MODA Tequila
... Our Process... Moda Tequila is charred barrel aged in high-grade oak wood drums where it absorbs the rich amber/black from the cured barrel lining naturally. But more than just coloring the alcohol, the charred aging imbues a warm, rustic savor into the agave notes, with a medium intensity that evok ...
... Our Process... Moda Tequila is charred barrel aged in high-grade oak wood drums where it absorbs the rich amber/black from the cured barrel lining naturally. But more than just coloring the alcohol, the charred aging imbues a warm, rustic savor into the agave notes, with a medium intensity that evok ...
File - Chem with Appleby
... The Direction of the Chemical Equation and K-12 The equilibrium constant of a reaction in the reverse reaction is the ______________of the equilibrium constant of the _____________________ reaction: ...
... The Direction of the Chemical Equation and K-12 The equilibrium constant of a reaction in the reverse reaction is the ______________of the equilibrium constant of the _____________________ reaction: ...
Chapter 17 - Cengage Learning
... INTRODUCTION Because atoms and molecules are so tiny, it is hard to imagine what happens when they react and form new products. In this chapter you will learn what is necessary for a reaction to occur, why some reactions stop before all the reactants have been used up, and how to speed up a reaction ...
... INTRODUCTION Because atoms and molecules are so tiny, it is hard to imagine what happens when they react and form new products. In this chapter you will learn what is necessary for a reaction to occur, why some reactions stop before all the reactants have been used up, and how to speed up a reaction ...
03-Chemical Rxns n Stoichiometry
... We know the reactants’ identities, but may need to establish identities of products via further research, etc. When identities of reactants and products are established, we can write their formulas. Still, the number of atoms of each element must be the same on both sides of the equation to adhere t ...
... We know the reactants’ identities, but may need to establish identities of products via further research, etc. When identities of reactants and products are established, we can write their formulas. Still, the number of atoms of each element must be the same on both sides of the equation to adhere t ...
Reactions Balancing Chemical Equations uses Law of conservation
... Solution reactions Gas phase reactions ...
... Solution reactions Gas phase reactions ...
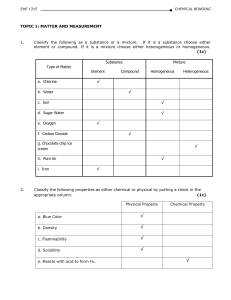
DEPARTMENT OF CHEMISTRY, CFS, IIUM
... variety of matter is recognized is called a property. A characteristic that depends upon the amount of matter in the sample is called an extensive property. A characteristic that does not depend upon the amount of matter is called an intensive property. A characteristic that can be observed without ...
... variety of matter is recognized is called a property. A characteristic that depends upon the amount of matter in the sample is called an extensive property. A characteristic that does not depend upon the amount of matter is called an intensive property. A characteristic that can be observed without ...
Chapter 9 – Reaction Energetics
... result. If we knew the energies of all of the interactions involved, we could determine the enthalpy of reaction by determining how much energy must be supplied to break all of the interactions that had to be broken and subtracting the energy that is released when all of the new interactions form. B ...
... result. If we knew the energies of all of the interactions involved, we could determine the enthalpy of reaction by determining how much energy must be supplied to break all of the interactions that had to be broken and subtracting the energy that is released when all of the new interactions form. B ...
Inorganometallic Chemistry
... be used to form molecular orbitals. On the other hand, if such a metalloid (p-block elements, i.e. main groups 13, 14, except C, 15, except N and Se and Te from 16) replaces the carbon atom in the metal-carbon bond, then they really form a metalnonmetal bonding and, for the main part, p-block – d-bl ...
... be used to form molecular orbitals. On the other hand, if such a metalloid (p-block elements, i.e. main groups 13, 14, except C, 15, except N and Se and Te from 16) replaces the carbon atom in the metal-carbon bond, then they really form a metalnonmetal bonding and, for the main part, p-block – d-bl ...
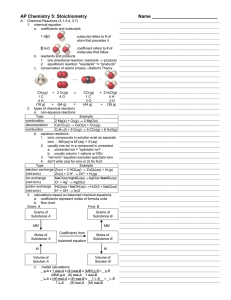
AP Chemistry
... in which ions exchange "partners"; and (6) proton exchange reactions, in which atoms exchange H+. The last three reactions will be discussed in more detail later in the year. Aqueous reactions (electron exchange, ion exchange and proton exchange) usually involve ions, where only one of the two ions ...
... in which ions exchange "partners"; and (6) proton exchange reactions, in which atoms exchange H+. The last three reactions will be discussed in more detail later in the year. Aqueous reactions (electron exchange, ion exchange and proton exchange) usually involve ions, where only one of the two ions ...
Fundamentals Diagnostic Quiz
... b) Most of the space of an atom is empty space. *c) The nuclear model in which the positive charge is held densely in the center of the atom is no longer ...
... b) Most of the space of an atom is empty space. *c) The nuclear model in which the positive charge is held densely in the center of the atom is no longer ...
vce chemistry trial exam 1
... C is incorrect because 8.60 g is the mass of the oxide. The mass of oxygen is the mass of the oxide minus the mass of lanthanum; i.e. m(O) = m(oxide) – m(La). D is incorrect because the molar mass of oxygen is 16.0 g mol–1, not 32.0 g mol–1. ...
... C is incorrect because 8.60 g is the mass of the oxide. The mass of oxygen is the mass of the oxide minus the mass of lanthanum; i.e. m(O) = m(oxide) – m(La). D is incorrect because the molar mass of oxygen is 16.0 g mol–1, not 32.0 g mol–1. ...
Membrane bioreactor system for treating synthetic oilfield
... inorganic contaminants from refinery wastewater streams employing ultrafiltration and reverse osmosis was provided. To remove divalent and trivalent metal cations prior to being passed to the reverse osmosis a sequential chemical softening system employed to ...
... inorganic contaminants from refinery wastewater streams employing ultrafiltration and reverse osmosis was provided. To remove divalent and trivalent metal cations prior to being passed to the reverse osmosis a sequential chemical softening system employed to ...
Multiple-choice questions : 1. The following graph shows the volume
... Each question below consists of two separate statements. Decide whether each of the two statements is true or false; if both are true, then decide whether or not the second statement is a correct explanation of the first statement. Then select one option from A to D according to the following table: ...
... Each question below consists of two separate statements. Decide whether each of the two statements is true or false; if both are true, then decide whether or not the second statement is a correct explanation of the first statement. Then select one option from A to D according to the following table: ...
aq - HCC Learning Web
... • Formulas and symbols are used to describe a chemical reaction as a chemical equation A+BC+D • A and B are reactants and C and D are products • Physical state is abbreviated by either (s), (l), (g), or (aq) • A catalyst is a substance that speeds up a reaction without being consumed or permanently ...
... • Formulas and symbols are used to describe a chemical reaction as a chemical equation A+BC+D • A and B are reactants and C and D are products • Physical state is abbreviated by either (s), (l), (g), or (aq) • A catalyst is a substance that speeds up a reaction without being consumed or permanently ...
Problem 5. The Second Law of thermodynamics
... 0.565 . All the efficiencies from 0 to 0.565 are possible, if we go in more steps. ...
... 0.565 . All the efficiencies from 0 to 0.565 are possible, if we go in more steps. ...