Periodic Table Properties Notes s1
... gained electrons and has a negative or positive charge. • Atoms that lose electrons have a positive charge. Atoms that gain electrons have a negative charge. • What ion an atom will form can be determined by its group on the periodic table. ...
... gained electrons and has a negative or positive charge. • Atoms that lose electrons have a positive charge. Atoms that gain electrons have a negative charge. • What ion an atom will form can be determined by its group on the periodic table. ...
Power Point Chapter 5
... • The periodic table was expanded by one group at the far right of the periodic table with the discovery of argon in 1894. • Helium, neon, krypton, xenon, and radon were subsequently discovered in the next 5 years. • They were originally called the inert gases. ...
... • The periodic table was expanded by one group at the far right of the periodic table with the discovery of argon in 1894. • Helium, neon, krypton, xenon, and radon were subsequently discovered in the next 5 years. • They were originally called the inert gases. ...
Chapter 5
... • The periodic table was expanded by one group at the far right of the periodic table with the discovery of argon in 1894. • Helium, neon, krypton, xenon, and radon were subsequently discovered in the next 5 years. • They were originally called the inert gases. ...
... • The periodic table was expanded by one group at the far right of the periodic table with the discovery of argon in 1894. • Helium, neon, krypton, xenon, and radon were subsequently discovered in the next 5 years. • They were originally called the inert gases. ...
Inorganic Chemistry ELEMENTS AND
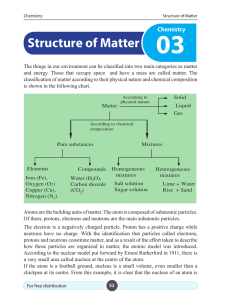
... Chemical Bond : Chemical bond may be defined as the attractive force that binds together the constituent atoms in a molecule. Following are some different types of chemical bonds which usually occur in various molecules. (i) Electrovalent bond (Ionic bond) : This type of bond is formed by transfer o ...
... Chemical Bond : Chemical bond may be defined as the attractive force that binds together the constituent atoms in a molecule. Following are some different types of chemical bonds which usually occur in various molecules. (i) Electrovalent bond (Ionic bond) : This type of bond is formed by transfer o ...
Structure of Matter - e
... carbon. In no other element, the atomic number is equal to 6. The atomic number of an element is symbolised by Z. In a neutral atom, the number of protons is equal to the number of electrons in it. So, it implies that the atomic number of an element is equal to the number of electrons in an atom of ...
... carbon. In no other element, the atomic number is equal to 6. The atomic number of an element is symbolised by Z. In a neutral atom, the number of protons is equal to the number of electrons in it. So, it implies that the atomic number of an element is equal to the number of electrons in an atom of ...
c1l2ch06
... nonmetals, metal atoms tend to lose electrons, and nonmetal atoms tend to gain electrons. The transfer has a predictable effect on the size of the ions that form. ...
... nonmetals, metal atoms tend to lose electrons, and nonmetal atoms tend to gain electrons. The transfer has a predictable effect on the size of the ions that form. ...
ch 6 ppt - Madison County Schools
... • Ionization energies generally increase from left to right across a period, and decrease as you move down a group. • The octet rule states that atoms gain, lose, or share electrons to acquire a full set of eight valence electrons. • Electronegativity generally increases from left to right across a ...
... • Ionization energies generally increase from left to right across a period, and decrease as you move down a group. • The octet rule states that atoms gain, lose, or share electrons to acquire a full set of eight valence electrons. • Electronegativity generally increases from left to right across a ...
Document
... • Ionization energies generally increase from left to right across a period, and decrease as you move down a group. • The octet rule states that atoms gain, lose, or share electrons to acquire a full set of eight valence electrons. • Electronegativity generally increases from left to right across a ...
... • Ionization energies generally increase from left to right across a period, and decrease as you move down a group. • The octet rule states that atoms gain, lose, or share electrons to acquire a full set of eight valence electrons. • Electronegativity generally increases from left to right across a ...
Chapter 6 PP
... • Ionization energies generally increase from left to right across a period, and decrease as you move down a group. • The octet rule states that atoms gain, lose, or share electrons to acquire a full set of eight valence electrons. • Electronegativity generally increases from left to right across a ...
... • Ionization energies generally increase from left to right across a period, and decrease as you move down a group. • The octet rule states that atoms gain, lose, or share electrons to acquire a full set of eight valence electrons. • Electronegativity generally increases from left to right across a ...
D. - Telluride Middle/High School
... • Ionization energies generally increase from left to right across a period, and decrease as you move down a group. • The octet rule states that atoms gain, lose, or share electrons to acquire a full set of eight valence electrons. • Electronegativity generally increases from left to right across a ...
