Periodic Properties of the Elements
... 3. Record the element’s properties from the list provided in part 1 of Table 1. 4. Note that you can select various periodic trends for the element and period. These trends appear as graphs on the right side of the screen. 5. Observe the graph for atomic radius (pm). Describe the relationship betwee ...
... 3. Record the element’s properties from the list provided in part 1 of Table 1. 4. Note that you can select various periodic trends for the element and period. These trends appear as graphs on the right side of the screen. 5. Observe the graph for atomic radius (pm). Describe the relationship betwee ...
Periodic Trends Studyguide with Questions and Answers
... The following information can be found in the box of each element. LOOK on the Periodic Table for these two elements: Oxygen and Gold NOTE all the information you can get from the box of each element. ...
... The following information can be found in the box of each element. LOOK on the Periodic Table for these two elements: Oxygen and Gold NOTE all the information you can get from the box of each element. ...
Student Exploration Sheet: Growing Plants
... _________________________________________________________________________ 7. Predict: On the ATOMIC RADIUS tab click Clear. Select the PERIODIC TABLE tab. Elements in the same column of the periodic table are called chemical families, or groups. How do you think the size of atoms will change from to ...
... _________________________________________________________________________ 7. Predict: On the ATOMIC RADIUS tab click Clear. Select the PERIODIC TABLE tab. Elements in the same column of the periodic table are called chemical families, or groups. How do you think the size of atoms will change from to ...
Record: 1 THE EVOLUTION OF THE PERIODIC SYSTEM Page 1 of
... Instead Mendeleev's ability to accommodate the already known elements may have contributed as much to the acceptance of the periodic system as did his striking predictions. Although numerous scientists helped to develop the periodic system, Mendeleev receives most of the credit for discovering chemi ...
... Instead Mendeleev's ability to accommodate the already known elements may have contributed as much to the acceptance of the periodic system as did his striking predictions. Although numerous scientists helped to develop the periodic system, Mendeleev receives most of the credit for discovering chemi ...
Physical Science
... Each row in the table of elements is a period. • Hydrogen, the first element in Period 1, has one electron in its first energy level. • Lithium, the first element in Period 2, has one electron in its second energy level. • Sodium, the first element in Period 3, has one electron in its third energy l ...
... Each row in the table of elements is a period. • Hydrogen, the first element in Period 1, has one electron in its first energy level. • Lithium, the first element in Period 2, has one electron in its second energy level. • Sodium, the first element in Period 3, has one electron in its third energy l ...
The Periodic Table and Chemical Properties
... The designer of this table wanted to emphasize the periods, so it has two periods of 8 elements, then two of 18 elements, then two of 32, and so on. The “arms” that stick out from the spiral are the lanthanides and actinides. These are the elements that you will find in those two rows down below the ...
... The designer of this table wanted to emphasize the periods, so it has two periods of 8 elements, then two of 18 elements, then two of 32, and so on. The “arms” that stick out from the spiral are the lanthanides and actinides. These are the elements that you will find in those two rows down below the ...
Period
... 1. What is the atomic number of cobalt? __________ 2. What is the atomic number of osmium? __________ 3. What is the atomic mass of germanium? __________ 4. What element is in group 1 and period 2? __________ 5. What element is in group 6 and period 4? __________ 6. What element is in group 12 and p ...
... 1. What is the atomic number of cobalt? __________ 2. What is the atomic number of osmium? __________ 3. What is the atomic mass of germanium? __________ 4. What element is in group 1 and period 2? __________ 5. What element is in group 6 and period 4? __________ 6. What element is in group 12 and p ...
Atomic Structure and the Periodic Table of Elements: The Secret
... o Element Reactivity/Stability – Atoms are generally most stable and least reactive when their outermost energy level is either completely filled with or empty of electrons. Reactivity refers to an atoms likelihood or desire to bond with another atom. See below for a chart of reactivity in the Peri ...
... o Element Reactivity/Stability – Atoms are generally most stable and least reactive when their outermost energy level is either completely filled with or empty of electrons. Reactivity refers to an atoms likelihood or desire to bond with another atom. See below for a chart of reactivity in the Peri ...
Periodic Classification of Elements
... 3A. Main Features of Long form of the Periodic Table: The long form of the periodic table relates the properties of elements to their electronic configuration. This is otherwise called "Modern Periodic Table". 1. It consist of 7 periods and 18 groups. 2. Every period starts with alkali metal and end ...
... 3A. Main Features of Long form of the Periodic Table: The long form of the periodic table relates the properties of elements to their electronic configuration. This is otherwise called "Modern Periodic Table". 1. It consist of 7 periods and 18 groups. 2. Every period starts with alkali metal and end ...
Periodic Table Presentation Lesson
... The elements are arranged on the periodic table according to their atomic numbers. On the table the atomic numbers increase from left to right. ...
