling, members of John’s biology class investigated the effect of various recycled products on plant growth. John’s lab group compared the effect of different aged grass compost on bean plants. Because decomposition is necessary for release of nutrients, the group hypothesiz ...
Answers
... a) What is the empirical formula for the substance? b) What is the molecular formula for the substance? 8) How many liters of ammonia gas would be produced by 3.00 grams of magnesium nitride, according to the reaction Mg3N2 + 6 H2O --> 3 Mg(OH)2 + 2 NH3? 9) Solid iron (II) sulfide reacts with aqueou ...
... a) What is the empirical formula for the substance? b) What is the molecular formula for the substance? 8) How many liters of ammonia gas would be produced by 3.00 grams of magnesium nitride, according to the reaction Mg3N2 + 6 H2O --> 3 Mg(OH)2 + 2 NH3? 9) Solid iron (II) sulfide reacts with aqueou ...
OCR Chemistry C2 - Wey Valley School
... extracted by heating its ore with carbon; can be purified by electrolysis; recycling is cheaper/saves resources electrolysis; impure copper as anode; pure copper as cathode; copper (II) sulphate solution as electrolyte mixture of two elements one of which is a metal brass (copper/zinc); bronze (copp ...
... extracted by heating its ore with carbon; can be purified by electrolysis; recycling is cheaper/saves resources electrolysis; impure copper as anode; pure copper as cathode; copper (II) sulphate solution as electrolyte mixture of two elements one of which is a metal brass (copper/zinc); bronze (copp ...
File
... Molecular equation: KCl(aq)+AgNO3(aq)AgCl(s)+KNO3(aq) Complete ionic equation: K+(aq)+Cl-(aq)+Ag++NO3-AgCl(s)+K+(aq)+NO3-(aq) In this case, K+ and NO3- do not precipitate, therefore they are spectator ions. Net ionic equation: leaving out the spectator ions, we get Cl-(aq)+Ag+(aq)AgCl(s) ...
... Molecular equation: KCl(aq)+AgNO3(aq)AgCl(s)+KNO3(aq) Complete ionic equation: K+(aq)+Cl-(aq)+Ag++NO3-AgCl(s)+K+(aq)+NO3-(aq) In this case, K+ and NO3- do not precipitate, therefore they are spectator ions. Net ionic equation: leaving out the spectator ions, we get Cl-(aq)+Ag+(aq)AgCl(s) ...
KEY CONCEPT Enzymes are catalysts for chemical
... A catalyst lowers activation energy. • Catalysts are substances that speed up chemical reactions. – decrease activation energy – increase reaction rate ...
... A catalyst lowers activation energy. • Catalysts are substances that speed up chemical reactions. – decrease activation energy – increase reaction rate ...
Chem 101 notes review
... 1. draw the Lewis dot structure 2. draw circles around each atom and the electrons associated with it. Remember that formal charges are associated with covalent bonds and that all electrons are shared equally. 3. compare to the group number for that atom. If the number is larger the formal charge is ...
... 1. draw the Lewis dot structure 2. draw circles around each atom and the electrons associated with it. Remember that formal charges are associated with covalent bonds and that all electrons are shared equally. 3. compare to the group number for that atom. If the number is larger the formal charge is ...
Oregon State University, Summer 2009 Chemistry 121 Midterm
... Fill in the front page of the Scantron answer sheet with your last name, first name, middle initial, and student identification number. Leave the class section number and the test form number blank. This exam consists of 20 multiple-choice questions. Each multiple-choice question has 5 points associ ...
... Fill in the front page of the Scantron answer sheet with your last name, first name, middle initial, and student identification number. Leave the class section number and the test form number blank. This exam consists of 20 multiple-choice questions. Each multiple-choice question has 5 points associ ...
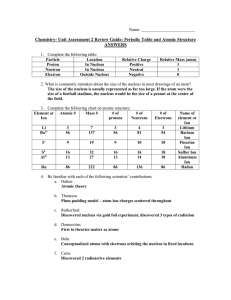
Chem Unit 2 Review Guide ANSWERS
... number (protons), but different masses because Hydrogen-2 has one neutron, while Hydrogen-3 has two neutrons. 8. Rubidium has two common isotopes, Rb-85 and Rb-87. If the atomic mass of Rubidium is 85.56 amu, what isotope is more abundant? Explain. Rb-85 is more abundant. If the amounts were equal, ...
... number (protons), but different masses because Hydrogen-2 has one neutron, while Hydrogen-3 has two neutrons. 8. Rubidium has two common isotopes, Rb-85 and Rb-87. If the atomic mass of Rubidium is 85.56 amu, what isotope is more abundant? Explain. Rb-85 is more abundant. If the amounts were equal, ...
