Chemistry 11 – Course Review
... 14. Circle the most reactive element in the following: Na Mg Si Al Ar 15. Circle the most reactive element in the following: Na K Rb Cs Li 16. Circle the most reactive element in the following: Cl Br I At Ne 17. Circle the element with the largest atomic radius of these: Na Mg Si Al Ar 18. Circle th ...
... 14. Circle the most reactive element in the following: Na Mg Si Al Ar 15. Circle the most reactive element in the following: Na K Rb Cs Li 16. Circle the most reactive element in the following: Cl Br I At Ne 17. Circle the element with the largest atomic radius of these: Na Mg Si Al Ar 18. Circle th ...
(NH 3 ) 2 - GZ @ Science Class Online
... Carry out procedures to identify ions involves collecting primary data and using these observations to identify ions in a solution using a procedure provided. Identification of ions must be supported by experimental observations and identification of all precipitates formed. Ions to be identified wi ...
... Carry out procedures to identify ions involves collecting primary data and using these observations to identify ions in a solution using a procedure provided. Identification of ions must be supported by experimental observations and identification of all precipitates formed. Ions to be identified wi ...
Topic 1: Quantitative chemistry
... involving oxygen, that during any chemical reaction no atoms were destroyed. 2. Law of Definite Proportions: analytical tests showed that any compound consists of the same elements in definite proportions e.g. in every water molecule 88.88% of its mass is oxygen. 3. Law of Combining Volumes of Gases ...
... involving oxygen, that during any chemical reaction no atoms were destroyed. 2. Law of Definite Proportions: analytical tests showed that any compound consists of the same elements in definite proportions e.g. in every water molecule 88.88% of its mass is oxygen. 3. Law of Combining Volumes of Gases ...
Personal Tutoring Help on Questions and Problems
... 3.11 Earth’s population is about 6.5 billion. Suppose that every person on Earth participates in a process of counting identical particles at the rate of two particles per second. How many years would it take to count 6.0 ⫻ 1023 particles? Assume that there are 365 days in a year. 3.12 The thickness ...
... 3.11 Earth’s population is about 6.5 billion. Suppose that every person on Earth participates in a process of counting identical particles at the rate of two particles per second. How many years would it take to count 6.0 ⫻ 1023 particles? Assume that there are 365 days in a year. 3.12 The thickness ...
CHAPTER 3 STOICHIOMETRY:
... Answer: 1 mol H2O (6 ×1023O atoms) < 3 ×1023molecules O3(9 ×1023O atoms) < 1 mol CO2(12 ×1023O atoms) ...
... Answer: 1 mol H2O (6 ×1023O atoms) < 3 ×1023molecules O3(9 ×1023O atoms) < 1 mol CO2(12 ×1023O atoms) ...
Grade 11 review answers
... b) Why is the second ionization energy of K greater than that of Ca while the first ionization energy of Calcium is higher than that of Potassium? Since Potassium has only one electron in its outer shell, the second electron must be removed from the next shell in, which takes a lot more energy. Calc ...
... b) Why is the second ionization energy of K greater than that of Ca while the first ionization energy of Calcium is higher than that of Potassium? Since Potassium has only one electron in its outer shell, the second electron must be removed from the next shell in, which takes a lot more energy. Calc ...
Use the following answers for questions 1
... 73. The volume of distilled water that should be added to 10.0 mL of 6.00 M HCl(aq) in order to prepare a 0.500 M HCl(aq) solution is approximately A) 50.0 mL B) 60.0 mL C) 100. mL D) 110. mL E) 120. mL 21. When a sample of oxygen gas in a closed container of constant volume is heated until its abso ...
... 73. The volume of distilled water that should be added to 10.0 mL of 6.00 M HCl(aq) in order to prepare a 0.500 M HCl(aq) solution is approximately A) 50.0 mL B) 60.0 mL C) 100. mL D) 110. mL E) 120. mL 21. When a sample of oxygen gas in a closed container of constant volume is heated until its abso ...
College Grossmont 115
... or numbers obtained by definition. For example, we can count the fingers on our hand and get an exact number (most people have 5). There is no uncertainty in this result, but we cannot count large groups of objects without some degree of uncertainty. For example, the number of stars in our galaxy is ...
... or numbers obtained by definition. For example, we can count the fingers on our hand and get an exact number (most people have 5). There is no uncertainty in this result, but we cannot count large groups of objects without some degree of uncertainty. For example, the number of stars in our galaxy is ...
Atomic Masses
... Topic: AMU’s & Atomic Mass Objectives: Day 1 of 3 • To learn how we define 1 amu (atomic mass unit) • To learn how we derive atomic mass from amu (atomic mass unit) • To learn how atomic mass is calculated using the average natural abundance of isotopes ...
... Topic: AMU’s & Atomic Mass Objectives: Day 1 of 3 • To learn how we define 1 amu (atomic mass unit) • To learn how we derive atomic mass from amu (atomic mass unit) • To learn how atomic mass is calculated using the average natural abundance of isotopes ...
