B. The Physical Properties of Matter
... suitable solvent. The solvent is allowed to slowly evaporate causing some of the desired solid to come out of solution as crystals; however, not all the solvent is allowed to evaporate. The crystals that are formed can then be separated by HAND SEPARATION or FILTRATION. ...
... suitable solvent. The solvent is allowed to slowly evaporate causing some of the desired solid to come out of solution as crystals; however, not all the solvent is allowed to evaporate. The crystals that are formed can then be separated by HAND SEPARATION or FILTRATION. ...
You Can Eat Crystal and Glass??
... It has to do with how they are made. The rock candy on the right slowly formed crystals from sugar dissolved in water. The sugar molecules are in an organized pattern. The lollipop on the left was made from sugar solution heated to about 300 °F. It cooled so quickly from the liquid that there was no ...
... It has to do with how they are made. The rock candy on the right slowly formed crystals from sugar dissolved in water. The sugar molecules are in an organized pattern. The lollipop on the left was made from sugar solution heated to about 300 °F. It cooled so quickly from the liquid that there was no ...
The Physical Properties And Physical Changes of Substances
... suitable solvent. The solvent is allowed to slowly evaporate causing some of the desired solid to come out of solution as crystals; however, not all the solvent is allowed to evaporate. The crystals that are formed can then be separated by HAND SEPARATION or FILTRATION. ...
... suitable solvent. The solvent is allowed to slowly evaporate causing some of the desired solid to come out of solution as crystals; however, not all the solvent is allowed to evaporate. The crystals that are formed can then be separated by HAND SEPARATION or FILTRATION. ...
freshman engineering laboratory
... We will begin by discussing what liquid crystals are, how they polarize light, and how polarized light is used to make LCDs. We will then disassemble an LCD wristwatch and examine its components. We will examine the LCD in detail and then finally reassemble the LCD wristwatch. Liquid Crystals Liquid ...
... We will begin by discussing what liquid crystals are, how they polarize light, and how polarized light is used to make LCDs. We will then disassemble an LCD wristwatch and examine its components. We will examine the LCD in detail and then finally reassemble the LCD wristwatch. Liquid Crystals Liquid ...
lecture 5 phase equilibria
... • a sample is placed in a cavity between two gem-quality diamonds and then pressure is exerted by turning a screw. Pressures up to ~2 Mbar can be achieved. • one application is the study the transition of covalent solids to metallic solids. ...
... • a sample is placed in a cavity between two gem-quality diamonds and then pressure is exerted by turning a screw. Pressures up to ~2 Mbar can be achieved. • one application is the study the transition of covalent solids to metallic solids. ...
Pdf
... solve rigorously for the thermodynamic properties of our lattice model, which is equivalent to a spin-4 Ising model subject to spatially uniform external fields. However, we propose that the widely used “mean-field approximation” is applicable (11), and should suffice to replicate the desired phase ...
... solve rigorously for the thermodynamic properties of our lattice model, which is equivalent to a spin-4 Ising model subject to spatially uniform external fields. However, we propose that the widely used “mean-field approximation” is applicable (11), and should suffice to replicate the desired phase ...
Polarization-controlled contrasted images using dye
... system in which a delta-like filter is placed at the center of its Fourier plane. Ideally, the filter induces a phase delay between the spectral component corresponding to the zero spatial frequency and the rest of the spectrum. The consequence of this filtering process is the superposition of a pla ...
... system in which a delta-like filter is placed at the center of its Fourier plane. Ideally, the filter induces a phase delay between the spectral component corresponding to the zero spatial frequency and the rest of the spectrum. The consequence of this filtering process is the superposition of a pla ...
Intermolecular forces liquids and Solids
... • Intermolecular forces: attractions and repulsions between molecules that hold them together. • Intermolecular forces (van der Waals forces) hold molecules together in liquid and solid phases. – Ion-dipole force: interaction between an ion and partial charges in a polar molecule. – Dipole-dipole fo ...
... • Intermolecular forces: attractions and repulsions between molecules that hold them together. • Intermolecular forces (van der Waals forces) hold molecules together in liquid and solid phases. – Ion-dipole force: interaction between an ion and partial charges in a polar molecule. – Dipole-dipole fo ...
PowerPoint Chapter 14 - Preparatory Chemistry
... more polar? The greater the ΔEN is, the more polar the bond. ...
... more polar? The greater the ΔEN is, the more polar the bond. ...
Phase Transformations Some Definitions Some Definitions, 2
... Phase diagrams are extremely useful for systems with multiple components, and serve to describe physical and chemical equilibria over a range of different compositions, as well as points where substances are mutually miscible, or even when a system has to be brought to a specific set of conditions f ...
... Phase diagrams are extremely useful for systems with multiple components, and serve to describe physical and chemical equilibria over a range of different compositions, as well as points where substances are mutually miscible, or even when a system has to be brought to a specific set of conditions f ...
