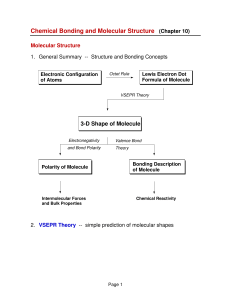
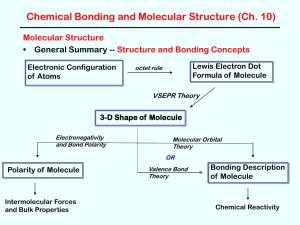
Polarity of Molecules
... subshell, but how can Be bind 2 fluorine atoms when it has no unpaired electrons? According to the VB model a bond results from sharing of unpaired electrons via overlap of AOs… ...
... subshell, but how can Be bind 2 fluorine atoms when it has no unpaired electrons? According to the VB model a bond results from sharing of unpaired electrons via overlap of AOs… ...
3-D Shape of Molecule
... 3. MO's for 2nd Row Diatomic Molecules (e.g., N2, O2, F2, etc.) AO combinations -- from s orbitals and from p orbitals MO energy level diagram -- Page 467 ...
... 3. MO's for 2nd Row Diatomic Molecules (e.g., N2, O2, F2, etc.) AO combinations -- from s orbitals and from p orbitals MO energy level diagram -- Page 467 ...
Carbene Singlets, Triplets, and the Physics that
... while knowing a set of rules for writing them allows them to be consistent with experiments in cases where there are no extremely similar energy levels in the interaction diagram, they are not suitable for discerning numerical results. However, as will be shown in more detail later, these molecular ...
... while knowing a set of rules for writing them allows them to be consistent with experiments in cases where there are no extremely similar energy levels in the interaction diagram, they are not suitable for discerning numerical results. However, as will be shown in more detail later, these molecular ...
Lectures 6-7 - U of L Class Index
... where Δx is the uncertainty about position, Δp is the uncertainty about momentum (i.e. difference between maximum and minimum possible momentum values), and h is Planck’s constant. Scientists often use ħ to stand for h/2, so this formula can also be written as: ...
... where Δx is the uncertainty about position, Δp is the uncertainty about momentum (i.e. difference between maximum and minimum possible momentum values), and h is Planck’s constant. Scientists often use ħ to stand for h/2, so this formula can also be written as: ...
Lectures 6-7
... For four of the d orbitals, both of these nodes are planes, giving a ‘petal-shaped’ orbital. For the fifth d orbital (_____),the nodes look more like a pair of inverted cones. This gives an orbital that looks a bit like a p orbital with a doughnut around it. (Note the phases, though; they are differ ...
... For four of the d orbitals, both of these nodes are planes, giving a ‘petal-shaped’ orbital. For the fifth d orbital (_____),the nodes look more like a pair of inverted cones. This gives an orbital that looks a bit like a p orbital with a doughnut around it. (Note the phases, though; they are differ ...
Resonance States of Atomic Anions
... a common belief that due to a strong correlation effect among several equivalent electrons the underlying physics is much more complicated than a simple model could suggest, and full-scale calcu- ...
... a common belief that due to a strong correlation effect among several equivalent electrons the underlying physics is much more complicated than a simple model could suggest, and full-scale calcu- ...
Lectures 10-11 - U of L Class Index
... For four of the d orbitals, both of these nodes are planes, giving a ‘petal-shaped’ orbital. For the fifth d orbital (_____),the nodes look more like a pair of inverted cones. This gives an orbital that looks a bit like a p orbital with a doughnut around it. (Note the phases, though; they are differ ...
... For four of the d orbitals, both of these nodes are planes, giving a ‘petal-shaped’ orbital. For the fifth d orbital (_____),the nodes look more like a pair of inverted cones. This gives an orbital that looks a bit like a p orbital with a doughnut around it. (Note the phases, though; they are differ ...
Lectures 10-11
... For four of the d orbitals, both of these nodes are planes, giving a ‘petal-shaped’ orbital. For the fifth d orbital (_____),the nodes look more like a pair of inverted cones. This gives an orbital that looks a bit like a p orbital with a doughnut around it. (Note the phases, though; they are differ ...
... For four of the d orbitals, both of these nodes are planes, giving a ‘petal-shaped’ orbital. For the fifth d orbital (_____),the nodes look more like a pair of inverted cones. This gives an orbital that looks a bit like a p orbital with a doughnut around it. (Note the phases, though; they are differ ...
