Form A 1 Chem 130 Name______________________________
... If we look at the reaction, there are three moles of gas on the reactant side and only two moles of gas on the product side, therefore, we expect the S for this process to be negative. So, we will have a situation where we have a negative H and a negative S. Since G = H - TS, the spontaneity o ...
... If we look at the reaction, there are three moles of gas on the reactant side and only two moles of gas on the product side, therefore, we expect the S for this process to be negative. So, we will have a situation where we have a negative H and a negative S. Since G = H - TS, the spontaneity o ...
____ 1. The energy required to convert a ground
... 46. Equal numbers of moles of He(g), Ar(g), and Ne(g) are placed in a glass vessel at room temperature. If the vessel has a pinhole-sized leak, which of the following will be true regarding the relative values of the partial pressures of the gases remaining in the vessel after some of the gas mixtur ...
... 46. Equal numbers of moles of He(g), Ar(g), and Ne(g) are placed in a glass vessel at room temperature. If the vessel has a pinhole-sized leak, which of the following will be true regarding the relative values of the partial pressures of the gases remaining in the vessel after some of the gas mixtur ...
CHAPTER 1 Differentiate b/w Mendeleev`s periodic law and modern
... The electronegativities of group IV-A decrease down the group along with their increasing sizes. These two parameters are responsible for creating van der Waal's forces of attraction among the hydrides. In this way, their melting and boiling points increase. Water is liquid at room temperature while ...
... The electronegativities of group IV-A decrease down the group along with their increasing sizes. These two parameters are responsible for creating van der Waal's forces of attraction among the hydrides. In this way, their melting and boiling points increase. Water is liquid at room temperature while ...
Balancing and Predicting Chemical Reactions:
... What is the net ionic equation for the reaction between aqueous calcium hydroxide and nitric acid? The products of this reaction are aqueous calcium nitrate and water. How does this net ionic equation compare to the net ionic equation shown on the earlier slide? ...
... What is the net ionic equation for the reaction between aqueous calcium hydroxide and nitric acid? The products of this reaction are aqueous calcium nitrate and water. How does this net ionic equation compare to the net ionic equation shown on the earlier slide? ...
Chem 173: Final Exam Review Short Answer and Problems 1
... Limestone is composed of calcium carbonate (CaCO3) as well as other compounds. In an analysis, a chemist takes a sample of limestone which has a mass of 413 mg and treats it with oxalic acid (H2C 2O4). A chemical reaction occurs between the calcium carbonate and the acid producing calcium oxalate an ...
... Limestone is composed of calcium carbonate (CaCO3) as well as other compounds. In an analysis, a chemist takes a sample of limestone which has a mass of 413 mg and treats it with oxalic acid (H2C 2O4). A chemical reaction occurs between the calcium carbonate and the acid producing calcium oxalate an ...
New method for determination of surface pK, using second harmonic
... the positively charged monolayer C2*NH: the B@(O) term favors an alignment of the bulk water molecules with the oxygen directed towards the positively charged interface. Thus the ,$*) contribution from the interface water molecules and the xc3’contribution which is electric field dependent and const ...
... the positively charged monolayer C2*NH: the B@(O) term favors an alignment of the bulk water molecules with the oxygen directed towards the positively charged interface. Thus the ,$*) contribution from the interface water molecules and the xc3’contribution which is electric field dependent and const ...
Final Exam Practice Problems: R = 0.0821 Latm/molK NA = 6.022
... 2. A substance that can't be chemically broken down into simpler substances is considered to be A) a homogeneous mixture. B) an element. C) a heterogeneous mixture. D) a compound. E) an electron. 3. Which of the following are examples of a chemical change? A) coffee brewing B) water boiling C) leave ...
... 2. A substance that can't be chemically broken down into simpler substances is considered to be A) a homogeneous mixture. B) an element. C) a heterogeneous mixture. D) a compound. E) an electron. 3. Which of the following are examples of a chemical change? A) coffee brewing B) water boiling C) leave ...
