Learning Guide 3
... 24. Group 18 elements were once called the inert gas group because they do not react with other elements, due to a gull valance shell of 8 electrons. They are now called noble gases. 25. The trend in BP going down group 18 is increasing (look it up on ref. table S: He BP = 4K Rn Bp =211 K ...
... 24. Group 18 elements were once called the inert gas group because they do not react with other elements, due to a gull valance shell of 8 electrons. They are now called noble gases. 25. The trend in BP going down group 18 is increasing (look it up on ref. table S: He BP = 4K Rn Bp =211 K ...
Chapt 6: Arrangement of the Elements
... The Noble Gases • The periodic table was expanded by one group at the far right of the periodic table with the discovery of argon in 1894. • Helium, neon, krypton, xenon, and radon were subsequently discovered in the next 5 years. • They were originally called the inert gases. • Recently, several co ...
... The Noble Gases • The periodic table was expanded by one group at the far right of the periodic table with the discovery of argon in 1894. • Helium, neon, krypton, xenon, and radon were subsequently discovered in the next 5 years. • They were originally called the inert gases. • Recently, several co ...
18HYD13_F_Layout 1
... position in the periodic table, for example, Berelium. Limitations: 1. No fix position was given to hydro -gen in the Mendeleev’s periodic table. 2. Position of isotopes of all elements was not certain according to his periodic table. 3. Atomic masses did not increase in a regular manner is going fr ...
... position in the periodic table, for example, Berelium. Limitations: 1. No fix position was given to hydro -gen in the Mendeleev’s periodic table. 2. Position of isotopes of all elements was not certain according to his periodic table. 3. Atomic masses did not increase in a regular manner is going fr ...
The Periodic Table and Periodic Law
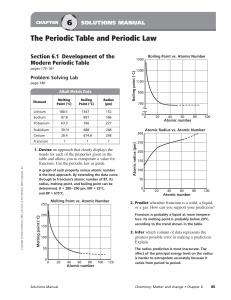
... electrons pulls the valence electrons (being added to the same energy level) closer to the nucleus. ...
... electrons pulls the valence electrons (being added to the same energy level) closer to the nucleus. ...
alkali metal
... This diagram compares the relative sizes of atoms and ions for selected alkali metals and halogens. The data are given in picometers. Comparing and Contrasting: ◦ What happens to the radius when an atom forms a cation? ...
... This diagram compares the relative sizes of atoms and ions for selected alkali metals and halogens. The data are given in picometers. Comparing and Contrasting: ◦ What happens to the radius when an atom forms a cation? ...
Revising the Periodic Table
... The Periodic Table is one of the central organising themes in chemistry, and one of the areas where ideas about the macroscopic (ie properties of elements) and molecular (eg electronic configuration) properties are related (and often confused) – see Chapter 6 of the Teachers’ notes. This exercise is ...
... The Periodic Table is one of the central organising themes in chemistry, and one of the areas where ideas about the macroscopic (ie properties of elements) and molecular (eg electronic configuration) properties are related (and often confused) – see Chapter 6 of the Teachers’ notes. This exercise is ...
Chapter 2 - UBC Physics
... of helium ions, He+2 , and the β-rays were later determined to be a stream of high-speed electrons. The α-rays were clear evidence that uranium atoms had disintegrated to give off helium. No longer could atoms be considered uncharged particles. Geiger and Marsden sent the He+2 ions into thin metal f ...
... of helium ions, He+2 , and the β-rays were later determined to be a stream of high-speed electrons. The α-rays were clear evidence that uranium atoms had disintegrated to give off helium. No longer could atoms be considered uncharged particles. Geiger and Marsden sent the He+2 ions into thin metal f ...
The Periodic Table and Periodic Law
... Metalloids have physical and chemical properties of both metals and non-metals, such as silicon and germanium. They are found along the stair step of the table starting with Boron ...
... Metalloids have physical and chemical properties of both metals and non-metals, such as silicon and germanium. They are found along the stair step of the table starting with Boron ...
Periodic Table of Elements
... Some groups have specific names like the halogens or noble gases. Elements within the same period or group have similar properties. ...
... Some groups have specific names like the halogens or noble gases. Elements within the same period or group have similar properties. ...
Study Material - Tiwariacademy.net
... Mendeleev's periodic law :– The properties of elements are the periodic function of their atomic mass. Mendeleev's periodic table based on the chemical properties of elements. Contain eight vertical columns called groups and seven horizontal rows called periods form Mendeleev’s peridic table. Achiev ...
... Mendeleev's periodic law :– The properties of elements are the periodic function of their atomic mass. Mendeleev's periodic table based on the chemical properties of elements. Contain eight vertical columns called groups and seven horizontal rows called periods form Mendeleev’s peridic table. Achiev ...
Unit 3 Notes: Periodic Table Notes
... • He noticed that both the chemical and physical properties repeated every 8 elements and called this the ____Law of Octaves ___________. • In 1869 both Lothar Meyer and Dmitri Mendeleev showed a connection between atomic mass and an element’s properties. • Mendeleev published first, and is given cr ...
... • He noticed that both the chemical and physical properties repeated every 8 elements and called this the ____Law of Octaves ___________. • In 1869 both Lothar Meyer and Dmitri Mendeleev showed a connection between atomic mass and an element’s properties. • Mendeleev published first, and is given cr ...
