Sustainable Oxidation Catalysis for Synthesis
... problematic on a larger scale. There is a need to develop efficient catalysts that use sustainable terminal oxidants such as molecular oxygen or hydrogen peroxide. Although such methods are employed in the preparation of commodity chemicals, they are rarely used for the synthesis of fine chemicals, ...
... problematic on a larger scale. There is a need to develop efficient catalysts that use sustainable terminal oxidants such as molecular oxygen or hydrogen peroxide. Although such methods are employed in the preparation of commodity chemicals, they are rarely used for the synthesis of fine chemicals, ...
8.4 Weak Acids and Bases, Continued
... The Autoionization of Water, Kw • Water can act as either a weak acid or a base depending on whether a base or acid is present in solution. • In pure water, the water molecules spontaneously react with each other as shown. ...
... The Autoionization of Water, Kw • Water can act as either a weak acid or a base depending on whether a base or acid is present in solution. • In pure water, the water molecules spontaneously react with each other as shown. ...
Chapter 4: Types of Chemical Reactions and Solution Stoichiometry
... Strong Electrolytes • Produce ions in aqueous solution and conduct electricity well. • Strong electrolytes are soluble salts, strong acids and strong bases. • Strong acids produce H+ ions when they dissolve in water. • HCl, … , HNO3 and H2SO4 are strong acids HNO3(aq) → H+(aq) + NO3-(aq) • NaOH and ...
... Strong Electrolytes • Produce ions in aqueous solution and conduct electricity well. • Strong electrolytes are soluble salts, strong acids and strong bases. • Strong acids produce H+ ions when they dissolve in water. • HCl, … , HNO3 and H2SO4 are strong acids HNO3(aq) → H+(aq) + NO3-(aq) • NaOH and ...
Aromatic Chemistry - heckgrammar.co.uk
... the concentration wrong and therefore the pH another likely error with a weak acid where xs alkali has not been added is assuming that the remaining acid is fully dissociated i.e. forgetting to use Ka to determine [H+(aq)] ...
... the concentration wrong and therefore the pH another likely error with a weak acid where xs alkali has not been added is assuming that the remaining acid is fully dissociated i.e. forgetting to use Ka to determine [H+(aq)] ...
unit-4-notes-1_enthalpy-and-entropy
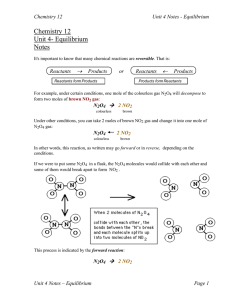
... and no NO2. The forward reaction rate was high at the start, but the reverse reaction rate eventually "caught up", the rates became equal and equilibrium was established. Can you guess what would happen if we had started with pure NO2 instead (no N2O4 )? The reverse rate would start out high and the ...
... and no NO2. The forward reaction rate was high at the start, but the reverse reaction rate eventually "caught up", the rates became equal and equilibrium was established. Can you guess what would happen if we had started with pure NO2 instead (no N2O4 )? The reverse rate would start out high and the ...
(NH 3 ) 2 - GZ @ Science Class Online
... Carry out procedures to identify ions involves collecting primary data and using these observations to identify ions in a solution using a procedure provided. Identification of ions must be supported by experimental observations and identification of all precipitates formed. Ions to be identified wi ...
... Carry out procedures to identify ions involves collecting primary data and using these observations to identify ions in a solution using a procedure provided. Identification of ions must be supported by experimental observations and identification of all precipitates formed. Ions to be identified wi ...
At equilibrium
... After studying this lecture you should be able to: • Understand the concepts of: the chemical equilibrium condition, dynamic equilibrium as the balance of forward and reverse reaction rates. • Know the definition of Le Chatelier’s Principle, and understand its application to the prediction of the di ...
... After studying this lecture you should be able to: • Understand the concepts of: the chemical equilibrium condition, dynamic equilibrium as the balance of forward and reverse reaction rates. • Know the definition of Le Chatelier’s Principle, and understand its application to the prediction of the di ...
Solutions Manual
... Cellulose is made from repeating units of β-glucose with inversion of every second unit. This produces long, straight chains of cellulose which are linked to each other by hydrogen bonding. In plants, cellulose acts as a structural material. Starch (both amylase and amylopectin) is made from long-ch ...
... Cellulose is made from repeating units of β-glucose with inversion of every second unit. This produces long, straight chains of cellulose which are linked to each other by hydrogen bonding. In plants, cellulose acts as a structural material. Starch (both amylase and amylopectin) is made from long-ch ...
The reaction pathways of hydrogen peroxide in
... enthalpy, entropy and free energy of the transition states of the formation and breakdown of the intermediate have been calculated. The metal-catalyzed pathway of hydrogen peroxide is dealing with the effect of hydroxyl radicals created by the Fenton reaction and their potential to oxidize the disul ...
