Diphenyldichlorophosphonium Trichloride−Chlorine Solvate 1:1
... onto a solidified solution of 2.20 g (7.5 mmol) of PPh2Cl3 dissolved in 5 mL of chloroform in a 50 mL Schlenk pressure vessel immersed in liquid N2. After all the chlorine had been added, the vessel was closed and left to warm to ambient temperature. When the contents of the vessel melted, a yellow ...
... onto a solidified solution of 2.20 g (7.5 mmol) of PPh2Cl3 dissolved in 5 mL of chloroform in a 50 mL Schlenk pressure vessel immersed in liquid N2. After all the chlorine had been added, the vessel was closed and left to warm to ambient temperature. When the contents of the vessel melted, a yellow ...
crystals - MDPI.com
... technique. Li et al. (2008) [12] reported the synthesis of a zinc(II) 4-(5H-tetrazolyl)benzoic coordination polymer exhibiting a GSI topology. This particular MOF was produced in a one pot hydrothermal synthesis set at 140 °C utilizing sodium 4-cyanobenzoic acid, sodium azide, and zinc bromide as th ...
... technique. Li et al. (2008) [12] reported the synthesis of a zinc(II) 4-(5H-tetrazolyl)benzoic coordination polymer exhibiting a GSI topology. This particular MOF was produced in a one pot hydrothermal synthesis set at 140 °C utilizing sodium 4-cyanobenzoic acid, sodium azide, and zinc bromide as th ...
Effects of Reaction Gel Dehydration on the Synthesis of Cu(NC5H5
... circles represent copper, vanadium, oxygen, fluorine, nitrogen, and carbon, respectively. Selected pyridine rings and non-water hydrogen atoms are omitted for clarity. Dashed black lines denote hydrogen bonding. ...
... circles represent copper, vanadium, oxygen, fluorine, nitrogen, and carbon, respectively. Selected pyridine rings and non-water hydrogen atoms are omitted for clarity. Dashed black lines denote hydrogen bonding. ...
Structural Studies of the Catalytic Reaction
... scaled with DENZO and SCALEPACK (13) and are summarized in Table I. The Matthews coefficient (14) for the tmHspAT crystals was 2.53 Å3 Da⫺1, corresponding to a solvent content of about 51%. SOLVE version 2.02 (15) was used to determine the heavy atom substructure using the x-ray data covering the re ...
... scaled with DENZO and SCALEPACK (13) and are summarized in Table I. The Matthews coefficient (14) for the tmHspAT crystals was 2.53 Å3 Da⫺1, corresponding to a solvent content of about 51%. SOLVE version 2.02 (15) was used to determine the heavy atom substructure using the x-ray data covering the re ...
Mineral Groups
... group. Feldspar and quartz are the two most common silicate minerals. Both are extremely common rock-forming minerals. The basic building block for all silicate minerals is the silica tetrahedron, which is illustrated in Figure 1.1. To create the wide variety of silicate minerals, this pyramid-shape ...
... group. Feldspar and quartz are the two most common silicate minerals. Both are extremely common rock-forming minerals. The basic building block for all silicate minerals is the silica tetrahedron, which is illustrated in Figure 1.1. To create the wide variety of silicate minerals, this pyramid-shape ...
Mineral Groups - cloudfront.net
... group. Feldspar and quartz are the two most common silicate minerals. Both are extremely common rock-forming minerals. The basic building block for all silicate minerals is the silica tetrahedron, which is illustrated in Figure 1.1. To create the wide variety of silicate minerals, this pyramid-shape ...
... group. Feldspar and quartz are the two most common silicate minerals. Both are extremely common rock-forming minerals. The basic building block for all silicate minerals is the silica tetrahedron, which is illustrated in Figure 1.1. To create the wide variety of silicate minerals, this pyramid-shape ...
Crystal Modelling
... Make a crystal of the substance. The crystal must be very pure and perfect. Stage 2 ...
... Make a crystal of the substance. The crystal must be very pure and perfect. Stage 2 ...
Key Stage 3 – Crystal Modelling
... Make a crystal of the substance. The crystal must be very pure and perfect. Stage 2 ...
... Make a crystal of the substance. The crystal must be very pure and perfect. Stage 2 ...
Drug-containing coordination and hydrogen bonding networks
... solubility and bioavailability of the free with respect to the coordinated drug. Recently,1 we have shown that coordination compounds of the neuroleptic drug gabapentin with zinc and copper can be obtained easily and quantitatively by mechanical co-grinding of solid gabapentin and of the inorganic s ...
... solubility and bioavailability of the free with respect to the coordinated drug. Recently,1 we have shown that coordination compounds of the neuroleptic drug gabapentin with zinc and copper can be obtained easily and quantitatively by mechanical co-grinding of solid gabapentin and of the inorganic s ...
THE CRYSTAL STRUCTURE OF ALLOCHALCOSELITE, Cu
... The crystal structure of allochalcoselite, Cu+Cu2+5PbO2(SeO3)2Cl5, a new mineral species from the fumaroles of the Great Tolbachik fissure eruption, in the Kamchatka Peninsula, Russia, has been solved from single-crystal X-ray-diffraction data by direct methods and refined to R1 = 0.047 for 1235 uni ...
