1.2 Calculations
... Avogadro's Number There are 6.022 x 1023 atoms in 12 grams of carbon-12. Therefore explained in simpler terms 'One mole of any specified entity contains 6.022 x 1023 of ...
... Avogadro's Number There are 6.022 x 1023 atoms in 12 grams of carbon-12. Therefore explained in simpler terms 'One mole of any specified entity contains 6.022 x 1023 of ...
Chp 5 Circle the correct answer Consider three 1
... a) The rms velocity of Ar in both containers is the same. b) The average kinetic energy of Ar in both containers is the same. c) The force in which the Ar atoms collide with their container is the same in both containers. d) The frequency with which the Ar atoms collide with their container is the s ...
... a) The rms velocity of Ar in both containers is the same. b) The average kinetic energy of Ar in both containers is the same. c) The force in which the Ar atoms collide with their container is the same in both containers. d) The frequency with which the Ar atoms collide with their container is the s ...
Final Exam - KFUPM Faculty List
... also only a small (maybe too small) negative Eea C) elements with equal values of Ei and Eea no, when they are equal, both are endothermic (Ei is always positive) and need energy D) an element with a large Ei and an element with a large negative Eea no, because the cation formation needs a large Ei ...
... also only a small (maybe too small) negative Eea C) elements with equal values of Ei and Eea no, when they are equal, both are endothermic (Ei is always positive) and need energy D) an element with a large Ei and an element with a large negative Eea no, because the cation formation needs a large Ei ...
GCE Getting Started - Edexcel
... © Pearson Education Ltd 2015. Copying permitted for purchasing institution only. This material is not copyright free. ...
... © Pearson Education Ltd 2015. Copying permitted for purchasing institution only. This material is not copyright free. ...
03 Stoichiometry
... o send the accelerated cations into a magnetic field o an accelerated cation creates it’s OWN magnetic field which perturbs the original magnetic field o this perturbation changes the path of the cation o the amount of deflection is proportional to the mass; heavy cations deflect little o ions hit a ...
... o send the accelerated cations into a magnetic field o an accelerated cation creates it’s OWN magnetic field which perturbs the original magnetic field o this perturbation changes the path of the cation o the amount of deflection is proportional to the mass; heavy cations deflect little o ions hit a ...
b - PianetaChimica
... curvature of the path is dependent upon the mass to charge ratio (m/z) of individual ions. Measurement of the radius of curvature thus gives an accurate measure of the m/z ratio for each ion. The abundance of ions detected for each m/z is plotted as a graph of the mass spectrum. Since ions with a mu ...
... curvature of the path is dependent upon the mass to charge ratio (m/z) of individual ions. Measurement of the radius of curvature thus gives an accurate measure of the m/z ratio for each ion. The abundance of ions detected for each m/z is plotted as a graph of the mass spectrum. Since ions with a mu ...
Differentiated Chemistry Worksheet and Laboratory
... Explain what happens when the electron of a hydrogen atom changes from a 2s orbital to a 5s orbital. ...
... Explain what happens when the electron of a hydrogen atom changes from a 2s orbital to a 5s orbital. ...
Topic 1: Quantitative chemistry
... involving oxygen, that during any chemical reaction no atoms were destroyed. 2. Law of Definite Proportions: analytical tests showed that any compound consists of the same elements in definite proportions e.g. in every water molecule 88.88% of its mass is oxygen. 3. Law of Combining Volumes of Gases ...
... involving oxygen, that during any chemical reaction no atoms were destroyed. 2. Law of Definite Proportions: analytical tests showed that any compound consists of the same elements in definite proportions e.g. in every water molecule 88.88% of its mass is oxygen. 3. Law of Combining Volumes of Gases ...
Stoichiometry, Lab Basics, Reactions
... ____ 19. Equal masses of three different ideal gases, X, Y, and Z, are mixed in a sealed rigid container. If the temperature of the system remains constant, which of the following statements about the partial pressure of gas X is correct? A) It is equal to 1/3 of the total pressure. B) It depends on ...
... ____ 19. Equal masses of three different ideal gases, X, Y, and Z, are mixed in a sealed rigid container. If the temperature of the system remains constant, which of the following statements about the partial pressure of gas X is correct? A) It is equal to 1/3 of the total pressure. B) It depends on ...
Discussion Questions
... 60. Terephthalic acid is an important chemical used in the manufacture of polyesters and plasticizers. It contains only C, H, and O. Combustion of 19.81 mg terephthalic acid produces 41.98 mg CO2 and 6.45 mg H2O. If 0.250 mole of terephthalic acid has a mass of 41.5 g, determine the molecular fo ...
... 60. Terephthalic acid is an important chemical used in the manufacture of polyesters and plasticizers. It contains only C, H, and O. Combustion of 19.81 mg terephthalic acid produces 41.98 mg CO2 and 6.45 mg H2O. If 0.250 mole of terephthalic acid has a mass of 41.5 g, determine the molecular fo ...
