Final Exam Review- no solutions
... 8. Assign oxidation states to each element in the following compounds. a. SO3 b. PO4 3c. Cr2O7 2d. HClO2 9. Use the changes in oxidation numbers to determine which elements are oxidized and which are reduced in these reactions. (Note: it is not necessary to use balanced equations) a. C + H2SO4 CO2 ...
... 8. Assign oxidation states to each element in the following compounds. a. SO3 b. PO4 3c. Cr2O7 2d. HClO2 9. Use the changes in oxidation numbers to determine which elements are oxidized and which are reduced in these reactions. (Note: it is not necessary to use balanced equations) a. C + H2SO4 CO2 ...
3.-Electrochemical-Cells-V2-
... are batteries which, when they go ‘flat’, can be charged and re-used. This means during recharging the chemicals used in the redox reactions are reformed. The lead-acid battery is the oldest type of rechargeable battery. The battery is made from plates and . A car battery is an example of this type ...
... are batteries which, when they go ‘flat’, can be charged and re-used. This means during recharging the chemicals used in the redox reactions are reformed. The lead-acid battery is the oldest type of rechargeable battery. The battery is made from plates and . A car battery is an example of this type ...
CHEMISTRY I Final..#1..rev 4KEY
... a. The atom is a hard sphereelectrons exist in orbitals outside the nucleusmost of the atom is empty space with a small dense nucleus. b. The atom is a hard spheremost of the atom is empty space with a small dense nucleuselectrons exist in orbitals outside the nucleus c. Most of the atom is empt ...
... a. The atom is a hard sphereelectrons exist in orbitals outside the nucleusmost of the atom is empty space with a small dense nucleus. b. The atom is a hard spheremost of the atom is empty space with a small dense nucleuselectrons exist in orbitals outside the nucleus c. Most of the atom is empt ...
Second review [Compatibility Mode]
... Moles of substance ⇔ heat release or adsorbed – Note that the heat is determined by the limiting reagent • Example How much ethane would need to be burned to heat 100 g of water by 10 K? 2C2H6(g) + 7O2(g) → 4CO2(g) + 6H2O(l) ...
... Moles of substance ⇔ heat release or adsorbed – Note that the heat is determined by the limiting reagent • Example How much ethane would need to be burned to heat 100 g of water by 10 K? 2C2H6(g) + 7O2(g) → 4CO2(g) + 6H2O(l) ...
C1 – Air and water information
... When a substance chemically combines with oxygen it is an example of oxidation. Combustion reactions are therefore oxidation. Some gases involved in combustion reactions can be identified by their chemical reactions. C1.2 Why are there temperature changes in chemical reactions? When a fuel is burned ...
... When a substance chemically combines with oxygen it is an example of oxidation. Combustion reactions are therefore oxidation. Some gases involved in combustion reactions can be identified by their chemical reactions. C1.2 Why are there temperature changes in chemical reactions? When a fuel is burned ...
Quiz 1
... 7. Which of the following is a correct statement concerning solution A with a pH of 11.5 compared to solution B with a pH of 10.0? Solution A… a. has a smaller [OH¯] than solution B b. has a larger number of [H+] than solution B c. is more basic than solution B d. is more acidic than solution B e. h ...
... 7. Which of the following is a correct statement concerning solution A with a pH of 11.5 compared to solution B with a pH of 10.0? Solution A… a. has a smaller [OH¯] than solution B b. has a larger number of [H+] than solution B c. is more basic than solution B d. is more acidic than solution B e. h ...
Cl Cl and
... Elements of group 6 react by sharing 2 electrons. Elements of group 7 react by sharing 1 electron 30. In H2O, why does hydrogen make one bond while oxygen makes two? O needs two electrons to become like Ne while H needs only one to become like He. 31. How do you explain that oxygen forms O2 molecule ...
