Chem expo 12
... weighted in terms of assessment to ensure that they have been successfully met. School-assessed coursework in Unit 3 contributes 17% of the total study score and the mid-year examination in June contributes a further 33%. This split is repeated in the second half of the year, with a final examinatio ...
... weighted in terms of assessment to ensure that they have been successfully met. School-assessed coursework in Unit 3 contributes 17% of the total study score and the mid-year examination in June contributes a further 33%. This split is repeated in the second half of the year, with a final examinatio ...
Reflection of a Ray of Light Introduction: Purpose
... __ 3. Find the midpoint of this line. Label it the point of incidence. __ 4. Draw a dotted line perpendicular to the reflecting surface at the point of incidence. The line should go from the point of incidence past the edge of the protractor at the 90o mark. Label this line the normal. (place ...
... __ 3. Find the midpoint of this line. Label it the point of incidence. __ 4. Draw a dotted line perpendicular to the reflecting surface at the point of incidence. The line should go from the point of incidence past the edge of the protractor at the 90o mark. Label this line the normal. (place ...
Efficient generation of a maximally entangled state by
... In fact, various interesting and peculiar phenomena are predicted on the basis of highly nonclassical states, and entanglement plays a key role in quantum information protocols [1]. They all rely on the generation of nontrivial states and are not realized without establishing strategies for the prep ...
... In fact, various interesting and peculiar phenomena are predicted on the basis of highly nonclassical states, and entanglement plays a key role in quantum information protocols [1]. They all rely on the generation of nontrivial states and are not realized without establishing strategies for the prep ...
Dielectric and thermodynamic response of a
... previously analyzed the orientational structure (Kirkwood factor Gk) of SPC water using both the conventional dipolar reaction field3 and the Ewald sum12 methods to treat the long range interactions. The orientational correlation functions provide a sensitive test of the effect of the long range int ...
... previously analyzed the orientational structure (Kirkwood factor Gk) of SPC water using both the conventional dipolar reaction field3 and the Ewald sum12 methods to treat the long range interactions. The orientational correlation functions provide a sensitive test of the effect of the long range int ...
PERCENT COMPOSITION of A HYDRATE LAB
... When many ionic compounds are crystallized from a water solution, they include individual water molecules as part of their crystalline structure. Such crystals are known as hydrates. If the substances are heated, this water of crystallization may be driven off and leaves behind the pure anhydrous cr ...
... When many ionic compounds are crystallized from a water solution, they include individual water molecules as part of their crystalline structure. Such crystals are known as hydrates. If the substances are heated, this water of crystallization may be driven off and leaves behind the pure anhydrous cr ...
Energy Efficiency trends and policies in the - ODYSSEE-MURE
... The total primary energy consumption has increased until 2007, while the final consumption remained more or less stable between 2000 and 2012 (see figure 1.2). The primary energy intensity (primary energy consumption per unit of GDP) has increased up to 2005 and then decreased. Final energy intensit ...
... The total primary energy consumption has increased until 2007, while the final consumption remained more or less stable between 2000 and 2012 (see figure 1.2). The primary energy intensity (primary energy consumption per unit of GDP) has increased up to 2005 and then decreased. Final energy intensit ...
NIckel AdsorptIoN oNto cArboN ANode dust modIfIed by AcetIc AcId
... binding affinity to Ni (II) ions [7]. It is biodegradable and widely used in the food industry. In the literature [8] we can see that there is growing interest in alkaline hydroxide activation process and KOH has ...
... binding affinity to Ni (II) ions [7]. It is biodegradable and widely used in the food industry. In the literature [8] we can see that there is growing interest in alkaline hydroxide activation process and KOH has ...
Unit 13 Stoichiometry (Theoretical)
... e. How many moles of Al2O3 are formed when 0.78 moles of O2 reacts with aluminum? (Ans. 0.52 mol Al2O3) f. How many grams of aluminum are required to produce 98.6 grams of aluminum oxide? ...
... e. How many moles of Al2O3 are formed when 0.78 moles of O2 reacts with aluminum? (Ans. 0.52 mol Al2O3) f. How many grams of aluminum are required to produce 98.6 grams of aluminum oxide? ...
REDOX EQUILIBRIA SL - chemistryatdulwich
... used to keep track of how many electrons are lost or gained by each atom or ion. They are used to identify oxidation and reduction reactions. Rules for working out oxidation number: 1. The oxidation number of an atom in its element form or uncombined form is always 0. For example the atoms in Na, He ...
... used to keep track of how many electrons are lost or gained by each atom or ion. They are used to identify oxidation and reduction reactions. Rules for working out oxidation number: 1. The oxidation number of an atom in its element form or uncombined form is always 0. For example the atoms in Na, He ...