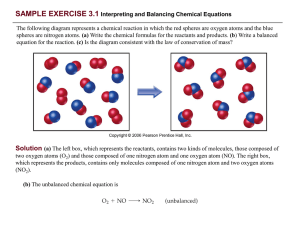
PRACTICE EXERCISE - Needham.K12.ma.us
... (c) The left box (reactants) contains four O2 molecules and eight NO molecules. Thus, the molecular ratio is one O2 for each two NO as required by the balanced equation. The right box (products) contains eight NO 2 molecules. The number of NO2 molecules on the right equals the number of NO molecules ...
... (c) The left box (reactants) contains four O2 molecules and eight NO molecules. Thus, the molecular ratio is one O2 for each two NO as required by the balanced equation. The right box (products) contains eight NO 2 molecules. The number of NO2 molecules on the right equals the number of NO molecules ...
- sartep.com
... Volume of the solid sample = 11.0 cubic centimeters The data above were gathered in order to determine the density of an unknown solid. The density of the sample should be reported as: (A) 0.5 g/cm3 (B) 0.50 g/cm3 (C) 2.0 g/cm3 (D) 2.00 g/cm3 18. __________A method of separation where a mobile phase ...
... Volume of the solid sample = 11.0 cubic centimeters The data above were gathered in order to determine the density of an unknown solid. The density of the sample should be reported as: (A) 0.5 g/cm3 (B) 0.50 g/cm3 (C) 2.0 g/cm3 (D) 2.00 g/cm3 18. __________A method of separation where a mobile phase ...
chemistry 2.1
... This achievement standard requires the student to present a report on the ‘Chemistry used in the development of a current technology’, using appropriate chemistry vocabulary, symbols and conventions (including names, formulae and equations). Before beginning this assessment, students will need to ha ...
... This achievement standard requires the student to present a report on the ‘Chemistry used in the development of a current technology’, using appropriate chemistry vocabulary, symbols and conventions (including names, formulae and equations). Before beginning this assessment, students will need to ha ...
5. Formulae, equations and amounts of substance
... same number of particles as there are atoms in 12 grams of carbon-12. DEFINITION: Relative atomic mass is the average mass of one atom compared to one twelfth of the mass of one atom of carbon-12 ...
... same number of particles as there are atoms in 12 grams of carbon-12. DEFINITION: Relative atomic mass is the average mass of one atom compared to one twelfth of the mass of one atom of carbon-12 ...
Chapter 10 Chemical Quantities - CNG Chemistry | Resources
... - difficult to measure their masses But, we will still need to know how many moles of gas we have we must know how many particles (atoms, ion, molecules) are taking part in chemical reactions Easy to measure the volume of a gas, but 2 things effect volumes of molar amounts of gases: a) Tempera ...
... - difficult to measure their masses But, we will still need to know how many moles of gas we have we must know how many particles (atoms, ion, molecules) are taking part in chemical reactions Easy to measure the volume of a gas, but 2 things effect volumes of molar amounts of gases: a) Tempera ...
mole concept type 1 - teko classes bhopal
... [Useful when only two reactant are there] By calculating amount of any one product obtained taking each reactant one by one irrespective of other reactants. The one giving least product is limiting reagent. Divide given moles of each reactant by their stoichiometric coefficient, the one with least r ...
... [Useful when only two reactant are there] By calculating amount of any one product obtained taking each reactant one by one irrespective of other reactants. The one giving least product is limiting reagent. Divide given moles of each reactant by their stoichiometric coefficient, the one with least r ...
UNITS OF CONCENTRATION
... takes into account the actual number of reacting species per mole of reagent (i.e., protons in the case of acid/base reactions or electrons in the case of redox reactions). For acids, an equivalent is defined as one mole of protons. The equivalent amount of any acid is the amount of acid that delive ...
... takes into account the actual number of reacting species per mole of reagent (i.e., protons in the case of acid/base reactions or electrons in the case of redox reactions). For acids, an equivalent is defined as one mole of protons. The equivalent amount of any acid is the amount of acid that delive ...
ХИМИЯ НА АНГЛИЙСКОМ ЯЗЫКЕ
... distilled water. A 10-mL aliquot of the solution was pipeted into a 500-mL volumetric flask and diluted to volume. Express the concentration of Mn in parts per million, and estimate uncertainty by a propagation of uncertainty calculation. Would the uncertainty in the solution’s concentration be impr ...
... distilled water. A 10-mL aliquot of the solution was pipeted into a 500-mL volumetric flask and diluted to volume. Express the concentration of Mn in parts per million, and estimate uncertainty by a propagation of uncertainty calculation. Would the uncertainty in the solution’s concentration be impr ...