... Elements of group 6 react by sharing 2 electrons. Elements of group 7 react by sharing 1 electron 30. In H2O, why does hydrogen make one bond while oxygen makes two? O needs two electrons to become like Ne while H needs only one to become like He. 31. How do you explain that oxygen forms O2 molecule ...
Sugárkémiai áttekintés Schiller Róbert
... β irradiation of oxidized iron surfaces, after that electrode Impedance in aquoeus SO32- solution (hole capture). Equivalent circuit: ...
... β irradiation of oxidized iron surfaces, after that electrode Impedance in aquoeus SO32- solution (hole capture). Equivalent circuit: ...
CHAPTER 9 HYDROGEN Position of Hydrogen in Periodic Table
... to form normal covalent bonds is called electron rich hydride. For example, hydrides of group 15 to 17 (NH3, PH3, H2O, H2S, H2Se, H2Te, HF etc.) (iii) Metallic or non-stoichiometric hydrides: These are formed by many d-block and f-block elements These hydrides conducts heat and electricity though ...
... to form normal covalent bonds is called electron rich hydride. For example, hydrides of group 15 to 17 (NH3, PH3, H2O, H2S, H2Se, H2Te, HF etc.) (iii) Metallic or non-stoichiometric hydrides: These are formed by many d-block and f-block elements These hydrides conducts heat and electricity though ...
Answers - U of L Class Index
... Why do deposits build up in pots and kettles which are regularly used to heat hard water? Heating water reduces the solubility of gases such as CO2. This makes the water less acidic and some of the soluble bicarbonates are converted back to carbonates and precipitate out. This is an example of Le Ch ...
... Why do deposits build up in pots and kettles which are regularly used to heat hard water? Heating water reduces the solubility of gases such as CO2. This makes the water less acidic and some of the soluble bicarbonates are converted back to carbonates and precipitate out. This is an example of Le Ch ...
Chemistry Final Exam Review 2006-2007
... Name 3 factors that increase the rate of dissolution of a substance. Describe solution equilibrium. What is the effect of temperature and pressure above a gas on gas solubility? What is the effect of temperature on the solubility for most ionic solids? What is the molarity of 4.5 moles of Ba(OH)2 in ...
... Name 3 factors that increase the rate of dissolution of a substance. Describe solution equilibrium. What is the effect of temperature and pressure above a gas on gas solubility? What is the effect of temperature on the solubility for most ionic solids? What is the molarity of 4.5 moles of Ba(OH)2 in ...
ch19 MSJ jlm
... a list of metals arranged in order of ease of oxidation.* The lower a metal is on the SRP table, the more active that metal is, i.e., the more easily it is oxidized. Any metal can be oxidized by the ions of elements above it. *Caution! The Activity series of Ch 5 is in reverse order of the SRP table ...
... a list of metals arranged in order of ease of oxidation.* The lower a metal is on the SRP table, the more active that metal is, i.e., the more easily it is oxidized. Any metal can be oxidized by the ions of elements above it. *Caution! The Activity series of Ch 5 is in reverse order of the SRP table ...
1 2016-17 Honors Chemistry Review for the Final Exam Each unit
... (b) How many moles of magnesium chloride are found in a sample containing 4.50 x 1022 formula units of magnesium chloride? ...
... (b) How many moles of magnesium chloride are found in a sample containing 4.50 x 1022 formula units of magnesium chloride? ...
AQA C2 revision book
... 2) The volumes of acid and alkali used are noted, and the experiment is repeated using the same volumes, but no indicator. 3) The solution is evaporated to leave the salt Method 2 Where an acid is reacted with an insoluble substance (base, metal or carbonate). 1) Some acid is measured into a beaker ...
... 2) The volumes of acid and alkali used are noted, and the experiment is repeated using the same volumes, but no indicator. 3) The solution is evaporated to leave the salt Method 2 Where an acid is reacted with an insoluble substance (base, metal or carbonate). 1) Some acid is measured into a beaker ...
