The Kinetic Theory of Gases - Department of Applied Physics
... n Chapter 19 we discussed the properties of an ideal gas, using such macroscopic variables as pressure, volume, and temperature. We shall now show that such large-scale properties can be described on a microscopic scale, where matter is treated as a collection of molecules. Newton’s laws of motion a ...
... n Chapter 19 we discussed the properties of an ideal gas, using such macroscopic variables as pressure, volume, and temperature. We shall now show that such large-scale properties can be described on a microscopic scale, where matter is treated as a collection of molecules. Newton’s laws of motion a ...
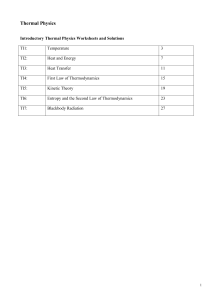
thermodynamics type 1
... uniform throughout, made up of one phase only, pure liquid. solid, gas. A system is said to be heterogeneous if it consists of two or more phases, liquid in contact with vapour. STATE OF A SYSTEM : The state of a system is defined by a particular set of its measurable properties. For example, we can ...
... uniform throughout, made up of one phase only, pure liquid. solid, gas. A system is said to be heterogeneous if it consists of two or more phases, liquid in contact with vapour. STATE OF A SYSTEM : The state of a system is defined by a particular set of its measurable properties. For example, we can ...
Thermochemistry Thermochemistry
... Enthalpy and change of state • There is usually a flow of heat when there is a change of state (solid to liquid, liquid to vapour) • This is usually expressed as the enthalpy of fusion or enthalpy of vaporization. E.g. H2O(s) 6 H2O(l) ∆H=6.01 kJ mol-1 H2O(l) 6 H2O(g) ∆H=44.0 kJ mol-1 • This is ofte ...
... Enthalpy and change of state • There is usually a flow of heat when there is a change of state (solid to liquid, liquid to vapour) • This is usually expressed as the enthalpy of fusion or enthalpy of vaporization. E.g. H2O(s) 6 H2O(l) ∆H=6.01 kJ mol-1 H2O(l) 6 H2O(g) ∆H=44.0 kJ mol-1 • This is ofte ...
1 CHAPTER 17 CHEMICAL THERMODYNAMICS 17.1 Equilibrium
... quoted as molar quantities at a specific temperature (often 25 oC) and pressure (often one atmosphere). The usual convention is to write A + B → C ...
... quoted as molar quantities at a specific temperature (often 25 oC) and pressure (often one atmosphere). The usual convention is to write A + B → C ...
PDF
... motor can operate fully loaded 24 hours a day. If ―intermediate‖ is shown, a time interval will also appear. This means the motor can operate at full load for the specified period. The motor should then be stopped and allowed to cool before starting again. Considerations of steady state heat transfe ...
... motor can operate fully loaded 24 hours a day. If ―intermediate‖ is shown, a time interval will also appear. This means the motor can operate at full load for the specified period. The motor should then be stopped and allowed to cool before starting again. Considerations of steady state heat transfe ...
Second Law of Thermodynamics
... many practical applications. For example it explains the limits of efficiency for heat engines and refrigerators. To develop a better understanding of this law, try these conceptual questions. ...
... many practical applications. For example it explains the limits of efficiency for heat engines and refrigerators. To develop a better understanding of this law, try these conceptual questions. ...
THERMODYNAMICS LECTURE NOTES
... understanding the patterns of energy change. More specifically, thermodynamics deals with (a) energy conversion and (b) the direction of change. Basis of thermodynamics is experimental observation. In that sense it is an empirical science. The principles of thermodynamics are summarized in the form ...
... understanding the patterns of energy change. More specifically, thermodynamics deals with (a) energy conversion and (b) the direction of change. Basis of thermodynamics is experimental observation. In that sense it is an empirical science. The principles of thermodynamics are summarized in the form ...
Entropy
... value, because teh mole fractions are less than one, and ln of a number less than one will be negative. ...
... value, because teh mole fractions are less than one, and ln of a number less than one will be negative. ...