... The elements are arranged on the periodic table according to their atomic numbers. On the table the atomic numbers increase from left to right. ...
2 Atoms, Bonding, and the Periodic Table
... number of valance electrons, and help them appreciate the significance of valence electrons. Teach Point out the pattern of valence electrons in the electron dot diagrams in Figure 11. Ask: Which two elements are in Group 1? (Lithium and sodium) How many valence electrons does each one have? (One) I ...
... number of valance electrons, and help them appreciate the significance of valence electrons. Teach Point out the pattern of valence electrons in the electron dot diagrams in Figure 11. Ask: Which two elements are in Group 1? (Lithium and sodium) How many valence electrons does each one have? (One) I ...
Periodic Table Packet
... • Identify the properties of metals, nonmetals, and seniimetals • Identify and describe allotropes • Define ionization energy • Use Table S to find the first ionization energies of the elements • Explain the trend in ionization energy across the periodic table and within a group • Define electronega ...
... • Identify the properties of metals, nonmetals, and seniimetals • Identify and describe allotropes • Define ionization energy • Use Table S to find the first ionization energies of the elements • Explain the trend in ionization energy across the periodic table and within a group • Define electronega ...
Document
... • The elements were first organized by increasing atomic mass, which led to inconsistencies. Later, they were organized by increasing atomic number. • The periodic law states that when the elements are arranged by increasing atomic number, there is a periodic repetition of their chemical and physica ...
... • The elements were first organized by increasing atomic mass, which led to inconsistencies. Later, they were organized by increasing atomic number. • The periodic law states that when the elements are arranged by increasing atomic number, there is a periodic repetition of their chemical and physica ...
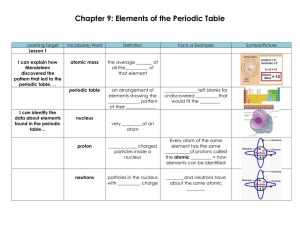
Chapter 9: Elements of the Periodic Table
... do not ordinarily form _____________ because atoms of noble gases do not usually ______, lose or _________ electrons ...
... do not ordinarily form _____________ because atoms of noble gases do not usually ______, lose or _________ electrons ...
Periods and Blocks of the Periodic Table
... • Mendeleev noticed that when the elements were arranged in order of increasing atomic mass, certain similarities in their chemical properties appeared at regular intervals. • Repeating patterns are referred to as periodic. • Mendeleev created a table in which elements with similar properties were g ...
... • Mendeleev noticed that when the elements were arranged in order of increasing atomic mass, certain similarities in their chemical properties appeared at regular intervals. • Repeating patterns are referred to as periodic. • Mendeleev created a table in which elements with similar properties were g ...
Document
... elements. Scientists during Dmitri’s time were trying to figure out an easy way in which they could organize the elements of matter so that it would be easy for them to communicate about their properties (i.e. scientists, even back then, were lazy and they didn’t want to have to memorize all the ele ...
... elements. Scientists during Dmitri’s time were trying to figure out an easy way in which they could organize the elements of matter so that it would be easy for them to communicate about their properties (i.e. scientists, even back then, were lazy and they didn’t want to have to memorize all the ele ...
Organizing the periodic table
... The vertical columns of the periodic table are known as a group. Another name for each group is a “family”. Each group is filled with atoms which have similar characteristics. There are eighteen groups in the periodic table. The lanthanides and actinides do not fit in the periodic table because the ...
... The vertical columns of the periodic table are known as a group. Another name for each group is a “family”. Each group is filled with atoms which have similar characteristics. There are eighteen groups in the periodic table. The lanthanides and actinides do not fit in the periodic table because the ...
(Periodic Trends) - stroh
... Rank the following sets of elements in order of decreasing electronegativity (big small). Set A: Cl, At, I, F, Br Set B: Te, Xe, Sn, In Set C: Rb, K, Sr, Ca ...
... Rank the following sets of elements in order of decreasing electronegativity (big small). Set A: Cl, At, I, F, Br Set B: Te, Xe, Sn, In Set C: Rb, K, Sr, Ca ...
General Structure of the Periodic Table
... its name). In 1879, Mendeleyev's periodic table received a powerful boost in general acceptance when it predicted the existence of the elements gallium, germanium, and scandium. Through time, Mendeleyev's periodic table has undergone some small changes. Many, many new elements have been added. The d ...
... its name). In 1879, Mendeleyev's periodic table received a powerful boost in general acceptance when it predicted the existence of the elements gallium, germanium, and scandium. Through time, Mendeleyev's periodic table has undergone some small changes. Many, many new elements have been added. The d ...
Periodic Table Study Guide
... Use a periodic table to answer the following questions: 17) Where are the metals located on the periodic table? 18) Where are the nonmetals located on the periodic table? 19) What classification of elements are between the metals and nonmetals on the periodic table? 20) What is the standard state f ...
