Chemical Equations and Stoichiometry
... Revolution. Hydrogen would be a more environmentally-friendly fuel. Based on what you know about chemistry, would hydrogen burn to produce .... ...
... Revolution. Hydrogen would be a more environmentally-friendly fuel. Based on what you know about chemistry, would hydrogen burn to produce .... ...
Chemistry Standardized Test Practice: Student Edition
... Welcome to Chemistry Standardized Test Practice This workbook is designed to strengthen your knowledge of the NSCS (National Science Content Standards) and provide additional chapter review of your Glencoe textbook, Chemistry: Matter and Change. For each chapter in the Glencoe textbook, Chemistry: M ...
... Welcome to Chemistry Standardized Test Practice This workbook is designed to strengthen your knowledge of the NSCS (National Science Content Standards) and provide additional chapter review of your Glencoe textbook, Chemistry: Matter and Change. For each chapter in the Glencoe textbook, Chemistry: M ...
North Carolina Test of Chemistry RELEASED
... North Carolina Test of Chemistry. Form A RELEASED Fall 2009 ...
... North Carolina Test of Chemistry. Form A RELEASED Fall 2009 ...
Chemistry 12 - Correspondence Studies
... calorimeter's bomb itself. The calorimeter is usually calibrated, showing the heat capacity of the entire bomb calorimeter. Knowing the heat capacity of the calorimeter in J/ºC and knowing the temperature change, ∆t, we can calculate the energy transferred from the combustion reaction to the entire ...
... calorimeter's bomb itself. The calorimeter is usually calibrated, showing the heat capacity of the entire bomb calorimeter. Knowing the heat capacity of the calorimeter in J/ºC and knowing the temperature change, ∆t, we can calculate the energy transferred from the combustion reaction to the entire ...
Acids - Beck-Shop
... 1 Add a measured volume of one solution to a conical flask using a pipette. 2 Add the other solution to a burette, and record the initial burette reading to the nearest 0.05 cm3. 3 Add a few drops of an indicator to the solution in the conical flask. 4 Run the solution in the burette into the so ...
... 1 Add a measured volume of one solution to a conical flask using a pipette. 2 Add the other solution to a burette, and record the initial burette reading to the nearest 0.05 cm3. 3 Add a few drops of an indicator to the solution in the conical flask. 4 Run the solution in the burette into the so ...
CHEM 1411 Exam #2 - HCC Learning Web
... 22. Physical states have to be included for both equations in a & b for full credit. a. Complete the following reaction Pb(NO3)2 (aq) + NaCl(aq) b. Write the complete ionic equation c. Identify the spectator ions ...
... 22. Physical states have to be included for both equations in a & b for full credit. a. Complete the following reaction Pb(NO3)2 (aq) + NaCl(aq) b. Write the complete ionic equation c. Identify the spectator ions ...
Gases - Chemistry 504
... **Note: For temperature the proportional situation (direct relationship) is only occurs for the Kelvin scale, therefore when using this equation the temperature must be in Kelvin.** We obtain the Combined gas law equation where the amount (moles) of gas does not change (cancels out). P1 V1 = P2 V2 n ...
... **Note: For temperature the proportional situation (direct relationship) is only occurs for the Kelvin scale, therefore when using this equation the temperature must be in Kelvin.** We obtain the Combined gas law equation where the amount (moles) of gas does not change (cancels out). P1 V1 = P2 V2 n ...
Solubility of Hexafluorobenzene in Aqueous Salt Solutions from
... the experimental measured solubilities for the (ethylbenzene + water) system at several temperatures. A comparison between the measured and literature data is presented in Figure 1. The good agreement between the literature and the experimental values measured here shows the reliability of the techn ...
... the experimental measured solubilities for the (ethylbenzene + water) system at several temperatures. A comparison between the measured and literature data is presented in Figure 1. The good agreement between the literature and the experimental values measured here shows the reliability of the techn ...
ACTIVATION ENERGY VARIATION DURING IGNITION OF
... occurrence. A very specific property characterizing the system is the induction period of the ignition and its dependence on the mixture pressure and composition as well as on the surface temperature [5y7]. The variation of this property on the system variables gives valuable information on the kine ...
