ppt
... 1669, Nicholas Steno, a Danish physician and natural scientist, discovered through analysis of numerous samples of the same mineral, when measured at the same temperature, the angles between similar crystal faces remain constant regardless of the size or the shape of the crystal. Steno's law is ...
... 1669, Nicholas Steno, a Danish physician and natural scientist, discovered through analysis of numerous samples of the same mineral, when measured at the same temperature, the angles between similar crystal faces remain constant regardless of the size or the shape of the crystal. Steno's law is ...
Poly[(tri-`butyl-phosphine)gold(I)]ammonhim
... formed in the early stages of decomposition. The 31P NMR spectra of the solutions show several peaks other than at d = 97.4 ppm, which have not yet been assigned. The salt in question can also be obtained in a metathesis reaction of the chloride salt [23] with AgBF4 in ethanol (Exp. Part). The produ ...
... formed in the early stages of decomposition. The 31P NMR spectra of the solutions show several peaks other than at d = 97.4 ppm, which have not yet been assigned. The salt in question can also be obtained in a metathesis reaction of the chloride salt [23] with AgBF4 in ethanol (Exp. Part). The produ ...
of a mineral? - Bakersfield College
... • Considered a pure substance • each element has its own atomic number • About 118 known elements (92 natural, 26 synthetic) ...
... • Considered a pure substance • each element has its own atomic number • About 118 known elements (92 natural, 26 synthetic) ...
Chapter 3: Minerals Why do we study minerals? They are the
... [NaCl model] The shape of crystals (habit) is closely linked to the atomic structure. Note that well formed crystal faces will be formed only under certain conditions (e.g. unimpeded growth into a cavity, early crystallization from a melt). In most rocks, crystals have to compete for space and so ar ...
... [NaCl model] The shape of crystals (habit) is closely linked to the atomic structure. Note that well formed crystal faces will be formed only under certain conditions (e.g. unimpeded growth into a cavity, early crystallization from a melt). In most rocks, crystals have to compete for space and so ar ...
CLAY MINERALS
... If no ion substitutes for Si, the entire framework has the composition SiO2 and all valence bonds are satisfied. When Al substitutes for Si in the tetrahedra, interstitial cations are required to maintain charge balance. The openness of these framework structures results in rather large interstitial ...
... If no ion substitutes for Si, the entire framework has the composition SiO2 and all valence bonds are satisfied. When Al substitutes for Si in the tetrahedra, interstitial cations are required to maintain charge balance. The openness of these framework structures results in rather large interstitial ...
Ch_ 2-1Mineral
... Covalent bond: or electron sharing bond is the strongest chemical bond. sharing of election when orbitals overlap Electrons in outer orbital are filled as in stable inert gas configuration. Hence ionic bonding also shares some electrons and covalent bond often contain electrostatic charge, therefor ...
... Covalent bond: or electron sharing bond is the strongest chemical bond. sharing of election when orbitals overlap Electrons in outer orbital are filled as in stable inert gas configuration. Hence ionic bonding also shares some electrons and covalent bond often contain electrostatic charge, therefor ...
as a PDF
... nal with space group С 66 , Z=2 (KLiSO4). In researched crystals atoms of oxygen form different number nonequivalent sublattices. In Li2SO4 their number is four and lengths of bonds S-O1=1.470, S-O2=1.471, S-O3=1.474, S-O4=1.478 Å [2]; in Na2SO4 – two [3] and K2SO4 it is three [4]. In monoclinic pha ...
... nal with space group С 66 , Z=2 (KLiSO4). In researched crystals atoms of oxygen form different number nonequivalent sublattices. In Li2SO4 their number is four and lengths of bonds S-O1=1.470, S-O2=1.471, S-O3=1.474, S-O4=1.478 Å [2]; in Na2SO4 – two [3] and K2SO4 it is three [4]. In monoclinic pha ...
UNIT 2: Minerals
... I can… Identify and describe the criteria for crystals classification Explain the concept of the Unit Cell and tell why it is necessary in explaining mineral structures Determine the identity of minerals based ont their properties. ...
... I can… Identify and describe the criteria for crystals classification Explain the concept of the Unit Cell and tell why it is necessary in explaining mineral structures Determine the identity of minerals based ont their properties. ...
Self-Assembly of the First Copper (II) Infinite 2D Network with Large
... titmb ligands with “up” and “down” orientations and the plane containing copper atoms Cu1, Cu1A, Cu1B, Cu1C, and Cu1D are strictly parallel with each other. The distance between these two benzene rings is 10.00 Å, and the plane of copper atoms is at the middle of the two benzene ring planes; i.e., e ...