Station 1: The Periodic Table 1a. Students know how to relate the
... Ionization energy increases from left to right and decreases from top to bottom. Inonization energy (IE) is the energy required to remove an electron. o IE increases from left to right because the elements increase in nuclear charge in this direction. o IE decreases from top to bottom because as ...
... Ionization energy increases from left to right and decreases from top to bottom. Inonization energy (IE) is the energy required to remove an electron. o IE increases from left to right because the elements increase in nuclear charge in this direction. o IE decreases from top to bottom because as ...
Oct 17-Oct 21
... I can list the three major subatomic particles and distinguish among their location, charges, and relative masses. ...
... I can list the three major subatomic particles and distinguish among their location, charges, and relative masses. ...
synopsis - Mindfiesta
... The p-Block Elements : The p-Block Elements comprise those belonging to group 13 to 18 and these together with the s- Block Elements are called the Representative Elements or Main Group Elements. The d-Block Elements (Transition Elements) : These are the elements of Group 3 to 12 in the centre of t ...
... The p-Block Elements : The p-Block Elements comprise those belonging to group 13 to 18 and these together with the s- Block Elements are called the Representative Elements or Main Group Elements. The d-Block Elements (Transition Elements) : These are the elements of Group 3 to 12 in the centre of t ...
Chapter Twelve: Atoms and the Periodic Table
... The first periodic table • Russian scientist Demitri Mendeleev discovered a set of patterns that applied to all the elements. • Mendeleev noticed a pattern of properties when he arranged the elements in order of increasing atomic mass. • Mendeleev used the melting point, density, color, atomic mass ...
... The first periodic table • Russian scientist Demitri Mendeleev discovered a set of patterns that applied to all the elements. • Mendeleev noticed a pattern of properties when he arranged the elements in order of increasing atomic mass. • Mendeleev used the melting point, density, color, atomic mass ...
Periodic Table Virtual Activity http://my.uzinggo.com/cplogin/ The
... 1. What is the first element of the periodic table ad why? ...
... 1. What is the first element of the periodic table ad why? ...
Mendeleev`s Periodic Table - Scotch Plains
... Nonmetals: to the right of the zig zag line most active nonmetal = F (top right, group 17, not 18) nonmetals have high ionization energies, have high electronegativities, gain electrons, form negative ions when with metals, share electrons with nonmetals, lack luster, are brittle, are poor conductor ...
... Nonmetals: to the right of the zig zag line most active nonmetal = F (top right, group 17, not 18) nonmetals have high ionization energies, have high electronegativities, gain electrons, form negative ions when with metals, share electrons with nonmetals, lack luster, are brittle, are poor conductor ...
Chapter 5 The Periodic Table Section 1 Organizing the Elements
... Changing the Arrangement > How are elements arranged in the modern periodic table? > The modern periodic table organizes elements by atomic number. When the elements are arranged in this way, elements that have similar properties appear at regular intervals. • As scientists learned more about the st ...
... Changing the Arrangement > How are elements arranged in the modern periodic table? > The modern periodic table organizes elements by atomic number. When the elements are arranged in this way, elements that have similar properties appear at regular intervals. • As scientists learned more about the st ...
Noble gas

The noble gases make a group of chemical elements with similar properties. Under standard conditions, they are all odorless, colorless, monatomic gases with very low chemical reactivity. The six noble gases that occur naturally are helium (He), neon (Ne), argon (Ar), krypton (Kr), xenon (Xe), and the radioactive radon (Rn).For the first six periods of the periodic table, the noble gases are exactly the members of group 18 of the periodic table.It is possible that due to relativistic effects, the group 14 element flerovium exhibits some noble-gas-like properties, instead of the group 18 element ununoctium. Noble gases are typically highly unreactive except when under particular extreme conditions. The inertness of noble gases makes them very suitable in applications where reactions are not wanted. For example: argon is used in lightbulbs to prevent the hot tungsten filament from oxidizing; also, helium is breathed by deep-sea divers to prevent oxygen and nitrogen toxicity.The properties of the noble gases can be well explained by modern theories of atomic structure: their outer shell of valence electrons is considered to be ""full"", giving them little tendency to participate in chemical reactions, and it has been possible to prepare only a few hundred noble gas compounds. The melting and boiling points for a given noble gas are close together, differing by less than 10 °C (18 °F); that is, they are liquids over only a small temperature range.Neon, argon, krypton, and xenon are obtained from air in an air separation unit using the methods of liquefaction of gases and fractional distillation. Helium is sourced from natural gas fields which have high concentrations of helium in the natural gas, using cryogenic gas separation techniques, and radon is usually isolated from the radioactive decay of dissolved radium, thorium, or uranium compounds (since those compounds give off alpha particles). Noble gases have several important applications in industries such as lighting, welding, and space exploration. A helium-oxygen breathing gas is often used by deep-sea divers at depths of seawater over 55 m (180 ft) to keep the diver from experiencing oxygen toxemia, the lethal effect of high-pressure oxygen, and nitrogen narcosis, the distracting narcotic effect of the nitrogen in air beyond this partial-pressure threshold. After the risks caused by the flammability of hydrogen became apparent, it was replaced with helium in blimps and balloons.