Recording Measurements
... 40. According to Reference Table J, which of these metals will react most readily with 1.0 M HCl to produce H2(g)? (1) Ca (2) K (3) Mg (4) Zn 41. Under standard conditions, which metal will react with 0.1 M HCl to liberate hydrogen gas? (1) Ag (2) Au (3) Cu (4) Mg 42. Because tap water is slightly a ...
... 40. According to Reference Table J, which of these metals will react most readily with 1.0 M HCl to produce H2(g)? (1) Ca (2) K (3) Mg (4) Zn 41. Under standard conditions, which metal will react with 0.1 M HCl to liberate hydrogen gas? (1) Ag (2) Au (3) Cu (4) Mg 42. Because tap water is slightly a ...
Redox
... whether electrons are lost or gained, but it is less easy to tell when complex ions or covalent molecules are involved. Oxidation number is a useful concept for helping to decide in these more awkward cases. ...
... whether electrons are lost or gained, but it is less easy to tell when complex ions or covalent molecules are involved. Oxidation number is a useful concept for helping to decide in these more awkward cases. ...
Practice Qs - Unit 10 Acid Base
... 40. According to Reference Table J, which of these metals will react most readily with 1.0 M HCl to produce H2(g)? (1) Ca (2) K (3) Mg (4) Zn 41. Under standard conditions, which metal will react with 0.1 M HCl to liberate hydrogen gas? (1) Ag (2) Au (3) Cu (4) Mg 42. Because tap water is slightly a ...
... 40. According to Reference Table J, which of these metals will react most readily with 1.0 M HCl to produce H2(g)? (1) Ca (2) K (3) Mg (4) Zn 41. Under standard conditions, which metal will react with 0.1 M HCl to liberate hydrogen gas? (1) Ag (2) Au (3) Cu (4) Mg 42. Because tap water is slightly a ...
Section 1e chemical formulae and chemical equations
... experiment with gypsum. Weigh a crucible empty. Weigh accurately about 30 g of gypsum into the crucible. Heat the crucible strongly for 10 minutes. Then allow it to cool, and reweigh. Heat the crucible again; cool and reweigh. The results she obtained are listed below. Mass (in g) Mass of crucible ...
... experiment with gypsum. Weigh a crucible empty. Weigh accurately about 30 g of gypsum into the crucible. Heat the crucible strongly for 10 minutes. Then allow it to cool, and reweigh. Heat the crucible again; cool and reweigh. The results she obtained are listed below. Mass (in g) Mass of crucible ...
Chapter8 - Louisiana Tech University
... of joules) or in kcal (kilocalories). ΔH˚ is the enthalpy measure at standard thermodynamic temprearture and pressure: 25˚C (298 K) and 1 atm (760 torr). If ΔH is negative for the reaction, then energy has been released. This type of reaction is referred to as exothermic (literally, outside heat, or ...
... of joules) or in kcal (kilocalories). ΔH˚ is the enthalpy measure at standard thermodynamic temprearture and pressure: 25˚C (298 K) and 1 atm (760 torr). If ΔH is negative for the reaction, then energy has been released. This type of reaction is referred to as exothermic (literally, outside heat, or ...
Chapter 7: Thermochemistry
... the bomb and its contents, the water in which the bomb is immersed, the thermometer, the stirrer, and so on. The system is isolated from its surroundings. ...
... the bomb and its contents, the water in which the bomb is immersed, the thermometer, the stirrer, and so on. The system is isolated from its surroundings. ...
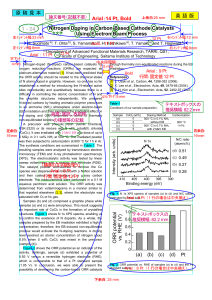
Effects of antioxidants for the degradation of flame
... 6 MGy in 0.1 vol% NH3 at 500 °C. The irradiated powder was then subjected to carbonization at 800 °C for 1 h in Ar. The synthesis conditions are summarized in Table 1. The resulting samples were analyzed by transmission electron microscopy (TEM) and X-ray photoelectron spectroscopy (XPS). The electr ...
... 6 MGy in 0.1 vol% NH3 at 500 °C. The irradiated powder was then subjected to carbonization at 800 °C for 1 h in Ar. The synthesis conditions are summarized in Table 1. The resulting samples were analyzed by transmission electron microscopy (TEM) and X-ray photoelectron spectroscopy (XPS). The electr ...