Physical concept of the surface tension of the liquid until some time
... Abstract: It is proved that the physical concept of liquid surface tension is a part of the concept of internal energy. The physical model of liquid evaporation and formula obtained for calculating surface tension of the liquid are suggested. The formula was tested on reference values for 64 substan ...
... Abstract: It is proved that the physical concept of liquid surface tension is a part of the concept of internal energy. The physical model of liquid evaporation and formula obtained for calculating surface tension of the liquid are suggested. The formula was tested on reference values for 64 substan ...
Expt. 5: Binary Phase Diagram CHEM 366 V-1 Binary Solid
... may behave more or less independent of each other but merely diluted, i.e., an ideal solution or mixture, or there may be substantial chemical interaction or complex formation between the constituents. The study of such mixtures can lead to an understanding of the most fundamental intermolecular int ...
... may behave more or less independent of each other but merely diluted, i.e., an ideal solution or mixture, or there may be substantial chemical interaction or complex formation between the constituents. The study of such mixtures can lead to an understanding of the most fundamental intermolecular int ...
High pressure differential scanning calorimetry of the hydrothermal
... As in naturally occurring salt solutions typically more than one salt is dissolved, higher order mixtures are also of interest. In the phase diagrams of such solutions the properties of one binary mixture transform into the properties of the second binary mixture, and this leads to either continuous ...
... As in naturally occurring salt solutions typically more than one salt is dissolved, higher order mixtures are also of interest. In the phase diagrams of such solutions the properties of one binary mixture transform into the properties of the second binary mixture, and this leads to either continuous ...
Phase-separation in ion-containing mixtures in electric fields
... From Eq. 6 we see that a liquid mixture phase- of the A and B liquids by ηA and ηB , respectively. separates into its components when put under the In the absence of field (mixed state) and under coninfluence of an electric field in some reasonable condi- stant applied external stress, the mixture w ...
... From Eq. 6 we see that a liquid mixture phase- of the A and B liquids by ηA and ηB , respectively. separates into its components when put under the In the absence of field (mixed state) and under coninfluence of an electric field in some reasonable condi- stant applied external stress, the mixture w ...
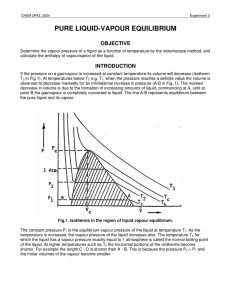
pure liquid-vapour equilibrium - Theoretical and Computational
... in Fig.1 includes all states of the system which correspond to the existence of two phases, liquid and vapour. Part of the shaded area, up to the normal boiling point, Tb, of the liquid, is the subject of this experiment in which the vapour pressure dependence on temperature will be investigated. Th ...
... in Fig.1 includes all states of the system which correspond to the existence of two phases, liquid and vapour. Part of the shaded area, up to the normal boiling point, Tb, of the liquid, is the subject of this experiment in which the vapour pressure dependence on temperature will be investigated. Th ...
Liquid Fundamental Measurements on an Aggregated Dye Crystal
... Lyotropic chromonic liquid crystals form a liquid crystal phase in the aggregate, which distinguishes them from thermotropic liquid crystals. In most of the liquid crystals that have been studied, including the lyotropic liquid crystal described above, the particles that form the liquid crystal phas ...
... Lyotropic chromonic liquid crystals form a liquid crystal phase in the aggregate, which distinguishes them from thermotropic liquid crystals. In most of the liquid crystals that have been studied, including the lyotropic liquid crystal described above, the particles that form the liquid crystal phas ...
Chapter 27
... Wall coated (WCOT) <1 m thick liquid coating on inside of silica tubing support-coated (SCOT) 30 m thick coating of liquid-coated support on inside of silica ...
... Wall coated (WCOT) <1 m thick liquid coating on inside of silica tubing support-coated (SCOT) 30 m thick coating of liquid-coated support on inside of silica ...
Crystallization of hydroxide cobalt carbonate Co2CO3(OH)2
... hydroxide carbonate Co2CO3(OH)2 and its hydrated phases Co2CO3(OH)2•nH2O have been subject of interest in the scientific literature because they can be precursors of the socalled low-dimensional cobalt oxide-based nanomaterials. Various cobalt salts can be precursors of Co3O4, but carbonates and hyd ...
... hydroxide carbonate Co2CO3(OH)2 and its hydrated phases Co2CO3(OH)2•nH2O have been subject of interest in the scientific literature because they can be precursors of the socalled low-dimensional cobalt oxide-based nanomaterials. Various cobalt salts can be precursors of Co3O4, but carbonates and hyd ...