... Use a periodic table to answer the following questions: 17) Where are the metals located on the periodic table? 18) Where are the nonmetals located on the periodic table? 19) What classification of elements are between the metals and nonmetals on the periodic table? 20) What is the standard state f ...
module-21 (worksheet-1)
... Answer the following questions: 1) Name two elements you would expect to show chemical reactions similar to magnesium. What is the basis for your choice? ___________________________________________________________________________ ______________________________________________________________________ ...
... Answer the following questions: 1) Name two elements you would expect to show chemical reactions similar to magnesium. What is the basis for your choice? ___________________________________________________________________________ ______________________________________________________________________ ...
TEST-Periodic Table
... a. Cl (chlorine) c. F (fluorine) b. I (iodine) d. Br (bromine) ____ 21. Among the alkali metals, the tendency to react with other substances a. does not vary among the members of the group. b. increases from top to bottom within the group. c. varies in an unpredictable way within the group. d. decre ...
... a. Cl (chlorine) c. F (fluorine) b. I (iodine) d. Br (bromine) ____ 21. Among the alkali metals, the tendency to react with other substances a. does not vary among the members of the group. b. increases from top to bottom within the group. c. varies in an unpredictable way within the group. d. decre ...
CHAPTER-3 CLASSIFICATION OF ELEMENTS
... Ans- The 1st ionisation enthalpy of magnesium is higher than that of Na due to higher nuclear charge and slightly smaller atomic radius of Mg than Na. After the loss of first electron, Na+ formed has the electronic configuration of neon (2,8). The higher stability of the completely filled noble gas ...
... Ans- The 1st ionisation enthalpy of magnesium is higher than that of Na due to higher nuclear charge and slightly smaller atomic radius of Mg than Na. After the loss of first electron, Na+ formed has the electronic configuration of neon (2,8). The higher stability of the completely filled noble gas ...
File
... temperature Most are metals Success Criteria: Can I recognize that all matter consists of atoms? (SPI0807.9.1) Can I use the Periodic Table to determine the properties of an element? (SPI0807.9.9) ...
... temperature Most are metals Success Criteria: Can I recognize that all matter consists of atoms? (SPI0807.9.1) Can I use the Periodic Table to determine the properties of an element? (SPI0807.9.9) ...
periods - Madeira City Schools
... ¥ Elements at the bottom of the periodic table have electrons that are more “unhappy” than the elements at the top of the periodic table. ¥ Elements at the bottom of the periodic table are larger (greater atomic radius, remember the trend!) because they have more energy levels. Those electrons in ...
... ¥ Elements at the bottom of the periodic table have electrons that are more “unhappy” than the elements at the top of the periodic table. ¥ Elements at the bottom of the periodic table are larger (greater atomic radius, remember the trend!) because they have more energy levels. Those electrons in ...
Noble gas

The noble gases make a group of chemical elements with similar properties. Under standard conditions, they are all odorless, colorless, monatomic gases with very low chemical reactivity. The six noble gases that occur naturally are helium (He), neon (Ne), argon (Ar), krypton (Kr), xenon (Xe), and the radioactive radon (Rn).For the first six periods of the periodic table, the noble gases are exactly the members of group 18 of the periodic table.It is possible that due to relativistic effects, the group 14 element flerovium exhibits some noble-gas-like properties, instead of the group 18 element ununoctium. Noble gases are typically highly unreactive except when under particular extreme conditions. The inertness of noble gases makes them very suitable in applications where reactions are not wanted. For example: argon is used in lightbulbs to prevent the hot tungsten filament from oxidizing; also, helium is breathed by deep-sea divers to prevent oxygen and nitrogen toxicity.The properties of the noble gases can be well explained by modern theories of atomic structure: their outer shell of valence electrons is considered to be ""full"", giving them little tendency to participate in chemical reactions, and it has been possible to prepare only a few hundred noble gas compounds. The melting and boiling points for a given noble gas are close together, differing by less than 10 °C (18 °F); that is, they are liquids over only a small temperature range.Neon, argon, krypton, and xenon are obtained from air in an air separation unit using the methods of liquefaction of gases and fractional distillation. Helium is sourced from natural gas fields which have high concentrations of helium in the natural gas, using cryogenic gas separation techniques, and radon is usually isolated from the radioactive decay of dissolved radium, thorium, or uranium compounds (since those compounds give off alpha particles). Noble gases have several important applications in industries such as lighting, welding, and space exploration. A helium-oxygen breathing gas is often used by deep-sea divers at depths of seawater over 55 m (180 ft) to keep the diver from experiencing oxygen toxemia, the lethal effect of high-pressure oxygen, and nitrogen narcosis, the distracting narcotic effect of the nitrogen in air beyond this partial-pressure threshold. After the risks caused by the flammability of hydrogen became apparent, it was replaced with helium in blimps and balloons.