Energy
... obtained by multiplying the value of ΔH in the original equation by that same factor. When a chemical equation is reversed, the sign of H is reversed. ...
... obtained by multiplying the value of ΔH in the original equation by that same factor. When a chemical equation is reversed, the sign of H is reversed. ...
CHEMSTRY FREE-RESPONSE QUESTIONS (Form B)
... Answer question 4 below. The section II score weighting for this question is 10 percent. 4. For each of the following three reactions in part (i) write a balanced equation for the reaction and in part (ii) answer the question about the reaction, In part (i), coefficients should be in terms of lowest ...
... Answer question 4 below. The section II score weighting for this question is 10 percent. 4. For each of the following three reactions in part (i) write a balanced equation for the reaction and in part (ii) answer the question about the reaction, In part (i), coefficients should be in terms of lowest ...
Coherence Domain - Conference on the Physics, Chemistry and
... • Water CDs can thus get excited by collecting ambient energy and get de-excited by activating “guest molecules” producing so a CD oscillation. • This oscillatory regime allows the onset of a coherence among coherence domains that tune together the oscillations of single CDs. • In this way coherence ...
... • Water CDs can thus get excited by collecting ambient energy and get de-excited by activating “guest molecules” producing so a CD oscillation. • This oscillatory regime allows the onset of a coherence among coherence domains that tune together the oscillations of single CDs. • In this way coherence ...
C:\SUBJECTS\SUBJECTS\Chemistry
... change their shape C. are oxidized by atmospheric oxygen D. contract E. become mobile as the inter molecular forces are broken. The ration of the number of molecules in 2g of hydrogen to that in 16 g of oxygen is A. ...
... change their shape C. are oxidized by atmospheric oxygen D. contract E. become mobile as the inter molecular forces are broken. The ration of the number of molecules in 2g of hydrogen to that in 16 g of oxygen is A. ...
3. What is the empirical formula of a compound that is
... As you learned in health and biology, food energy typically comes from carbohydrates, proteins and fats. The amount of energy that the body can use per gram of these substances is not the same. The following balanced exothermic reaction represents combustion (respiration) of glucose (a carbohydrate) ...
... As you learned in health and biology, food energy typically comes from carbohydrates, proteins and fats. The amount of energy that the body can use per gram of these substances is not the same. The following balanced exothermic reaction represents combustion (respiration) of glucose (a carbohydrate) ...
Balanced Chemical Equation
... • Predict the products, if any, note oxidation #’s, and balance the reaction: 1. __Fe(s) + __CuSO4(aq) __FeSO4(aq) + __Cu(s) 2. __Br2(l) + __MgCl2(aq) No Reaction 3. __Fe(s) + __Na3PO4(aq) No Reaction 4. __Al(s) + _3CuCl(aq) __AlCl3(aq) + 3_Cu(s) ...
... • Predict the products, if any, note oxidation #’s, and balance the reaction: 1. __Fe(s) + __CuSO4(aq) __FeSO4(aq) + __Cu(s) 2. __Br2(l) + __MgCl2(aq) No Reaction 3. __Fe(s) + __Na3PO4(aq) No Reaction 4. __Al(s) + _3CuCl(aq) __AlCl3(aq) + 3_Cu(s) ...
Rates of Reaction: Chemical Kinetics 50
... adjacent office to telephone her agent in New York City. The agent has her secretary write a note to Dr. Goodall. The note is typed and faxed to Africa. The fax is placed in an envelope when it is received and given to a messenger who must travel a few kilometres by boat and a few hundred metres on ...
... adjacent office to telephone her agent in New York City. The agent has her secretary write a note to Dr. Goodall. The note is typed and faxed to Africa. The fax is placed in an envelope when it is received and given to a messenger who must travel a few kilometres by boat and a few hundred metres on ...
Electrolysis of water
Electrolysis of water is the decomposition of water (H2O) into oxygen (O2) and hydrogen gas (H2) due to an electric current being passed through the water.This technique can be used to make hydrogen fuel (hydrogen gas) and breathable oxygen; though currently most industrial methods make hydrogen fuel from natural gas instead.