... occurrence. A very specific property characterizing the system is the induction period of the ignition and its dependence on the mixture pressure and composition as well as on the surface temperature [5y7]. The variation of this property on the system variables gives valuable information on the kine ...
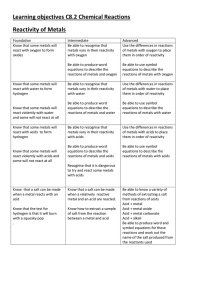
Learning objectives C8.2 Chemical Reactions Reactivity of Metals
... Know how to extract a sample of salt from the reaction between a metal and acid ...
... Know how to extract a sample of salt from the reaction between a metal and acid ...
Introduction to Qualitative Analysis
... Disproportionation (self-redox) reactions are a special case of redox reactions wherein part of the reagent is oxidized, and an equivalent part, according to the stoichiometry, is reduced. An example is the spontaneous decomposition of hydrogen peroxide where oxygen, in the –1 oxidation state in per ...
... Disproportionation (self-redox) reactions are a special case of redox reactions wherein part of the reagent is oxidized, and an equivalent part, according to the stoichiometry, is reduced. An example is the spontaneous decomposition of hydrogen peroxide where oxygen, in the –1 oxidation state in per ...
9.1 REDOX Introduction to Oxidation and Reduction
... e.g. MgO (Magnesium is +2 and Oxygen is -2) ...
... e.g. MgO (Magnesium is +2 and Oxygen is -2) ...
The Chemical Composition of the Earth`s Original Atmosphere
... Composition given as volume percent for each gas excluding water. Water is given as percentage of total gases. ...
... Composition given as volume percent for each gas excluding water. Water is given as percentage of total gases. ...
AP 2005 Chemistry Free-Response Questions
... one end that has been ignited and extinguished, but still contains hot, glowing, partially burned wood). (b) The following three mixtures have been prepared: CaO plus water, SiO2 plus water, and CO2 plus water. For each mixture, predict whether the pH is less than 7, equal to 7, or greater than 7. J ...
... one end that has been ignited and extinguished, but still contains hot, glowing, partially burned wood). (b) The following three mixtures have been prepared: CaO plus water, SiO2 plus water, and CO2 plus water. For each mixture, predict whether the pH is less than 7, equal to 7, or greater than 7. J ...
Chapter 19.1 Balancing Redox Equations
... Place the following compounds, methyl alcohol, CH3OH, oxygen, O2, and hydrogen chloride, HCl, in order of increasing melting point. ...
... Place the following compounds, methyl alcohol, CH3OH, oxygen, O2, and hydrogen chloride, HCl, in order of increasing melting point. ...
CHEMICAL REACTIONS Chapter 4
... 4 Al atoms + 3 O2 molecules ------> 2 formula units of Al2O3 4 moles of Al + 3 moles of O2 ----> 2 moles of Al2O3 ...
... 4 Al atoms + 3 O2 molecules ------> 2 formula units of Al2O3 4 moles of Al + 3 moles of O2 ----> 2 moles of Al2O3 ...
6.1 Moles and Molar Masses
... These calculations can also be applied to determine the number of water molecules within a hydrate. Instead of individual elements, compare the ratio of the compound to that of water. Ex.10) Copper (II) sulfate exists as a hydrate. In lab, a 2.60 g sample of the hydrate is heated in a crucible for ...
... These calculations can also be applied to determine the number of water molecules within a hydrate. Instead of individual elements, compare the ratio of the compound to that of water. Ex.10) Copper (II) sulfate exists as a hydrate. In lab, a 2.60 g sample of the hydrate is heated in a crucible for ...
1. Define the following term: system. A) The part of the universe that
... 41. What is the value of H if the equation is multiplied throughout by 2? A) –1452.8 kJ B) –2905.6 kJ C) -726.4 kJ 42. What is the value of H if the reaction is reversed so that the products become the reactants and vice versa? A) 0 kJ B) –1452.8 kJ C) 1452.8 kJ 43. What would the value of H be i ...