... The crystal structure of allochalcoselite, Cu+Cu2+5PbO2(SeO3)2Cl5, a new mineral species from the fumaroles of the Great Tolbachik fissure eruption, in the Kamchatka Peninsula, Russia, has been solved from single-crystal X-ray-diffraction data by direct methods and refined to R1 = 0.047 for 1235 uni ...
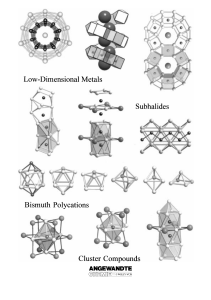
From the Metal to the Molecule
... intermetallic phases with bismuth. Previous studies suggest that this is a basic requirement for the formation of the corresponding subhalides. Consequently, we can speak of the partial oxidation of intermetallic phases. 2) The electronegativities of these transition metal elements hardly differ fro ...
... intermetallic phases with bismuth. Previous studies suggest that this is a basic requirement for the formation of the corresponding subhalides. Consequently, we can speak of the partial oxidation of intermetallic phases. 2) The electronegativities of these transition metal elements hardly differ fro ...
Full-Text PDF
... phase were synthesized via a one-step hydrothermal method using an ionic complex as template. The ionic complex template was formed by interaction between cetyltrimethylammonium ions and metal complex ion [M(EDTA)]2− (M = Co or Ni)]. The materials were characterized by X-ray diffraction, scanning el ...
... phase were synthesized via a one-step hydrothermal method using an ionic complex as template. The ionic complex template was formed by interaction between cetyltrimethylammonium ions and metal complex ion [M(EDTA)]2− (M = Co or Ni)]. The materials were characterized by X-ray diffraction, scanning el ...
Ionic compound
... Aqueous: Yes (if soluble) Boiling point and Melting Point High Solubility in water Often high Thermal conductivity Low ...
... Aqueous: Yes (if soluble) Boiling point and Melting Point High Solubility in water Often high Thermal conductivity Low ...
Slide 1
... PT contains exactly 6.02x1023 atoms of that element. • And is equal to how much 1 mole of that sample would weigh in grams ...
... PT contains exactly 6.02x1023 atoms of that element. • And is equal to how much 1 mole of that sample would weigh in grams ...
The real structure of Na3BiO4 by electron microscopy, HR
... exhibits several peaks of small amounts of sodium hydroxide and bismuth oxide. Data reduction was performed using the GUFI program [30]. Indexing with ITO [31] led to a hexagonal cell with lattice parameters given in Table 1. The number of formula units (Na0.75Bi0.25O) per unit cell was deduced to b ...
... exhibits several peaks of small amounts of sodium hydroxide and bismuth oxide. Data reduction was performed using the GUFI program [30]. Indexing with ITO [31] led to a hexagonal cell with lattice parameters given in Table 1. The number of formula units (Na0.75Bi0.25O) per unit cell was deduced to b ...
T-Shaped Molecular Building Units in the Porous Structure of Ag(4,4
... zeolite-like attributes2 and others having important electronic3 and magnetic4 properties. One of the simplest strategies employed in the production of such 3-D networks is schematically illustrated in Figure 1a,b, where open-framework diamondlike networks have been achieved by the addition copolyme ...
... zeolite-like attributes2 and others having important electronic3 and magnetic4 properties. One of the simplest strategies employed in the production of such 3-D networks is schematically illustrated in Figure 1a,b, where open-framework diamondlike networks have been achieved by the addition copolyme ...
Using APL format - Massachusetts Institute of Technology
... Much research in recent years has been focused on photonic crystals—periodic dielectric 共or metallic兲 structures with a photonic band gap 共PBG兲, a range of frequencies in which light is forbidden to propagate.1,2 Photonic crystals provide an unprecedented degree of control over light, introducing th ...
... Much research in recent years has been focused on photonic crystals—periodic dielectric 共or metallic兲 structures with a photonic band gap 共PBG兲, a range of frequencies in which light is forbidden to propagate.1,2 Photonic crystals provide an unprecedented degree of control over light, introducing th ...
Atomic disordering in YB56 detected by high
... the basis of the structure model of YB56 determined by X-ray di raction as listed in table 1. HREM images calculated along the [100] direction are shown in ® gure 2. The image calculations were carried out for various under-def ocus values and crystal thicknesses to determine the imaging condition ...
... the basis of the structure model of YB56 determined by X-ray di raction as listed in table 1. HREM images calculated along the [100] direction are shown in ® gure 2. The image calculations were carried out for various under-def ocus values and crystal thicknesses to determine the imaging condition ...
Chemical Equations
... C6H12O6 + O2 H2O + CO2 Start with either C or H. Oxygen appears in every formula; save it till last. ...
... C6H12O6 + O2 H2O + CO2 Start with either C or H. Oxygen appears in every formula; save it till last. ...