Lectures 10-11
... For four of the d orbitals, both of these nodes are planes, giving a ‘petal-shaped’ orbital. For the fifth d orbital (_____),the nodes look more like a pair of inverted cones. This gives an orbital that looks a bit like a p orbital with a doughnut around it. (Note the phases, though; they are differ ...
... For four of the d orbitals, both of these nodes are planes, giving a ‘petal-shaped’ orbital. For the fifth d orbital (_____),the nodes look more like a pair of inverted cones. This gives an orbital that looks a bit like a p orbital with a doughnut around it. (Note the phases, though; they are differ ...
Lectures 6-7
... For four of the d orbitals, both of these nodes are planes, giving a ‘petal-shaped’ orbital. For the fifth d orbital (_____),the nodes look more like a pair of inverted cones. This gives an orbital that looks a bit like a p orbital with a doughnut around it. (Note the phases, though; they are differ ...
... For four of the d orbitals, both of these nodes are planes, giving a ‘petal-shaped’ orbital. For the fifth d orbital (_____),the nodes look more like a pair of inverted cones. This gives an orbital that looks a bit like a p orbital with a doughnut around it. (Note the phases, though; they are differ ...
CHEMICAL BONDING
... Atomic Spectra and Bohr Energy of quantized state = - C/n2 • Only orbits where n = integral no. are permitted. • Radius of allowed orbitals = n2 • (0.0529 nm) • But note — same eqns. come from modern wave mechanics approach. • Results can be used to explain atomic spectra. ...
... Atomic Spectra and Bohr Energy of quantized state = - C/n2 • Only orbits where n = integral no. are permitted. • Radius of allowed orbitals = n2 • (0.0529 nm) • But note — same eqns. come from modern wave mechanics approach. • Results can be used to explain atomic spectra. ...
molecular modeling and electronic structure calculations
... first principles”) calculations solve the Schrödinger equation using the exact computational expression for the energy of the electrons.1 The particular ab initio method that we will use for this lab is called HartreeFock (HF). HF uses an approximate wavefunction to solve Schrödinger, so the resulti ...
... first principles”) calculations solve the Schrödinger equation using the exact computational expression for the energy of the electrons.1 The particular ab initio method that we will use for this lab is called HartreeFock (HF). HF uses an approximate wavefunction to solve Schrödinger, so the resulti ...
Lecture 1 Atomic Structure
... • Nodal plane – the plane cutting through a nucleus, separating the region of + and – sign of the wavefunction. (Note the bright and dark lope which is opposite to each other.) • No electrons at nodal plane • There are three ml values (-1, 0, and +1) for p-orbitals, representing px, py and pz. I wil ...
... • Nodal plane – the plane cutting through a nucleus, separating the region of + and – sign of the wavefunction. (Note the bright and dark lope which is opposite to each other.) • No electrons at nodal plane • There are three ml values (-1, 0, and +1) for p-orbitals, representing px, py and pz. I wil ...
Band Theories
... of a problem, the expectation value of the energy calculated from this function will always be HIGHER than the true energy of the ground state. ...
... of a problem, the expectation value of the energy calculated from this function will always be HIGHER than the true energy of the ground state. ...
The Use of Fock Spaces in Quantum Mechanics
... natural tool for quantum field theory. This mathematical construction is used to construct the quantum states of a multi-particle system from a single particle system. The creation and annihilation operators are used to account for the introduction and removal of particles, allowing us to describe a ...
... natural tool for quantum field theory. This mathematical construction is used to construct the quantum states of a multi-particle system from a single particle system. The creation and annihilation operators are used to account for the introduction and removal of particles, allowing us to describe a ...
Homework 2 - UCSB Physics
... 2. Orbitals with cubic symmetry: Consider the effect of cubic crystal fields on the fivefold degenerate d orbitals. The single particle potential on an electron, projected into this quintuplet, can in general be expressed as a function of the 3 orbital angular momentum ~ ·L ~ = `(` + 1) with ` = 2. ...
... 2. Orbitals with cubic symmetry: Consider the effect of cubic crystal fields on the fivefold degenerate d orbitals. The single particle potential on an electron, projected into this quintuplet, can in general be expressed as a function of the 3 orbital angular momentum ~ ·L ~ = `(` + 1) with ` = 2. ...