Introduction to Qualitative Analysis
... compound could be used to reveal that iron, potassium and oxalate ions are present. It could also be used to determine that the iron is in its +3 oxidation state and that the substance is a hydrate. On the other hand, quantitative analysis of the green crystals would reveal that it contains 11.4% Fe ...
... compound could be used to reveal that iron, potassium and oxalate ions are present. It could also be used to determine that the iron is in its +3 oxidation state and that the substance is a hydrate. On the other hand, quantitative analysis of the green crystals would reveal that it contains 11.4% Fe ...
Materials - Hodder Education
... using X-ray crystallography (see section 22.8). The majority of solids, including metals and their alloys as well as ceramics, are crystalline. A few plastics have some degree of crystallinity, for example high-density polythene (up to 70 per cent). Amorphous (disordered) materials have their partic ...
... using X-ray crystallography (see section 22.8). The majority of solids, including metals and their alloys as well as ceramics, are crystalline. A few plastics have some degree of crystallinity, for example high-density polythene (up to 70 per cent). Amorphous (disordered) materials have their partic ...
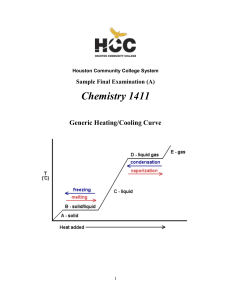
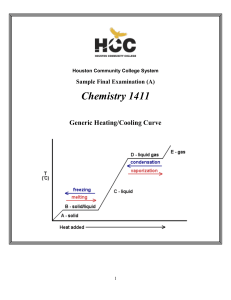
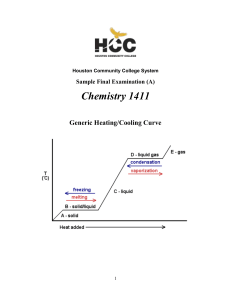
1411FINALSAMPLE+KEY - Houston Community College
... second structure, which has an “expanded octet” around the sulfur atom. Hybridizations that allow more than an octet of electrons around an atom are sp3d (10 electrons) and sp3d2 (12 electrons), but not sp, sp2, or sp3. Furthermore, to form a -bond, we must have unhybridized p-orbitals on the sulfu ...
... second structure, which has an “expanded octet” around the sulfur atom. Hybridizations that allow more than an octet of electrons around an atom are sp3d (10 electrons) and sp3d2 (12 electrons), but not sp, sp2, or sp3. Furthermore, to form a -bond, we must have unhybridized p-orbitals on the sulfu ...
CHEM-1411 Final Practice Exam
... second structure, which has an “expanded octet” around the sulfur atom. Hybridizations that allow more than an octet of electrons around an atom are sp3d (10 electrons) and sp3d2 (12 electrons), but not sp, sp2, or sp3. Furthermore, to form a -bond, we must have unhybridized p-orbitals on the sulfu ...
... second structure, which has an “expanded octet” around the sulfur atom. Hybridizations that allow more than an octet of electrons around an atom are sp3d (10 electrons) and sp3d2 (12 electrons), but not sp, sp2, or sp3. Furthermore, to form a -bond, we must have unhybridized p-orbitals on the sulfu ...
Chemical Quantities: Stoichiometry and the Mole
... of barium sulfate is obtained. What mass of barium sulfate was contained in the solution? 13) A 0.187 g sample of impure aluminum metaal was treated with excess sulfuric acid. 0.921 g of aluminum sulfate was obtained. Calculate the percent purity of the aluminum sample. 14) A solution containing 15. ...
... of barium sulfate is obtained. What mass of barium sulfate was contained in the solution? 13) A 0.187 g sample of impure aluminum metaal was treated with excess sulfuric acid. 0.921 g of aluminum sulfate was obtained. Calculate the percent purity of the aluminum sample. 14) A solution containing 15. ...
Chemistry
... Chapter 18 (electrochemistry) is not included on this Review Guide but will be on the Exam. Use the notes, handouts, homework, and textbook to review. It’s a short chapter and the topic we covered most recently, so it should be fresh in your mind. Chapter 8 – The Mole and Chemical Composition Topics ...
... Chapter 18 (electrochemistry) is not included on this Review Guide but will be on the Exam. Use the notes, handouts, homework, and textbook to review. It’s a short chapter and the topic we covered most recently, so it should be fresh in your mind. Chapter 8 – The Mole and Chemical Composition Topics ...
2014 HSC Chemistry Marking Guidelines
... Ethanol can also be used as a source of ethylene. Ethanol is dehydrated by heating it with concentrated sulfuric or phosphoric acid. ...
... Ethanol can also be used as a source of ethylene. Ethanol is dehydrated by heating it with concentrated sulfuric or phosphoric acid. ...
Year 11 C2 Mock Exam Revision Questions
... Phosphorus and fluorine form a covalent compound, phosphorus trifluoride. Complete the sentences below which are about this compound. Phosphorus trifluoride is made up of phosphorus and fluorine ................................ These are joined together by sharing pairs of .......................... ...
... Phosphorus and fluorine form a covalent compound, phosphorus trifluoride. Complete the sentences below which are about this compound. Phosphorus trifluoride is made up of phosphorus and fluorine ................................ These are joined together by sharing pairs of .......................... ...