... enthalpy, entropy and free energy of the transition states of the formation and breakdown of the intermediate have been calculated. The metal-catalyzed pathway of hydrogen peroxide is dealing with the effect of hydroxyl radicals created by the Fenton reaction and their potential to oxidize the disul ...
Chapter 4 Aqueous Reactions and Solution Stoichiometry
... • To form the net ionic equation, cross out anything that does not change from the left side of the equation to the right. • The only things left in the equation are those things that change (i.e. react) during the course of the reaction. • Those things that didn’t change (and were deleted from the ...
... • To form the net ionic equation, cross out anything that does not change from the left side of the equation to the right. • The only things left in the equation are those things that change (i.e. react) during the course of the reaction. • Those things that didn’t change (and were deleted from the ...
Phase behavior of clathrate hydrates: a model for single and
... cell contains 46 H2 O molecules, two 12-hedra (512 ), and six 14-hedra (512 62 ); where 512 is used to indicate that the polyhedron contains 12 9ve member ring faces (Sloan, 1998). Structure II (sII) is a body-centered cubic structure with a I (Mak & McMullen, 1965) for lattice parameter of 17:31 A ...
... cell contains 46 H2 O molecules, two 12-hedra (512 ), and six 14-hedra (512 62 ); where 512 is used to indicate that the polyhedron contains 12 9ve member ring faces (Sloan, 1998). Structure II (sII) is a body-centered cubic structure with a I (Mak & McMullen, 1965) for lattice parameter of 17:31 A ...
Detailed modeling of the evaporation and thermal decomposition of
... droplet increases uniformly which finally leads to the formation of a solid particle. But, when water vaporizes rapidly, urea concentration increases at the droplet surface which builds up a urea shell around the droplet. This may lead to the boiling of the water inside the urea shell and even to th ...
... droplet increases uniformly which finally leads to the formation of a solid particle. But, when water vaporizes rapidly, urea concentration increases at the droplet surface which builds up a urea shell around the droplet. This may lead to the boiling of the water inside the urea shell and even to th ...
CfE Higher Chemistry Unit 1: Chemical Changes and Structure
... concentration of reactants, particle size and temperature. How can collision theory explain these effects? A suitable reaction which will be used to study this over the next three activities is the reaction of hydrochloric acid with a sample of calcium carbonate (chalk). ...
... concentration of reactants, particle size and temperature. How can collision theory explain these effects? A suitable reaction which will be used to study this over the next three activities is the reaction of hydrochloric acid with a sample of calcium carbonate (chalk). ...
Problem 1-2
... Arrhenius stated that acids dissociate in hydronium cations and acid residue anions when dissolved in water and bases dissociate in metal cations and hydroxyl anions. Subsequently the acidic or basic reaction of aqueous solutions is based on a surplus of hydronium cations and hydroxyl anions, respec ...
... Arrhenius stated that acids dissociate in hydronium cations and acid residue anions when dissolved in water and bases dissociate in metal cations and hydroxyl anions. Subsequently the acidic or basic reaction of aqueous solutions is based on a surplus of hydronium cations and hydroxyl anions, respec ...
DOE Chemistry 1
... for use by DOE category A reactors. The subject areas, subject matter content, and level of detail of the Reactor Operator Fundamentals Manuals were determined from several sources. DOE Category A reactor training managers determined which materials should be included, and served as a primary refere ...
... for use by DOE category A reactors. The subject areas, subject matter content, and level of detail of the Reactor Operator Fundamentals Manuals were determined from several sources. DOE Category A reactor training managers determined which materials should be included, and served as a primary refere ...
document
... 0.0119 mole NaHCO3 reacts with 0.0119 mole HCl 0.0171 mole Mg(OH)2 reacts with 0.0342 mole HCl ...
... 0.0119 mole NaHCO3 reacts with 0.0119 mole HCl 0.0171 mole Mg(OH)2 reacts with 0.0342 mole HCl ...
Gas phase chemistry of neutral metal clusters
... for clusters with ionization energies larger than 10.5 eV is additionally possible, but should also not cause fragmentation of high ionization energy clusters. In general, for the majority of systems, the true neutral cluster distribution is obtained through 118 nm (or similar) single photon ionizat ...
... for clusters with ionization energies larger than 10.5 eV is additionally possible, but should also not cause fragmentation of high ionization energy clusters. In general, for the majority of systems, the true neutral cluster distribution is obtained through 118 nm (or similar) single photon ionizat ...
Electrolysis of water
Electrolysis of water is the decomposition of water (H2O) into oxygen (O2) and hydrogen gas (H2) due to an electric current being passed through the water.This technique can be used to make hydrogen fuel (hydrogen gas) and breathable oxygen; though currently most industrial methods make hydrogen fuel from natural gas instead.