Group 2 - UC Davis Canvas
... halogen is able to displace the members of the group below it, but not those above it. The only halogen with sufficient oxidizing power to displace O2(g) from water is F2(g). None of the halogens reacts with water to form H2(g). ...
... halogen is able to displace the members of the group below it, but not those above it. The only halogen with sufficient oxidizing power to displace O2(g) from water is F2(g). None of the halogens reacts with water to form H2(g). ...
FINAL EXAM REVIEW
... e) __Fe(HCO3)3 + __MgSe → __Fe2Se3 + __Mg(HCO3)2 f) __Br2 + __KCl → __KBr +__ Cl2 2) Complete and balance the following reactions and classify each reaction a) NO2 → b) HCl + KOH → c) Cu + Fe(NO3)2 → d) MgSO4 + BeCl2 → 3) How many grams of sodium oxide are produced when 73.2g of sodium react complet ...
... e) __Fe(HCO3)3 + __MgSe → __Fe2Se3 + __Mg(HCO3)2 f) __Br2 + __KCl → __KBr +__ Cl2 2) Complete and balance the following reactions and classify each reaction a) NO2 → b) HCl + KOH → c) Cu + Fe(NO3)2 → d) MgSO4 + BeCl2 → 3) How many grams of sodium oxide are produced when 73.2g of sodium react complet ...
Classifying Reactions: A good summary
... chemical substance undergoes both oxidation and reduction. NO2 and H2O2 are classic chemicals that have this ability. Example: 3NO2(g) + H2O ...
... chemical substance undergoes both oxidation and reduction. NO2 and H2O2 are classic chemicals that have this ability. Example: 3NO2(g) + H2O ...
DALTON`S ATOMIC THEORY - 1808: Publication of Dalton`s "A New
... You can't have a compound form in a chemical reaction that contains an element that was not in your starting materials. You can only make a certain amount of desired product from a fixed amount of starting material. ...
... You can't have a compound form in a chemical reaction that contains an element that was not in your starting materials. You can only make a certain amount of desired product from a fixed amount of starting material. ...
Honors Chemistry
... 10. Give the different waves of the magnetic spectrum. 11. Which wave has more energy: red or blue? Short or long? Microwave or x-ray? 12. What does Bohr’s Model say about the hydrogen atom? 13. What does it mean when an electron is excited? What happens when the excited electron returns to the grou ...
... 10. Give the different waves of the magnetic spectrum. 11. Which wave has more energy: red or blue? Short or long? Microwave or x-ray? 12. What does Bohr’s Model say about the hydrogen atom? 13. What does it mean when an electron is excited? What happens when the excited electron returns to the grou ...
AP Semestar Exam REVIEW
... d. enthalpy is a state property. e. in an endothermic process heat flows from the surroundings into the system. ...
... d. enthalpy is a state property. e. in an endothermic process heat flows from the surroundings into the system. ...
Redox

Redox reactions include all chemical reactions in which atoms have their oxidation state changed; in general, redox reactions involve the transfer of electrons between species. The term ""redox"" comes from two concepts involved with electron transfer: reduction and oxidation. It can be explained in simple terms: Oxidation is the loss of electrons or an increase in oxidation state by a molecule, atom, or ion. Reduction is the gain of electrons or a decrease in oxidation state by a molecule, atom, or ion.Although oxidation reactions are commonly associated with the formation of oxides from oxygen molecules, these are only specific examples of a more general concept of reactions involving electron transfer.Redox reactions, or oxidation-reduction reactions, have a number of similarities to acid–base reactions. Like acid–base reactions, redox reactions are a matched set, that is, there cannot be an oxidation reaction without a reduction reaction happening simultaneously. The oxidation alone and the reduction alone are each called a half-reaction, because two half-reactions always occur together to form a whole reaction. When writing half-reactions, the gained or lost electrons are typically included explicitly in order that the half-reaction be balanced with respect to electric charge.Though sufficient for many purposes, these descriptions are not precisely correct. Oxidation and reduction properly refer to a change in oxidation state — the actual transfer of electrons may never occur. The oxidation state of an atom is the fictitious charge that an atom would have if all bonds between atoms of different elements were 100% ionic. Thus, oxidation is better defined as an increase in oxidation state, and reduction as a decrease in oxidation state. In practice, the transfer of electrons will always cause a change in oxidation state, but there are many reactions that are classed as ""redox"" even though no electron transfer occurs (such as those involving covalent bonds).There are simple redox processes, such as the oxidation of carbon to yield carbon dioxide (CO2) or the reduction of carbon by hydrogen to yield methane (CH4), and more complex processes such as the oxidation of glucose (C6H12O6) in the human body through a series of complex electron transfer processes.