Experimental Competition
... attainable speed while watching the screen. As increases you might observe different frequency ranges over which the properties of the “image” are drastically different. To describe these observations complete the table on the answer form by adding a row to this table for each such frequency range ...
... attainable speed while watching the screen. As increases you might observe different frequency ranges over which the properties of the “image” are drastically different. To describe these observations complete the table on the answer form by adding a row to this table for each such frequency range ...
Structural basis for the fast phase change of DVD-RAM
... pattern of Ge2Sb2Te5 (953 K) and GeTe (1073 K) liquids show a typical halo pattern peculiar to non-crystalline materials, reflecting the lack of the long-range periodicity. These diffraction patterns show a highly disordered state (liquid state) in which both Ge2Sb2Te5 and GeTe have to undergo durin ...
... pattern of Ge2Sb2Te5 (953 K) and GeTe (1073 K) liquids show a typical halo pattern peculiar to non-crystalline materials, reflecting the lack of the long-range periodicity. These diffraction patterns show a highly disordered state (liquid state) in which both Ge2Sb2Te5 and GeTe have to undergo durin ...
Lecture 33 : Chiral molecules and Optical Activity
... The difference between the two refractive indices is known as the linear birefringence. In a case when sample is composed of chiral molecules, we can anticipate that the refractive indices for right and left circularly polarized light need not be same. We denote the difference between these two refr ...
... The difference between the two refractive indices is known as the linear birefringence. In a case when sample is composed of chiral molecules, we can anticipate that the refractive indices for right and left circularly polarized light need not be same. We denote the difference between these two refr ...
1 - contentextra
... Pentylcyanophenyl is used in liquid crystal display devices as it has the following properties: ...
... Pentylcyanophenyl is used in liquid crystal display devices as it has the following properties: ...
Refractive Indices, Order Parameter and Optical Transmittance
... S = 12 h3 cos2 θ − 1i, where θ is angle between optic axis and long molecular axis of a molecule. Macroscopic order parameter (Q) as determined by different properties differs among them and also differs from microscopic order parameter because the effect of the local field is different on different ...
... S = 12 h3 cos2 θ − 1i, where θ is angle between optic axis and long molecular axis of a molecule. Macroscopic order parameter (Q) as determined by different properties differs among them and also differs from microscopic order parameter because the effect of the local field is different on different ...
Experiment 4 Separation of a Mixture
... down by simple chemical means. The periodic table, which we have talked about in class, contains all the elements we know about up to this point. An example of an element is copper (Cu). At room temperature copper is a solid, if the temperature is raised above the melting point of copper it becomes ...
... down by simple chemical means. The periodic table, which we have talked about in class, contains all the elements we know about up to this point. An example of an element is copper (Cu). At room temperature copper is a solid, if the temperature is raised above the melting point of copper it becomes ...
Print Activity - Let`s Talk Science
... Why does it matter? All fluids have a measurable viscosity. Viscosity is the thickness or resistance to flow of a liquid. Water, for instance, has a low viscosity because it pours easily. In comparison, honey is a very thick liquid and can take a long time to pour, so it has a high viscosity. Normal ...
... Why does it matter? All fluids have a measurable viscosity. Viscosity is the thickness or resistance to flow of a liquid. Water, for instance, has a low viscosity because it pours easily. In comparison, honey is a very thick liquid and can take a long time to pour, so it has a high viscosity. Normal ...
Liquid crystal

Liquid crystals (LCs) are matter in a state that has properties between those of conventional liquid and those of solid crystal. For instance, a liquid crystal may flow like a liquid, but its molecules may be oriented in a crystal-like way. There are many different types of liquid-crystal phases, which can be distinguished by their different optical properties (such as birefringence). When viewed under a microscope using a polarized light source, different liquid crystal phases will appear to have distinct textures. The contrasting areas in the textures correspond to domains where the liquid-crystal molecules are oriented in different directions. Within a domain, however, the molecules are well ordered. LC materials may not always be in a liquid-crystal phase (just as water may turn into ice or steam).Liquid crystals can be divided into thermotropic, lyotropic and metallotropic phases. Thermotropic and lyotropic liquid crystals consist of organic molecules. Thermotropic LCs exhibit a phase transition into the liquid-crystal phase as temperature is changed. Lyotropic LCs exhibit phase transitions as a function of both temperature and concentration of the liquid-crystal molecules in a solvent (typically water). Metallotropic LCs are composed of both organic and inorganic molecules; their liquid-crystal transition depends not only on temperature and concentration, but also on the inorganic-organic composition ratio.Examples of liquid crystals can be found both in the natural world and in technological applications. Most contemporary electronic displays use liquid crystals. Lyotropic liquid-crystalline phases are abundant in living systems. For example, many proteins and cell membranes are liquid crystals. Other well-known examples of liquid crystals are solutions of soap and various related detergents, as well as the tobacco mosaic virus.