Key Words Questions and Problems
... accordance with the law of conservation of mass. The number of atoms of each element in the reactants must equal the number in the products. Stoichiometry is the quantitative study of products and reactants in chemical reactions. Stoichiometric calculations are best done by expressing both the known ...
... accordance with the law of conservation of mass. The number of atoms of each element in the reactants must equal the number in the products. Stoichiometry is the quantitative study of products and reactants in chemical reactions. Stoichiometric calculations are best done by expressing both the known ...
1 What is the angular momentum quantum number (l) value for the
... of all free radicals? They all: A have an unpaired electron. CORRECT: All free radicals, by definition, have one or more unpaired electrons. It is this characteristic that typically makes free radicals highly reactive. B are neutral atoms. INCORRECT: Free radicals can be molecular species and can ...
... of all free radicals? They all: A have an unpaired electron. CORRECT: All free radicals, by definition, have one or more unpaired electrons. It is this characteristic that typically makes free radicals highly reactive. B are neutral atoms. INCORRECT: Free radicals can be molecular species and can ...
Final Exam Review Notes
... SCIENTIFIC NOTATION Some numbers are very large or very small difficult to express. For example, Avogadro’s number = 602,000,000,000,000,000,000,000 an electron’s mass = 0.000 000 000 000 000 000 000 000 000 91 kg Also, it's not clear how many sig figs there are in some measurements. For example, ...
... SCIENTIFIC NOTATION Some numbers are very large or very small difficult to express. For example, Avogadro’s number = 602,000,000,000,000,000,000,000 an electron’s mass = 0.000 000 000 000 000 000 000 000 000 91 kg Also, it's not clear how many sig figs there are in some measurements. For example, ...
WORD - SSS Chemistry
... Bohr came up with an atomic model to explain the spectrum of ______________________. He said that the atom has certain _______________ levels which are allowed. These levels corresponded to ____________________ in which electrons move. If an electron absorbs a certain photon of energy, it will jump ...
... Bohr came up with an atomic model to explain the spectrum of ______________________. He said that the atom has certain _______________ levels which are allowed. These levels corresponded to ____________________ in which electrons move. If an electron absorbs a certain photon of energy, it will jump ...
1411FINALSAMPLEs and Key
... second structure, which has an “expanded octet” around the sulfur atom. Hybridizations that allow more than an octet of electrons around an atom are sp3d (10 electrons) and sp3d2 (12 electrons), but not sp, sp2, or sp3. Furthermore, to form a -bond, we must have unhybridized p-orbitals on the sulfu ...
... second structure, which has an “expanded octet” around the sulfur atom. Hybridizations that allow more than an octet of electrons around an atom are sp3d (10 electrons) and sp3d2 (12 electrons), but not sp, sp2, or sp3. Furthermore, to form a -bond, we must have unhybridized p-orbitals on the sulfu ...
ESO - ENCIGA
... tested by subsequent investigation and can be modified by its results. Science does not give statements of absolute eternal truth, it only provides theories. We know that those theories will probably be refined in the future, and some of them may even be discarded in favour of theories that make mor ...
... tested by subsequent investigation and can be modified by its results. Science does not give statements of absolute eternal truth, it only provides theories. We know that those theories will probably be refined in the future, and some of them may even be discarded in favour of theories that make mor ...
CH 151 Companion
... accurate observation of behavior of the system of interest. Conclusions are based solely on the observed data. Only by using experimental observations to study the behavior of matter, arranging the results of such studies in an orderly fashions, correlating the observed data and testing these correl ...
... accurate observation of behavior of the system of interest. Conclusions are based solely on the observed data. Only by using experimental observations to study the behavior of matter, arranging the results of such studies in an orderly fashions, correlating the observed data and testing these correl ...
ض ( ا ء ا ط ك ا رر 108 1) -
... Which state of the matter has definite volume and indefinite shape? solid B plasma C liquid D gas Which state of the matter has indefinite volume and indefinite shape? ...
... Which state of the matter has definite volume and indefinite shape? solid B plasma C liquid D gas Which state of the matter has indefinite volume and indefinite shape? ...
Inductively coupled plasma mass spectrometry

Inductively coupled plasma mass spectrometry (ICP-MS) is a type of mass spectrometry which is capable of detecting metals and several non-metals at concentrations as low as one part in 1015 (part per quadrillion, ppq) on non-interfered low-background isotopes. This is achieved by ionizing the sample with inductively coupled plasma and then using a mass spectrometer to separate and quantify those ions.Compared to atomic absorption techniques, ICP-MS has greater speed, precision, and sensitivity. However, compared with other types of mass spectrometry, such as Thermal ionization mass spectrometry (TIMS) and Glow Discharge Mass Spectrometry (GD-MS), ICP-MS introduces a lot of interfering species: argon from the plasma, component gasses of air that leak through the cone orifices, and contamination from glassware and the cones.The variety of applications exceeds that of inductively coupled plasma atomic emission spectroscopy and includes isotopic speciation. Due to possible applications in nuclear technologies, ICP-MS hardware is a subject for special exporting regulations.