... titmb ligands with “up” and “down” orientations and the plane containing copper atoms Cu1, Cu1A, Cu1B, Cu1C, and Cu1D are strictly parallel with each other. The distance between these two benzene rings is 10.00 Å, and the plane of copper atoms is at the middle of the two benzene ring planes; i.e., e ...
GEOLOGY PPT
... to describe their atomic "lattice" structure. A crystal's lattice is a three dimensional network of atoms that are arranged in a symmetrical pattern. ...
... to describe their atomic "lattice" structure. A crystal's lattice is a three dimensional network of atoms that are arranged in a symmetrical pattern. ...
(Ala-Leu-Ac7c-)2-OMe - Molecular Biophysics Unit
... programme (13). Partial structure expansion with the tangent formula led to the complete molecule in several cycles (14). Hydrogen atoms were added in idealized positions. Anisotropic refinement on F2 values resulted in R1 ¼ 15.1% for 497 parameters and 1477 independent data with |F0| > 2r (nearly t ...
... programme (13). Partial structure expansion with the tangent formula led to the complete molecule in several cycles (14). Hydrogen atoms were added in idealized positions. Anisotropic refinement on F2 values resulted in R1 ¼ 15.1% for 497 parameters and 1477 independent data with |F0| > 2r (nearly t ...
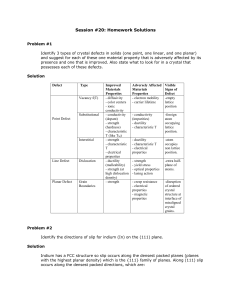
Session #20: Homework Solutions
... Problem #4 (a) List four different defects in crystalline solids. (b) What evidence is available supporting the actual existence of the listed defects? Solution Many answers are acceptable. For example: (1) dopant elements in semiconductors → affect electrical conductivity (2) vacancies in close pac ...
... Problem #4 (a) List four different defects in crystalline solids. (b) What evidence is available supporting the actual existence of the listed defects? Solution Many answers are acceptable. For example: (1) dopant elements in semiconductors → affect electrical conductivity (2) vacancies in close pac ...
LECTURE 5- CHEMISTRY OF MINERALS REVISED
... crystallographic structure. Each polymorph has its own physical properties and a distinct internal arrangement of its atoms and/or ions. Polymorphism is an expression of the fact that crystal structure is not determined exclusively by chemical composition. In other words, for some substances, the sa ...
... crystallographic structure. Each polymorph has its own physical properties and a distinct internal arrangement of its atoms and/or ions. Polymorphism is an expression of the fact that crystal structure is not determined exclusively by chemical composition. In other words, for some substances, the sa ...
Crystal Bonding
... The bonding of atoms in semiconductors has primary influence of forming the lattice of any solar cell and is accomplished by electrostatic forces and by the tendency of atoms to fill their outer shells. Interatomic attraction is balanced by short-range repulsion due to strong resistance of atoms aga ...
... The bonding of atoms in semiconductors has primary influence of forming the lattice of any solar cell and is accomplished by electrostatic forces and by the tendency of atoms to fill their outer shells. Interatomic attraction is balanced by short-range repulsion due to strong resistance of atoms aga ...
5-1
... 34. Each type of mineral crystal is characterized by what? _______________________________________________________________ 35. What hinders the growth of single, large crystals? _______________________________________________________________ __________________________________________________________ ...
... 34. Each type of mineral crystal is characterized by what? _______________________________________________________________ 35. What hinders the growth of single, large crystals? _______________________________________________________________ __________________________________________________________ ...
References
... The possibility of the simultaneous existence of different forms of nickel (II) complex in the presence of - diimine ligands with different structure can determine some of their physico-chemical properties . Thus, the non-equivalence of the two bromide ions in the complex I and different Ni-Br bond ...
... The possibility of the simultaneous existence of different forms of nickel (II) complex in the presence of - diimine ligands with different structure can determine some of their physico-chemical properties . Thus, the non-equivalence of the two bromide ions in the complex I and different Ni-Br bond ...
- Opus
... and 1.0 M NaOH (100 µL, 0.10 mmol) was carefully layered on top of an aqueous solution (1 mL) of metal nitrate hexahydrate salt (0.05 mmol) separated by a layer of DMF (0.5 mL). Data for 1: Co(NO3)2·6H2O (15.5 mg, 0.05 mmol). Pink crystals were harvested after a week. Yield 28 mg (74%). Found: C, 51 ...
