THERMODYNAMICS OF NONCOMMUTATIVE BLACK HOLE
... to the entropy of black hole due to noncommutative space time. Noncommutativity is expected to be relevant at the Planck scale where it is known that usual semi classical Considerations break down. It is therefore reasonable to expect that noncommutativity would modify the entropy. If we consider th ...
... to the entropy of black hole due to noncommutative space time. Noncommutativity is expected to be relevant at the Planck scale where it is known that usual semi classical Considerations break down. It is therefore reasonable to expect that noncommutativity would modify the entropy. If we consider th ...
PS5, Thermo Thermodynamics Standards: 3. Energy cannot be
... Thermodynamics Standards: 3. Energy cannot be created or destroyed, although in many processes energy is transferred to the environment as heat. As a basis for understanding this concept: a. Students know heat flow and work are two forms of energy transfer between systems. b. Students know that the ...
... Thermodynamics Standards: 3. Energy cannot be created or destroyed, although in many processes energy is transferred to the environment as heat. As a basis for understanding this concept: a. Students know heat flow and work are two forms of energy transfer between systems. b. Students know that the ...
Entropy and Free Energy
... State functions are properties that are determined by the state of the system, regardless of how that condition was achieved. energy, enthalpy, pressure, volume, temperature, entropy. ...
... State functions are properties that are determined by the state of the system, regardless of how that condition was achieved. energy, enthalpy, pressure, volume, temperature, entropy. ...
Document
... The internal energy,"u", is a "state function". This means that it is a property that depends only on the present state (T, P, phase) of the system. ...
... The internal energy,"u", is a "state function". This means that it is a property that depends only on the present state (T, P, phase) of the system. ...
Lecture 5
... V This is a simplified version of an engine converting heat to work that was important in the development of thermodynamics, which tells us the limits to the efficiency of such engines. In the Carnot Cycle a working fluid such as the one we have just investigated is taken through expansion-compressi ...
... V This is a simplified version of an engine converting heat to work that was important in the development of thermodynamics, which tells us the limits to the efficiency of such engines. In the Carnot Cycle a working fluid such as the one we have just investigated is taken through expansion-compressi ...
Defects - Script
... This is a simple relation always best suited for systems under constant pressure and also clarifying why we tend to think of enthalpy as heat. dH is a measure of of the energy needed to form a substance in a given state, it is occasionally also called the heat of formation (always refering to the di ...
... This is a simple relation always best suited for systems under constant pressure and also clarifying why we tend to think of enthalpy as heat. dH is a measure of of the energy needed to form a substance in a given state, it is occasionally also called the heat of formation (always refering to the di ...
Free Energy Examples
... If the volume increased the system did positive work on its surroundings. C. Since pressure is constant, enthalpy is conserved. D. If the volume increased heat must have been added to the system. E. If the enthalpy is constant, the volume must have decreased. ΔU= Q- W H=U+PV ...
... If the volume increased the system did positive work on its surroundings. C. Since pressure is constant, enthalpy is conserved. D. If the volume increased heat must have been added to the system. E. If the enthalpy is constant, the volume must have decreased. ΔU= Q- W H=U+PV ...
Lecture 4
... The entropy of an isolated system increases in any irreversible process and is unaltered in any reversible process. This is the principle of increasing entropy. The fact that the entropy of an isolated system can never decrease in a process provides a direction for the sequence of natural events. ...
... The entropy of an isolated system increases in any irreversible process and is unaltered in any reversible process. This is the principle of increasing entropy. The fact that the entropy of an isolated system can never decrease in a process provides a direction for the sequence of natural events. ...
EGU2016-10322 - CO Meeting Organizer
... evolution of the Earth. For instance, they influenced the evolution of a plausible primordial magma ocean, and, as a result, the differentiation of the Earth mantle and crust. Further, they control the dynamic of volcanic eruptions. Because of that, modelling the viscosity or the heat capacity of si ...
... evolution of the Earth. For instance, they influenced the evolution of a plausible primordial magma ocean, and, as a result, the differentiation of the Earth mantle and crust. Further, they control the dynamic of volcanic eruptions. Because of that, modelling the viscosity or the heat capacity of si ...
Entropy
... • Consider two scenarios: – An ideal gas reversibly and adiabatically expands from volume Vi to Vf . ∗ The change in entropy is zero, dS = n0, because dQrev = 0. – An ideal gas freely and adiabatically expands from volume Vi to Vf . ∗ No work is done in a free expansion. ∗ Since this is adiabatic, a ...
... • Consider two scenarios: – An ideal gas reversibly and adiabatically expands from volume Vi to Vf . ∗ The change in entropy is zero, dS = n0, because dQrev = 0. – An ideal gas freely and adiabatically expands from volume Vi to Vf . ∗ No work is done in a free expansion. ∗ Since this is adiabatic, a ...
BOLTZMANN ENTROPY: PROBABILITY AND INFORMATION The
... We have presented a rigorous axiomatic derivation of Boltzmann entropy on the basis of the axioms of additivity and increasing law of entropy consistent with two basic properties of thermodynamic entropy [11]. The method is superior, both mathematically and physically, to the existing methods (excep ...
... We have presented a rigorous axiomatic derivation of Boltzmann entropy on the basis of the axioms of additivity and increasing law of entropy consistent with two basic properties of thermodynamic entropy [11]. The method is superior, both mathematically and physically, to the existing methods (excep ...
