CH 301 Practice Test Questions
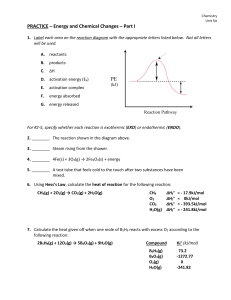
... 15. Estimate ΔH for the gas-phase reaction NCl3 + 3H2O NH3 + 3 HOCl, based on the bond energies N-Cl : 190 kJ/mol; O-H : 464 kJ/mol; N-H : 391 kJ/mol; O-Cl : 206 kJ/mol. 16. Calculate the enthalpy change for the reaction SO2(g) + ½ O2(g) SO3(g) given the following: SO2(g) S(s) + O2(g) 2SO3(g) ...
... 15. Estimate ΔH for the gas-phase reaction NCl3 + 3H2O NH3 + 3 HOCl, based on the bond energies N-Cl : 190 kJ/mol; O-H : 464 kJ/mol; N-H : 391 kJ/mol; O-Cl : 206 kJ/mol. 16. Calculate the enthalpy change for the reaction SO2(g) + ½ O2(g) SO3(g) given the following: SO2(g) S(s) + O2(g) 2SO3(g) ...
What are reactions? - UTLNET Secure Site
... If they are formed in a reaction you will see __________. This can be a sign that a chemical __________ has happened. 2. Other signs of a chemical reaction might be an increase in temperature if _____ is released or a change in ________. 3. Physical changes like _________ do not make new materials a ...
... If they are formed in a reaction you will see __________. This can be a sign that a chemical __________ has happened. 2. Other signs of a chemical reaction might be an increase in temperature if _____ is released or a change in ________. 3. Physical changes like _________ do not make new materials a ...
What are reactions?
... If they are formed in a reaction you will see __________. This can be a sign that a chemical __________ has happened. 2. Other signs of a chemical reaction might be an increase in temperature if _____ is released or a change in ________. 3. Physical changes like _________ do not make new materials a ...
... If they are formed in a reaction you will see __________. This can be a sign that a chemical __________ has happened. 2. Other signs of a chemical reaction might be an increase in temperature if _____ is released or a change in ________. 3. Physical changes like _________ do not make new materials a ...
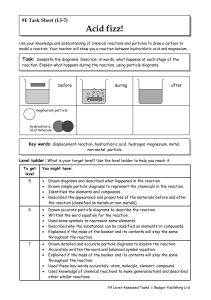
Year 9 Homework Task 9E-5 Reactions 5-7
... Drawn accurate particle diagrams to describe the reaction. Written the word equation for the reaction. Used some symbols to represent some elements. Described why the substances can be classified as elements or compounds. Explained if the mass of the beaker and its contents will stay the same throug ...
... Drawn accurate particle diagrams to describe the reaction. Written the word equation for the reaction. Used some symbols to represent some elements. Described why the substances can be classified as elements or compounds. Explained if the mass of the beaker and its contents will stay the same throug ...
Chemical Reactions
... • Uses chemical formulas and symbols to describe a chemical reaction and the product it produces (see below) Symbol ...
... • Uses chemical formulas and symbols to describe a chemical reaction and the product it produces (see below) Symbol ...
Ch 5.1 The Nature of Chemical Reactions
... reactants are rearranged to form one or more different substances • Old bonds are broken; new bonds form ...
... reactants are rearranged to form one or more different substances • Old bonds are broken; new bonds form ...
A.P. Chemistry Complexation Reactions
... Two compounds switch partners and form two new compounds. ...
... Two compounds switch partners and form two new compounds. ...
Collision Theory
... Reaction Kinetics: study of the factors that affect the rates of chemical reactions ...
... Reaction Kinetics: study of the factors that affect the rates of chemical reactions ...
1 ChE 505 WORKSHOP 1 1. Why are chemical reactions important
... Relate molar extent of reaction to the fractional conversion of the limiting reactant. What is the limiting reactant? ...
... Relate molar extent of reaction to the fractional conversion of the limiting reactant. What is the limiting reactant? ...
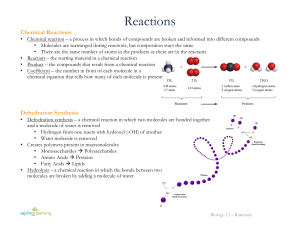
Reactions
... • Chemical reaction – a process in which bonds of compounds are broken and reformed into different compounds • Molecules are rearranged during reactions, but composition stays the same • There are the same number of atoms in the products as there are in the reactants • Reactant – the starting materi ...
... • Chemical reaction – a process in which bonds of compounds are broken and reformed into different compounds • Molecules are rearranged during reactions, but composition stays the same • There are the same number of atoms in the products as there are in the reactants • Reactant – the starting materi ...
Notes on Chapter 12 Chemical Equilibrium
... reaction (i.e. the energy required to break and form new bonds). ...
... reaction (i.e. the energy required to break and form new bonds). ...
Test 4
... A process that will occur without input of energy from a external source. Second law of thermodynamics In any spontaneous process the entropy of the universe always increases. Positional disorder Randomness that comes from the number of different arrangements of a system. Molar entropy of fusion The ...
... A process that will occur without input of energy from a external source. Second law of thermodynamics In any spontaneous process the entropy of the universe always increases. Positional disorder Randomness that comes from the number of different arrangements of a system. Molar entropy of fusion The ...
Bioorthogonal chemistry
The term bioorthogonal chemistry refers to any chemical reaction that can occur inside of living systems without interfering with native biochemical processes. The term was coined by Carolyn R. Bertozzi in 2003. Since its introduction, the concept of the bioorthogonal reaction has enabled the study of biomolecules such as glycans, proteins, and lipids in real time in living systems without cellular toxicity. A number of chemical ligation strategies have been developed that fulfill the requirements of bioorthogonality, including the 1,3-dipolar cycloaddition between azides and cyclooctynes (also termed copper-free click chemistry), between nitrones and cyclooctynes, oxime/hydrazone formation from aldehydes and ketones, the tetrazine ligation, the isocyanide-based click reaction, and most recently, the quadricyclane ligation.The use of bioorthogonal chemistry typically proceeds in two steps. First, a cellular substrate is modified with a bioorthogonal functional group (chemical reporter) and introduced to the cell; substrates include metabolites, enzyme inhibitors, etc. The chemical reporter must not alter the structure of the substrate dramatically to avoid affecting its bioactivity. Secondly, a probe containing the complementary functional group is introduced to react and label the substrate.Although effective bioorthogonal reactions such as copper-free click chemistry have been developed, development of new reactions continues to generate orthogonal methods for labeling to allow multiple methods of labeling to be used in the same biosystems.