... 41. What is the value of H if the equation is multiplied throughout by 2? A) –1452.8 kJ B) –2905.6 kJ C) -726.4 kJ 42. What is the value of H if the reaction is reversed so that the products become the reactants and vice versa? A) 0 kJ B) –1452.8 kJ C) 1452.8 kJ 43. What would the value of H be i ...
RES8_chemcontentchecklist
... Explain that addition reactions have an atom economy of 100% whereas substitution reactions are less efficient. Carry out calculations to determine the atom economy of a reaction. Describe the benefits of developing chemical processes with a high atom economy in terms of fewer waste materials. Expla ...
... Explain that addition reactions have an atom economy of 100% whereas substitution reactions are less efficient. Carry out calculations to determine the atom economy of a reaction. Describe the benefits of developing chemical processes with a high atom economy in terms of fewer waste materials. Expla ...
1.7AMINES
... 1. Amines have an unpleasant odours. (the odour of decaying fish is attributed to simple amines). 2. Because of the hydrogen bonding amines have higher BP than hydrocarbons of similar molar mass. Since a nitrogen—hydrogen bond is less polar than an oxygen—hydrogen bond, the hydrogen bonding in prima ...
... 1. Amines have an unpleasant odours. (the odour of decaying fish is attributed to simple amines). 2. Because of the hydrogen bonding amines have higher BP than hydrocarbons of similar molar mass. Since a nitrogen—hydrogen bond is less polar than an oxygen—hydrogen bond, the hydrogen bonding in prima ...
Explained answers - Admissions Testing Service
... Bile is alkaline and neutralises the acid material released from the stomach into the small intestine. This will increase the pH rather than reduce it. Therefore statement 2 is incorrect. Lipase is not present in human bile. Therefore statement 3 is incorrect. The answer is B ...
... Bile is alkaline and neutralises the acid material released from the stomach into the small intestine. This will increase the pH rather than reduce it. Therefore statement 2 is incorrect. Lipase is not present in human bile. Therefore statement 3 is incorrect. The answer is B ...
AP Chemistry - West Bloomfield School District
... In a certain experiment, 6.00 g of aluminum is burned in 24.0 g of bromine. What is the maximum amount of aluminum bromide that can be produced? 67. Acid-base neutralization reactions are very common in industrial processes. This is the reaction of sulfuric acid with sodium hydroxide: H2SO4 (aq) + 2 ...
... In a certain experiment, 6.00 g of aluminum is burned in 24.0 g of bromine. What is the maximum amount of aluminum bromide that can be produced? 67. Acid-base neutralization reactions are very common in industrial processes. This is the reaction of sulfuric acid with sodium hydroxide: H2SO4 (aq) + 2 ...
CHAPtER 9 Properties and reactions of organic compounds
... It is interesting to note that the melting points do not follow the same pattern as the boiling points. In the solid state, the trans isomers can pack more closely than the cis isomers, making the intermolecular forces more effective. cis and trans isomers can also occur in ring structures. cis–tran ...
... It is interesting to note that the melting points do not follow the same pattern as the boiling points. In the solid state, the trans isomers can pack more closely than the cis isomers, making the intermolecular forces more effective. cis and trans isomers can also occur in ring structures. cis–tran ...
practice test 4 CHM 112
... write an equilibrium constant in the usual way except that here one has concentrations that refer to solids in the solid solution. Determine the equilibrium constant for the formation of cementite from iron and carbon at 680C. [Given: for this reaction at 25C, H = 21 kJ/mol and S = 20.4 J/mol· ...
... write an equilibrium constant in the usual way except that here one has concentrations that refer to solids in the solid solution. Determine the equilibrium constant for the formation of cementite from iron and carbon at 680C. [Given: for this reaction at 25C, H = 21 kJ/mol and S = 20.4 J/mol· ...
Electrolysis of water
Electrolysis of water is the decomposition of water (H2O) into oxygen (O2) and hydrogen gas (H2) due to an electric current being passed through the water.This technique can be used to make hydrogen fuel (hydrogen gas) and breathable oxygen; though currently most industrial methods make hydrogen fuel from natural gas instead.