Heat Chapter 12: Thermodynamics
... • Heat does not flow spontaneously from a colder to a warmer body. • In a thermal cycle, heat energy cannot be completely transformed into mechanical work. • The total entropy of the universe increases in every natural process. The Third Law of Thermodynamics – It is not possible to lower temperatur ...
... • Heat does not flow spontaneously from a colder to a warmer body. • In a thermal cycle, heat energy cannot be completely transformed into mechanical work. • The total entropy of the universe increases in every natural process. The Third Law of Thermodynamics – It is not possible to lower temperatur ...
Entropy in chemical thermodynamics
... The statistical definition of entropy matches up with the thermodynamic formula for calculating entropy, because adding heat to a system, which increases its classical thermodynamic entropy, also increases the system's thermal fluctuations, so giving an increased lack of information about the exact ...
... The statistical definition of entropy matches up with the thermodynamic formula for calculating entropy, because adding heat to a system, which increases its classical thermodynamic entropy, also increases the system's thermal fluctuations, so giving an increased lack of information about the exact ...
Entropy change of an ideal gas determination with no reversible
... In simulation (b) the gas reaches the final equilibrium much faster than in simulation (a). This happens because in case (a) the walls have a lower thermal conductivity than in the case (b), which explains why the temperature rises over 400 K, due to the strong gas compression, before it decreases r ...
... In simulation (b) the gas reaches the final equilibrium much faster than in simulation (a). This happens because in case (a) the walls have a lower thermal conductivity than in the case (b), which explains why the temperature rises over 400 K, due to the strong gas compression, before it decreases r ...
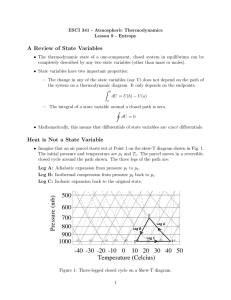
1. Introduction (Chapters 1 and 2 ) Goal: Review the empirical laws
... basically energy conservation. Note W and Q are not state functions since they are not functions of state variables (e.g. p, V for a gas with NA kmoles ). In differential form the first law of TD is: dU Q pdV For example one can move the system from points A to C through different paths (throug ...
... basically energy conservation. Note W and Q are not state functions since they are not functions of state variables (e.g. p, V for a gas with NA kmoles ). In differential form the first law of TD is: dU Q pdV For example one can move the system from points A to C through different paths (throug ...
Lecture 4
... 1 at x 0 , p0 , and particle 2 at x, p. So in this case 2 volumes of h2 correspond to a single state. In the N particle system in 3 dimensions there are N ! configurations of the particles amongst different phase space volumes of h3N that correspond to a single quantum state. This is the N! factor t ...
... 1 at x 0 , p0 , and particle 2 at x, p. So in this case 2 volumes of h2 correspond to a single state. In the N particle system in 3 dimensions there are N ! configurations of the particles amongst different phase space volumes of h3N that correspond to a single quantum state. This is the N! factor t ...
Chapter 18 - Evangel University
... (c) Heating hydrogen gas from 600C to 800C Randomness ________ ...
... (c) Heating hydrogen gas from 600C to 800C Randomness ________ ...
Chap 7 - College of Science | Oregon State University
... Fahrenheit based on human body temperature and freezing point of a mixture of water and salt. - Water freezes at 32oF; boils at 212oF. - There are 180 degrees between these two points. Kelvin based on absolute zero. - Also called absolute scale. - Absolute zero is the lowest possible temperature tha ...
... Fahrenheit based on human body temperature and freezing point of a mixture of water and salt. - Water freezes at 32oF; boils at 212oF. - There are 180 degrees between these two points. Kelvin based on absolute zero. - Also called absolute scale. - Absolute zero is the lowest possible temperature tha ...
+ p
... The second law of thermodynamics Joule and Carnot studied ways to improve the efficiency of steam engines. Is it possible for a thermodynamic system to move from state A to state B without any net energy being put into the system from outside? A single experimental quantity, dubbed entropy, made it ...
... The second law of thermodynamics Joule and Carnot studied ways to improve the efficiency of steam engines. Is it possible for a thermodynamic system to move from state A to state B without any net energy being put into the system from outside? A single experimental quantity, dubbed entropy, made it ...
Nonextensivity-Nonintensivity
... In thermodynamics, properties (variables) are classified as being either extensive or intensive. When properties of a system are independent of the number of particles present in the system, they are called "intensive properties (variables)". Otherwise, those properties (variables) are called extens ...
... In thermodynamics, properties (variables) are classified as being either extensive or intensive. When properties of a system are independent of the number of particles present in the system, they are called "intensive properties (variables)". Otherwise, those properties (variables) are called extens ...
Entropy in thermodynamics and information theory
There are close parallels between the mathematical expressions for the thermodynamic entropy, usually denoted by S, of a physical system in the statistical thermodynamics established by Ludwig Boltzmann and J. Willard Gibbs in the 1870s, and the information-theoretic entropy, usually expressed as H, of Claude Shannon and Ralph Hartley developed in the 1940s. Shannon, although not initially aware of this similarity, commented on it upon publicizing information theory in A Mathematical Theory of Communication.This article explores what links there are between the two concepts, and how far they can be regarded as connected.