... and 1.0 M NaOH (100 µL, 0.10 mmol) was carefully layered on top of an aqueous solution (1 mL) of metal nitrate hexahydrate salt (0.05 mmol) separated by a layer of DMF (0.5 mL). Data for 1: Co(NO3)2·6H2O (15.5 mg, 0.05 mmol). Pink crystals were harvested after a week. Yield 28 mg (74%). Found: C, 51 ...
PESUnit5minerals12-13
... – Atoms can be dissolved in solutions of water. If water evaporates or changes temperature or gets too many atoms in it the dissolved atoms may form crystalline solids…minerals! – Happens at bottom of bodies of water, especially where there is volcanically heated hot water dissolving minerals and th ...
... – Atoms can be dissolved in solutions of water. If water evaporates or changes temperature or gets too many atoms in it the dissolved atoms may form crystalline solids…minerals! – Happens at bottom of bodies of water, especially where there is volcanically heated hot water dissolving minerals and th ...
minerals!
... ○ It has never been a part of a living thing. has a definite chemical composition. ○ It has specific elements that make up its compounds. ○ NaCl is always halite, which is table salt. It is a compound of sodium and chlorine. ...
... ○ It has never been a part of a living thing. has a definite chemical composition. ○ It has specific elements that make up its compounds. ○ NaCl is always halite, which is table salt. It is a compound of sodium and chlorine. ...
Topic/Objective: Full Name: Class: Period: _____ Date: Tutor Use
... An element is matter that is made up of the atoms that have the same number of _protons _ in their nucleus. But not all atoms of the same element are the same. Some elements have the same number of protons but a different number of neutrons in the nucleus, these are called ___isotopes__. Most su ...
... An element is matter that is made up of the atoms that have the same number of _protons _ in their nucleus. But not all atoms of the same element are the same. Some elements have the same number of protons but a different number of neutrons in the nucleus, these are called ___isotopes__. Most su ...
Practice Qs - Unit 6a
... They all contain metals w/ more than 1 possible charge (oxidation state). + ion: always first (element name or ammonium Charge of metal ion goes as Roman numeral in ( ) ...
... They all contain metals w/ more than 1 possible charge (oxidation state). + ion: always first (element name or ammonium Charge of metal ion goes as Roman numeral in ( ) ...
Crystal structure of boron-rich metal borides

Metals, and specifically rare-earth elements (RE), form numerous chemical complexes with boron. Their crystal structure and chemical bonding depend strongly on the metal element M and on its atomic ratio to boron. When B/M ratio exceeds 12, boron atoms form B12 icosahedra which are linked into a three-dimensional boron framework, and the metal atoms reside in the voids of this framework. Those icosahedra are basic structural units of most allotropes of boron and boron-rich rare-earth borides. In such borides, metal atoms donate electrons to the boron polyhedra, and thus these compounds are regarded as electron-deficient solids.The crystal structures of many boron-rich borides can be attributed to certain types including MgAlB14, YB66, REB41Si1.2, B4C and other, more complex types such as RExB12C0.33Si3.0. Some of these formulas, for example B4C, YB66 and MgAlB14, historically reflect the idealistic structures, whereas the experimentally determined composition is nonstoichiometric and corresponds to fractional indexes. Boron-rich borides are usually characterized by large and complex unit cells, which can contain more than 1500 atomic sites and feature extended structures shaped as ""tubes"" and large modular polyhedra (""superpolyhedra""). Many of those sites have partial occupancy, meaning that the probability to find them occupied with a certain atom is smaller than one and thus that only some of them are filled with atoms. Scandium is distinguished among the rare-earth elements by that it forms numerous borides with uncommon structure types; this property of scandium is attributed to its relatively small atomic and ionic radii. Crystals of the specific rare-earth boride YB66 are used as X-ray monochromators for selecting X-rays with certain energies (in the 1–2 keV range) out of synchrotron radiation. Other rare-earth borides may find application as thermoelectric materials, owing to their low thermal conductivity; the latter originates from their complex, ""amorphous-like"", crystal structure.
![Poly[(tri-`butyl-phosphine)gold(I)]ammonhim](http://s1.studyres.com/store/data/016955699_1-3ddf66273799341f4663a1d22989efb5-300x300.png)