... • Ionization energies generally increase from left to right across a period, and decrease as you move down a group. • The octet rule states that atoms gain, lose, or share electrons to acquire a full set of eight valence electrons. • Electronegativity generally increases from left to right across a ...
Document
... Groups 18 Noble Gases – Not reactive – do not form ions – Filled, stable valence shell (8 electrons except He which has 2) ...
... Groups 18 Noble Gases – Not reactive – do not form ions – Filled, stable valence shell (8 electrons except He which has 2) ...
Describe the Periodic Table
... History of the Periodic Table • In 1869, Dmitri Mendeleev listed all the elements known at the time in order of their atomic weight. He arranged the list into a table of rows and columns. • In Mendeleev’s day, there were 63 known elements. Today, we know of almost 100 elements that can be found in ...
... History of the Periodic Table • In 1869, Dmitri Mendeleev listed all the elements known at the time in order of their atomic weight. He arranged the list into a table of rows and columns. • In Mendeleev’s day, there were 63 known elements. Today, we know of almost 100 elements that can be found in ...
powerpoint
... You ask such intelligent questions my friend! The periodic table is one of the most important tools you will need next year when you take chemistry.It was designed by a Russian chemist in 1869 by the name of Dimitri Mendeleev. The periodic table arranges all the elements in columns and rows in order ...
... You ask such intelligent questions my friend! The periodic table is one of the most important tools you will need next year when you take chemistry.It was designed by a Russian chemist in 1869 by the name of Dimitri Mendeleev. The periodic table arranges all the elements in columns and rows in order ...
5.3 Representative Groups PPT
... n Relate the number of valence electrons to groups in the periodic table and to properties of elements in those groups. n Predict the reactivity of some elements based on their locations within a group. n Identify some properties of common A group elements. ...
... n Relate the number of valence electrons to groups in the periodic table and to properties of elements in those groups. n Predict the reactivity of some elements based on their locations within a group. n Identify some properties of common A group elements. ...
THE PERIODIC TABLE and PERIODIC LAW
... 1. Definition---a vertical column of elements on the Periodic Table --also called a “Family” 2. Valence electrons and group number---each element within a group has the same number of valence electrons --In general, the group number is equal to the number of valence electrons --For group numbers gre ...
... 1. Definition---a vertical column of elements on the Periodic Table --also called a “Family” 2. Valence electrons and group number---each element within a group has the same number of valence electrons --In general, the group number is equal to the number of valence electrons --For group numbers gre ...
Mendeleef`s Periodic Table
... be discovered. e.g., he left spaces for Ga and Ge and named these elements as Ekaaluminium (Ga) and EKa-silicon (Ge)respectively 3. Atomic mass correction of doubtful elements on the basis of their expected positions and properties. Defects in the Mendeleef’s Periodic Table (i) Position of hydrogen ...
... be discovered. e.g., he left spaces for Ga and Ge and named these elements as Ekaaluminium (Ga) and EKa-silicon (Ge)respectively 3. Atomic mass correction of doubtful elements on the basis of their expected positions and properties. Defects in the Mendeleef’s Periodic Table (i) Position of hydrogen ...
Study Guide for Electrons Mini-Test - seys
... - important for modern technology - tungsten = in tiny coil of wire inside incandescent light bulbs - platinum = in the catalytic converters that reduce pollution from automobile engines - alloys = for many applications, two or more metals are combined to form alloys - can be stronger, less likely t ...
... - important for modern technology - tungsten = in tiny coil of wire inside incandescent light bulbs - platinum = in the catalytic converters that reduce pollution from automobile engines - alloys = for many applications, two or more metals are combined to form alloys - can be stronger, less likely t ...
POGIL: Periodic Table Trends
... b. Why does Hydrogen fit into this group? c. Why does Hydrogen NOT fit into this group? (Hint: Why does it make sense that European Periodic Tables show H in both Group 1 and 17?) ...
... b. Why does Hydrogen fit into this group? c. Why does Hydrogen NOT fit into this group? (Hint: Why does it make sense that European Periodic Tables show H in both Group 1 and 17?) ...
Review of Periodic Trends
... upper left hand corner of the periodic table lower left hand corner of the periodic table lower right hand corner of the periodic table upper right hand corner of the periodic table ...
... upper left hand corner of the periodic table lower left hand corner of the periodic table lower right hand corner of the periodic table upper right hand corner of the periodic table ...
Periodic Table Trends - Magoffin County Schools
... • Another trend that can easily be seen in the periodic table is that of ELECTRONEGATIVITY. • Simply put, Eneg is a measure of an atom’s ability to chemically bond (react) with other atoms. • For Eneg, each atom is assigned a number value from 0 (weakest) to 4 (strongest) . • The closer to 4, the mo ...