Class XII Chapter 1
... types are held by the electrostatic force of attraction. In metallic crystals, the electrostatic force acts between the positive ions and the electrons. In ionic crystals, it acts between the oppositely-charged ions. Hence, both have high melting points. The basis of differences between metallic and ...
... types are held by the electrostatic force of attraction. In metallic crystals, the electrostatic force acts between the positive ions and the electrons. In ionic crystals, it acts between the oppositely-charged ions. Hence, both have high melting points. The basis of differences between metallic and ...
ppt - National Single Crystal X
... • Reflections effected by twinning show-up in the least-squares refinement with F(obs) >> F(calc) • Overlapping reflections necessarily have the same Theta value within a tolerance. • Generate a list of implied possible twin axes based on the above observations. • Test each proposed twin law for its ...
... • Reflections effected by twinning show-up in the least-squares refinement with F(obs) >> F(calc) • Overlapping reflections necessarily have the same Theta value within a tolerance. • Generate a list of implied possible twin axes based on the above observations. • Test each proposed twin law for its ...
Crystal structure of oxygen-evolving photosystem II at 1.9 Ε
... YZ (D1-Tyr161) is located between the Mn4CaO5-cluster and PSII-RC, and functions to mediate electron transfer between the Mn4CaO5-cluster and PSII-RC. An extensive hydrogen-bonding network was found between D1-Tyr161 and the Mn4CaO5-cluster, and from D1-Tyr161 to the lumenal bulk phase. D1-Tyr161 wa ...
... YZ (D1-Tyr161) is located between the Mn4CaO5-cluster and PSII-RC, and functions to mediate electron transfer between the Mn4CaO5-cluster and PSII-RC. An extensive hydrogen-bonding network was found between D1-Tyr161 and the Mn4CaO5-cluster, and from D1-Tyr161 to the lumenal bulk phase. D1-Tyr161 wa ...
solid state - einstein classes
... 1. Tetrahedral void 2. Octahedral void Tetrahedral void : If one sphere is placed upon three other spheres which are touching one other tetrahedral structure results. ...
... 1. Tetrahedral void 2. Octahedral void Tetrahedral void : If one sphere is placed upon three other spheres which are touching one other tetrahedral structure results. ...
Crystal Properties and Growth of Semiconductors
... primitive vectors. Note that, in a primitive cell, the lattice points at the corners are shared with adjacent cells; thus, the effective number of lattice points belonging to the primitive cell is always unity. Since there are many different ways of placing atoms in a volume, the distances and orien ...
... primitive vectors. Note that, in a primitive cell, the lattice points at the corners are shared with adjacent cells; thus, the effective number of lattice points belonging to the primitive cell is always unity. Since there are many different ways of placing atoms in a volume, the distances and orien ...
solid state
... When a cation of higher valence is added to an ionic solid as an impurity to it, the cation of higher valence replaces more than one cation of lower valence so as to keep the crystal electrically neutral. As a result, some sites become vacant. For example, when Sr 2+ is added to NaCl, each Sr2+ ion ...
... When a cation of higher valence is added to an ionic solid as an impurity to it, the cation of higher valence replaces more than one cation of lower valence so as to keep the crystal electrically neutral. As a result, some sites become vacant. For example, when Sr 2+ is added to NaCl, each Sr2+ ion ...
Crystal structure of boron-rich metal borides

Metals, and specifically rare-earth elements (RE), form numerous chemical complexes with boron. Their crystal structure and chemical bonding depend strongly on the metal element M and on its atomic ratio to boron. When B/M ratio exceeds 12, boron atoms form B12 icosahedra which are linked into a three-dimensional boron framework, and the metal atoms reside in the voids of this framework. Those icosahedra are basic structural units of most allotropes of boron and boron-rich rare-earth borides. In such borides, metal atoms donate electrons to the boron polyhedra, and thus these compounds are regarded as electron-deficient solids.The crystal structures of many boron-rich borides can be attributed to certain types including MgAlB14, YB66, REB41Si1.2, B4C and other, more complex types such as RExB12C0.33Si3.0. Some of these formulas, for example B4C, YB66 and MgAlB14, historically reflect the idealistic structures, whereas the experimentally determined composition is nonstoichiometric and corresponds to fractional indexes. Boron-rich borides are usually characterized by large and complex unit cells, which can contain more than 1500 atomic sites and feature extended structures shaped as ""tubes"" and large modular polyhedra (""superpolyhedra""). Many of those sites have partial occupancy, meaning that the probability to find them occupied with a certain atom is smaller than one and thus that only some of them are filled with atoms. Scandium is distinguished among the rare-earth elements by that it forms numerous borides with uncommon structure types; this property of scandium is attributed to its relatively small atomic and ionic radii. Crystals of the specific rare-earth boride YB66 are used as X-ray monochromators for selecting X-rays with certain energies (in the 1–2 keV range) out of synchrotron radiation. Other rare-earth borides may find application as thermoelectric materials, owing to their low thermal conductivity; the latter originates from their complex, ""amorphous-like"", crystal structure.