CH 5 CHEM1A GASES - Santa Rosa Junior College
... fluorine. A 5.00-L flask containing 10.0 g of I2 is charged with 10.0 g of F2 and the reaction proceeds until one of the reactants is completely consumed. After the reaction is complete, the temperature in the flask is 125 ºC. a) What is the partial pressure of IF5 in the flask? b) What is the mole ...
... fluorine. A 5.00-L flask containing 10.0 g of I2 is charged with 10.0 g of F2 and the reaction proceeds until one of the reactants is completely consumed. After the reaction is complete, the temperature in the flask is 125 ºC. a) What is the partial pressure of IF5 in the flask? b) What is the mole ...
Bulk and Surface Micromachining
... Bulk and Surface Micromachining for the Miller Indices <110>:<100>:<111>, planar selectivity can be as high as 600:400:1. However, KOH is not used in micromachining because its potassium ion content bans it from clean room operations. Also, it is highly corrosive and attacks aluminum, which makes it ...
... Bulk and Surface Micromachining for the Miller Indices <110>:<100>:<111>, planar selectivity can be as high as 600:400:1. However, KOH is not used in micromachining because its potassium ion content bans it from clean room operations. Also, it is highly corrosive and attacks aluminum, which makes it ...
KISS Notes
... • metal can made into intricate shapes, such as j)..........................., not possible in stone. Later, bronze was replaced by k)...................... which is l)................ ...................... and....................., but requires more m)............................ for its extractio ...
... • metal can made into intricate shapes, such as j)..........................., not possible in stone. Later, bronze was replaced by k)...................... which is l)................ ...................... and....................., but requires more m)............................ for its extractio ...
2. CHEMICAL ACTIVITY of the METALS 3. PATTERNS of the
... f)............................... and ........................................... This produced the alloy g)......................................, which made tools and weapons with many advantages over stone: • metal is h)............................ and ............................ and is not i).. ...
... f)............................... and ........................................... This produced the alloy g)......................................, which made tools and weapons with many advantages over stone: • metal is h)............................ and ............................ and is not i).. ...
physical setting chemistry
... Answer all questions in this part. Directions (66–83): Record your answers in the spaces provided in your answer booklet. Some questions may require the use of the Reference Tables for Physical Setting/Chemistry. Base your answers to questions 66 and 67 on the information below. In 1897, J. J. Thoms ...
... Answer all questions in this part. Directions (66–83): Record your answers in the spaces provided in your answer booklet. Some questions may require the use of the Reference Tables for Physical Setting/Chemistry. Base your answers to questions 66 and 67 on the information below. In 1897, J. J. Thoms ...
N Goalby chemrevise.org 1 2.5 Transition Metals Substitution
... [Cu(H2O)6]2+ (aq) + EDTA4- (aq) [Cu (EDTA)]2- (aq) + 6H2O (l) The copper complex ion has changed from having unidentate ligands to a multidentate ligand. In this reaction there is an increase in the entropy because there are more moles of products than reactants (from 2 to 7), creating more disord ...
... [Cu(H2O)6]2+ (aq) + EDTA4- (aq) [Cu (EDTA)]2- (aq) + 6H2O (l) The copper complex ion has changed from having unidentate ligands to a multidentate ligand. In this reaction there is an increase in the entropy because there are more moles of products than reactants (from 2 to 7), creating more disord ...
ANALYSIS OF THE SILVER GROUP CATIONS
... may be possible to test for one particular ion in the presence of just one or two others. Alternatively, each subgroup of just a few ions may be separated further so that each ion in the subgroup ends up in a different test tube where its presence can be confirmed by other chemical tests. The chemic ...
... may be possible to test for one particular ion in the presence of just one or two others. Alternatively, each subgroup of just a few ions may be separated further so that each ion in the subgroup ends up in a different test tube where its presence can be confirmed by other chemical tests. The chemic ...
Inductively coupled plasma mass spectrometry

Inductively coupled plasma mass spectrometry (ICP-MS) is a type of mass spectrometry which is capable of detecting metals and several non-metals at concentrations as low as one part in 1015 (part per quadrillion, ppq) on non-interfered low-background isotopes. This is achieved by ionizing the sample with inductively coupled plasma and then using a mass spectrometer to separate and quantify those ions.Compared to atomic absorption techniques, ICP-MS has greater speed, precision, and sensitivity. However, compared with other types of mass spectrometry, such as Thermal ionization mass spectrometry (TIMS) and Glow Discharge Mass Spectrometry (GD-MS), ICP-MS introduces a lot of interfering species: argon from the plasma, component gasses of air that leak through the cone orifices, and contamination from glassware and the cones.The variety of applications exceeds that of inductively coupled plasma atomic emission spectroscopy and includes isotopic speciation. Due to possible applications in nuclear technologies, ICP-MS hardware is a subject for special exporting regulations.