... • Another trend that can easily be seen in the periodic table is that of ELECTRONEGATIVITY. • Simply put, Eneg is a measure of an atom’s ability to chemically bond (react) with other atoms. • For Eneg, each atom is assigned a number value from 0 (weakest) to 4 (strongest) . • The closer to 4, the mo ...
Unit 3 Notes: Periodic Table Notes
... He noticed that both the chemical and physical properties repeated every 8 elements and called this the ____Law of Octaves ___________. In 1869 both Lothar Meyer and Dmitri Mendeleev showed a connection between atomic mass and an element’s properties. Mendeleev published first, and is given cr ...
... He noticed that both the chemical and physical properties repeated every 8 elements and called this the ____Law of Octaves ___________. In 1869 both Lothar Meyer and Dmitri Mendeleev showed a connection between atomic mass and an element’s properties. Mendeleev published first, and is given cr ...
FSN 1500 Week 7 - Oakland Community College
... Organic Compounds Organic compounds - carbon-containing compounds where carbon forms the structural framework of the molecule Remember: to our knowledge all life on Earth is organic compound based! Millions of organic compounds exist; organic compounds are over 10X more abundant than all inorga ...
... Organic Compounds Organic compounds - carbon-containing compounds where carbon forms the structural framework of the molecule Remember: to our knowledge all life on Earth is organic compound based! Millions of organic compounds exist; organic compounds are over 10X more abundant than all inorga ...
ElectronConfigurationSE
... _________________________________________________________________________ 7. Predict: On the ATOMIC RADIUS tab click Clear. Select the PERIODIC TABLE tab. Elements in the same column of the periodic table are called chemical families, or groups. How do you think the size of atoms will change from to ...
... _________________________________________________________________________ 7. Predict: On the ATOMIC RADIUS tab click Clear. Select the PERIODIC TABLE tab. Elements in the same column of the periodic table are called chemical families, or groups. How do you think the size of atoms will change from to ...
CBSE Class 10 Physics Periodic classification of elements Notes
... note. For example cobalt and nickel are in the same slot and these are placed in the same column as fluorine, chlorine and bromine which have very different properties than these elements. Iron, which resembles cobalt and nickel in properties, has been placed differently away from these elements 3. ...
... note. For example cobalt and nickel are in the same slot and these are placed in the same column as fluorine, chlorine and bromine which have very different properties than these elements. Iron, which resembles cobalt and nickel in properties, has been placed differently away from these elements 3. ...
Noble gas

The noble gases make a group of chemical elements with similar properties. Under standard conditions, they are all odorless, colorless, monatomic gases with very low chemical reactivity. The six noble gases that occur naturally are helium (He), neon (Ne), argon (Ar), krypton (Kr), xenon (Xe), and the radioactive radon (Rn).For the first six periods of the periodic table, the noble gases are exactly the members of group 18 of the periodic table.It is possible that due to relativistic effects, the group 14 element flerovium exhibits some noble-gas-like properties, instead of the group 18 element ununoctium. Noble gases are typically highly unreactive except when under particular extreme conditions. The inertness of noble gases makes them very suitable in applications where reactions are not wanted. For example: argon is used in lightbulbs to prevent the hot tungsten filament from oxidizing; also, helium is breathed by deep-sea divers to prevent oxygen and nitrogen toxicity.The properties of the noble gases can be well explained by modern theories of atomic structure: their outer shell of valence electrons is considered to be ""full"", giving them little tendency to participate in chemical reactions, and it has been possible to prepare only a few hundred noble gas compounds. The melting and boiling points for a given noble gas are close together, differing by less than 10 °C (18 °F); that is, they are liquids over only a small temperature range.Neon, argon, krypton, and xenon are obtained from air in an air separation unit using the methods of liquefaction of gases and fractional distillation. Helium is sourced from natural gas fields which have high concentrations of helium in the natural gas, using cryogenic gas separation techniques, and radon is usually isolated from the radioactive decay of dissolved radium, thorium, or uranium compounds (since those compounds give off alpha particles). Noble gases have several important applications in industries such as lighting, welding, and space exploration. A helium-oxygen breathing gas is often used by deep-sea divers at depths of seawater over 55 m (180 ft) to keep the diver from experiencing oxygen toxemia, the lethal effect of high-pressure oxygen, and nitrogen narcosis, the distracting narcotic effect of the nitrogen in air beyond this partial-pressure threshold. After the risks caused by the flammability of hydrogen became apparent, it was replaced with helium in blimps and balloons.