On the leading energy correction for the statistical model of the atom
... Nm , ϕ can be understood as a coordinate transformation. One can easily see that these wave functions are automatically orthogonal to each other in the new coordinates thus, fulfilling the Pauli principle. By explicitly specifying the wave functions, one also ensures the quantum-mechanical foundatio ...
... Nm , ϕ can be understood as a coordinate transformation. One can easily see that these wave functions are automatically orthogonal to each other in the new coordinates thus, fulfilling the Pauli principle. By explicitly specifying the wave functions, one also ensures the quantum-mechanical foundatio ...
Detailed Notes CH. 6
... Line spectra of many electron atoms show each line as a closely spaced pair of lines. Stern and Gerlach designed an experiment to determine why. • A beam of atoms was passed through a slit and into a magnetic field and the atoms were then detected. • Two spots were found: one with the electrons spin ...
... Line spectra of many electron atoms show each line as a closely spaced pair of lines. Stern and Gerlach designed an experiment to determine why. • A beam of atoms was passed through a slit and into a magnetic field and the atoms were then detected. • Two spots were found: one with the electrons spin ...
An evolutionary algorithm to calculate the ground state of a quantum
... Before describing our approach we present rst a brief description of the GA. As we have mentioned before, the GA was developed to optimize (maximize or minimize) a given property, like an area, a volume or an energy. The property in question is a function of many variables of the system. In GA-lang ...
... Before describing our approach we present rst a brief description of the GA. As we have mentioned before, the GA was developed to optimize (maximize or minimize) a given property, like an area, a volume or an energy. The property in question is a function of many variables of the system. In GA-lang ...
C1403_Lecture9_10110..
... The energy of an orbital of a hydrogen atom or any one electron atom only depends on the value of n shell = all orbitals with the same value of n subshell = all orbitals with the same value of n and l an orbital is fully defined by three quantum numbers, n, l, and ml Each shell of QN = n contains n ...
... The energy of an orbital of a hydrogen atom or any one electron atom only depends on the value of n shell = all orbitals with the same value of n subshell = all orbitals with the same value of n and l an orbital is fully defined by three quantum numbers, n, l, and ml Each shell of QN = n contains n ...
Chapter 13 Review
... Bohr Model was the first model that showed the electrons located in ________ ________. energy levels ...
... Bohr Model was the first model that showed the electrons located in ________ ________. energy levels ...
Chapter1011
... • Polar or Non-Polar? – In very symmetrical structures (e.g. CO2 or CF4), the individual bond dipoles effectively cancel each other and the molecule is ...
... • Polar or Non-Polar? – In very symmetrical structures (e.g. CO2 or CF4), the individual bond dipoles effectively cancel each other and the molecule is ...
Atomic Orbitals
... Valence electrons – electrons in the outermost (highest) principal energy level of an atom Core electrons – inner electrons Elements with the same valence electron arrangement show very similar chemical behavior. ...
... Valence electrons – electrons in the outermost (highest) principal energy level of an atom Core electrons – inner electrons Elements with the same valence electron arrangement show very similar chemical behavior. ...
elmszerv3
... For CH4, the four C - H bonds { YI }, may also be formed from the four sp3 hybrid AO {i} and the 1s orbitals { Hi } of the four hydrogen atoms. 1 = a1 + bH1 2 = a2 + bH2 3 = a3 + bH3 4 = a4 + bH4 The linear combination of carbon HAO and hydrogen AO to form methane LMO. ...
... For CH4, the four C - H bonds { YI }, may also be formed from the four sp3 hybrid AO {i} and the 1s orbitals { Hi } of the four hydrogen atoms. 1 = a1 + bH1 2 = a2 + bH2 3 = a3 + bH3 4 = a4 + bH4 The linear combination of carbon HAO and hydrogen AO to form methane LMO. ...
Lecture 2 - Tufts University
... • A plane wave in two or three dimensions is like a sine wave in one dimension except that crests and troughs aren't points, but form lines (2D) or planes (3-D) perpendicular to the direction of wave propagation. The Figure shows a plane sine wave in two dimensions. The large arrow is a vector calle ...
... • A plane wave in two or three dimensions is like a sine wave in one dimension except that crests and troughs aren't points, but form lines (2D) or planes (3-D) perpendicular to the direction of wave propagation. The Figure shows a plane sine wave in two dimensions. The large arrow is a vector calle ...