The structure of the energy bands and optical absorption in osmium
... greater than 2 eV, and is manifested in a different width and intensity of the two main absorption peaks, C and D. Over the whole spectral range we studied, h < 5 eV, anisotropy occurs in the reflectivity of osmium, which is largest at a wavelength /Z = 0.823 p m (h= 1.5 eV), where R = 75% while R, ...
... greater than 2 eV, and is manifested in a different width and intensity of the two main absorption peaks, C and D. Over the whole spectral range we studied, h < 5 eV, anisotropy occurs in the reflectivity of osmium, which is largest at a wavelength /Z = 0.823 p m (h= 1.5 eV), where R = 75% while R, ...
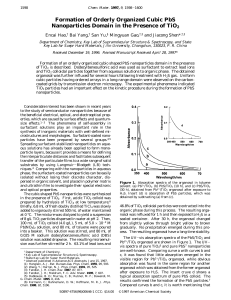
Formation of Orderly Organized Cubic PbS Nanoparticles Domain in
... reactive time was prolonged. This indicated that a slow crystallite growth occurred for a longer reactive time. Samples for transmission electron microscopy (TEM) were prepared as follows. The nanoparticles-containing toluene solution was dropped on the surface of water, which dispersed to be a laye ...
... reactive time was prolonged. This indicated that a slow crystallite growth occurred for a longer reactive time. Samples for transmission electron microscopy (TEM) were prepared as follows. The nanoparticles-containing toluene solution was dropped on the surface of water, which dispersed to be a laye ...
Chemistry 11 – Course Review
... ___________________________ measured the charge/mass ratio of an electron and came up with the so-called “plum pudding” model of the atom. ...
... ___________________________ measured the charge/mass ratio of an electron and came up with the so-called “plum pudding” model of the atom. ...
AP Chemistry - cloudfront.net
... 8.37 Which group in the periodic table has elements with high IE1 and very negative first electron affinities (EA1)? What is the charge on the ions that these atoms form? 8.59 Write the charge and full ground-state electron configuration of the monatomic ion most likely to be formed by each of the f ...
... 8.37 Which group in the periodic table has elements with high IE1 and very negative first electron affinities (EA1)? What is the charge on the ions that these atoms form? 8.59 Write the charge and full ground-state electron configuration of the monatomic ion most likely to be formed by each of the f ...
Chem101, 2nd Major Exam, term061
... The hydrogen atom has only one orbital. The size of the hydrogen 1s orbital is defined as the surface that contains 90% of the total electron probability. C) The square of the wave function represents the probability distribution of the elctron in the orbital. D) In the quantum mechanical model, the ...
... The hydrogen atom has only one orbital. The size of the hydrogen 1s orbital is defined as the surface that contains 90% of the total electron probability. C) The square of the wave function represents the probability distribution of the elctron in the orbital. D) In the quantum mechanical model, the ...
development of SPR sensors
... Low-cost micro-lens array and diffractive grating mirrors for focusing and redirecting light Molding and rapid fabrication techniques for mass production of optical components Sensitive fiber and waveguide-SPR approach Integrated multilayer and distributed Bragg reflector for improved sensitivity Im ...
... Low-cost micro-lens array and diffractive grating mirrors for focusing and redirecting light Molding and rapid fabrication techniques for mass production of optical components Sensitive fiber and waveguide-SPR approach Integrated multilayer and distributed Bragg reflector for improved sensitivity Im ...
Liquid-gas transition of neon in quasi-one
... transition is impossible when we have a finite-ranged interaction, and other authors,22–24 proved that a long-range potential decaying faster than 1/r 2 could not present a phase transition in purely 1D systems. All that means that the liquid-gas critical point seems to be at 0 K, as in the orderdis ...
... transition is impossible when we have a finite-ranged interaction, and other authors,22–24 proved that a long-range potential decaying faster than 1/r 2 could not present a phase transition in purely 1D systems. All that means that the liquid-gas critical point seems to be at 0 K, as in the orderdis ...
www.fahadsacademy.com
... Hydrogen atom has one valency. To become stable with hydrogen atom, it needs one more electron, just like helium which has 2 valency. When 2 hydrogen atoms join, they share their electrons, on which, the share becomes 2 electrons, which is now a noble gas configuration, being shared between these 2 ...
... Hydrogen atom has one valency. To become stable with hydrogen atom, it needs one more electron, just like helium which has 2 valency. When 2 hydrogen atoms join, they share their electrons, on which, the share becomes 2 electrons, which is now a noble gas configuration, being shared between these 2 ...
Rutherford backscattering spectrometry
Rutherford backscattering spectrometry (RBS) is an analytical technique used in materials science. Sometimes referred to as high-energy ion scattering (HEIS) spectrometry, RBS is used to determine the structure and composition of materials by measuring the backscattering of a beam of high energy ions (typically protons or alpha particles) impinging on a sample.