Final Preparation
... C) the hydrophobic heads point to the hydrophilic tails D) the hydrophobic tails of the molecules point toward each other 77. What is the [H3O+] concentration of a solution that has a pH = 11.61? A) 1.2 × 101 M B) 1.0 × 10-14 M C) 2.5 × 10-12 M 78. Each of the following can cause a protein to denatu ...
... C) the hydrophobic heads point to the hydrophilic tails D) the hydrophobic tails of the molecules point toward each other 77. What is the [H3O+] concentration of a solution that has a pH = 11.61? A) 1.2 × 101 M B) 1.0 × 10-14 M C) 2.5 × 10-12 M 78. Each of the following can cause a protein to denatu ...
2016 GK Advamce 1
... When an iron nail gets rusted, iron oxide is formed with increase in the weight of the nail Galvanised iron sheets have a coating of zinc Nonstick cooking utensils are coated with Teflon Soda water contains carbon dioxide Nitrous oxide is more commonly known as laughing gas? The filament of an elect ...
... When an iron nail gets rusted, iron oxide is formed with increase in the weight of the nail Galvanised iron sheets have a coating of zinc Nonstick cooking utensils are coated with Teflon Soda water contains carbon dioxide Nitrous oxide is more commonly known as laughing gas? The filament of an elect ...
Unit 2: Atoms and Ions Homework Booklet
... c. When coal is burned a gas is given off that is weakly soluble in water. What is the gas and what is formed when it dissolves in water? ...
... c. When coal is burned a gas is given off that is weakly soluble in water. What is the gas and what is formed when it dissolves in water? ...
Elements, Compounds, and Molecules
... What is the difference between a compound and a molecule? A molecule is formed when two or more atoms join together chemically. A compound is a molecule that contains at least two different elements. All compounds are molecules but not all molecules are compounds. ...
... What is the difference between a compound and a molecule? A molecule is formed when two or more atoms join together chemically. A compound is a molecule that contains at least two different elements. All compounds are molecules but not all molecules are compounds. ...
Lab Stoichiometry problems Dr. Baxley 1. Lithium metal reacts with
... a. Write a balanced equation for the reaction b. Draw a model of the balanced chemical equation, using labeled circles for each atom. c. Solve for how many units of lithium nitride can be made if 12 lithium atoms and 4 nitrogen molecules are allowed to react. Determine if there is any remaining reac ...
... a. Write a balanced equation for the reaction b. Draw a model of the balanced chemical equation, using labeled circles for each atom. c. Solve for how many units of lithium nitride can be made if 12 lithium atoms and 4 nitrogen molecules are allowed to react. Determine if there is any remaining reac ...
1. What are micelles? Give two examples of micellar systems. Sol. A
... Anode reaction: 2Fe => 2Fe2+ + 4eCathode reaction: O2 + 2H2O + 4e- => 4OHThere are obviously different anodic and cathodic reactions for different alloys exposed to various environments. These half cell reactions are thought to occur (at least initially) at microscopic anodes and cathodes covering a ...
... Anode reaction: 2Fe => 2Fe2+ + 4eCathode reaction: O2 + 2H2O + 4e- => 4OHThere are obviously different anodic and cathodic reactions for different alloys exposed to various environments. These half cell reactions are thought to occur (at least initially) at microscopic anodes and cathodes covering a ...
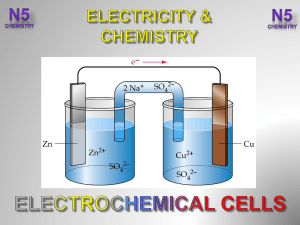
Electrolysis of water
Electrolysis of water is the decomposition of water (H2O) into oxygen (O2) and hydrogen gas (H2) due to an electric current being passed through the water.This technique can be used to make hydrogen fuel (hydrogen gas) and breathable oxygen; though currently most industrial methods make hydrogen fuel from natural gas instead.



![Second review [Compatibility Mode]](http://s1.studyres.com/store/data/003692853_1-a578e4717b0c8365c11d7e7f576654ae-300x300.png)