Chemistry 133 Problem Set Introduction
... 1.95 Round the mass values to two significant figures, and round the length values to three significant figures. (a) 11.345 kg (b) 122.22 km (c) 10.7500 cm (d) 6000 g (e) 5.280 × 10 3 m. 1.96 A small beaker is weighed on a balance and found to have a mass of 25.48 g. When it is carefully filled to ...
... 1.95 Round the mass values to two significant figures, and round the length values to three significant figures. (a) 11.345 kg (b) 122.22 km (c) 10.7500 cm (d) 6000 g (e) 5.280 × 10 3 m. 1.96 A small beaker is weighed on a balance and found to have a mass of 25.48 g. When it is carefully filled to ...
Exam 2 Key
... 10. At elevated temperatures, solid sodium chlorate (NaClO3, molar mass 106.44 g/mol) decomposes to produce sodium chloride and oxygen gas. In an experiment, a 0.8765 g sample of impure sodium chlorate was heated until the production of oxygen ceased. The O2 gas was collected over water. The collec ...
... 10. At elevated temperatures, solid sodium chlorate (NaClO3, molar mass 106.44 g/mol) decomposes to produce sodium chloride and oxygen gas. In an experiment, a 0.8765 g sample of impure sodium chlorate was heated until the production of oxygen ceased. The O2 gas was collected over water. The collec ...
å¾è湿çå¦
... 19 A 10.0 L cylinder containing neon gas with a measured pressure of 550 kPa at 298 K is connected through a valve to a 2.50 L cylinder containing 275 kPa of helium gas at 298 K. The valve is opened and the gases mix with no change in temperature. What is the final total pressure in the system? ...
... 19 A 10.0 L cylinder containing neon gas with a measured pressure of 550 kPa at 298 K is connected through a valve to a 2.50 L cylinder containing 275 kPa of helium gas at 298 K. The valve is opened and the gases mix with no change in temperature. What is the final total pressure in the system? ...
chapter twenty-one transition metals and coordination chemistry
... different. The tetrahedrally oriented ligands point differently in relationship to the d-orbitals than do the octahedrally oriented ligands. Plus, we have more ligands in an octahedral complex. See Figure 21.27 for the tetrahedral crystal field diagram. Notice that the orbitals are reverse of that i ...
... different. The tetrahedrally oriented ligands point differently in relationship to the d-orbitals than do the octahedrally oriented ligands. Plus, we have more ligands in an octahedral complex. See Figure 21.27 for the tetrahedral crystal field diagram. Notice that the orbitals are reverse of that i ...
odd - WWW2
... formed by the most electropositive metals. These may contain the dicarbide(2 ) ion, C22 , or the true carbide ion C4 . Both types of ionic carbides react with water to produce the appropriate hydrocarbon. Covalent carbides are formed by nonmetals, specifically boron and silicon, more electronegative ...
... formed by the most electropositive metals. These may contain the dicarbide(2 ) ion, C22 , or the true carbide ion C4 . Both types of ionic carbides react with water to produce the appropriate hydrocarbon. Covalent carbides are formed by nonmetals, specifically boron and silicon, more electronegative ...
noble gases
... Sc3+ has only 18 electrons that can be arranged as [Ar] 3d0 4s0. Hence no d-d transition of electrons and no colour. Vikasana - CET 2012 ...
... Sc3+ has only 18 electrons that can be arranged as [Ar] 3d0 4s0. Hence no d-d transition of electrons and no colour. Vikasana - CET 2012 ...
Non-ideal Plastic Behavior
... in a quality assurance laboratory. This procedure is often fairly timeconsuming and means that some of the product is usually wasted before a particular problem becomes apparent. For this reason, there is an ...
... in a quality assurance laboratory. This procedure is often fairly timeconsuming and means that some of the product is usually wasted before a particular problem becomes apparent. For this reason, there is an ...
application of hydroxyapatite in protein purification
... order to get pure hydroxyapatite which would be used as a matrix in protein purification for the next step of this research. So that, if there was any carbonate content in this research’s hydroxyapatite sample, it is only caused by incorporated CO2 from the air during precipitation process that can ...
... order to get pure hydroxyapatite which would be used as a matrix in protein purification for the next step of this research. So that, if there was any carbonate content in this research’s hydroxyapatite sample, it is only caused by incorporated CO2 from the air during precipitation process that can ...
CHAPTER 20 METALLURGY AND THE CHEMISTRY OF METALS
... Mg(s) + Cl2(g) → MgCl2(s) Magnesium will also react with HCl. Mg(s) + 2HCl(aq) → MgCl2(aq) + H2(g) Neither of the above methods are really practical because magnesium metal is expensive to produce (electrolysis of magnesium chloride!). Can you suggest a method starting with a magnesium compound like ...
... Mg(s) + Cl2(g) → MgCl2(s) Magnesium will also react with HCl. Mg(s) + 2HCl(aq) → MgCl2(aq) + H2(g) Neither of the above methods are really practical because magnesium metal is expensive to produce (electrolysis of magnesium chloride!). Can you suggest a method starting with a magnesium compound like ...
Chapter 2 - hrsbstaff.ednet.ns.ca
... ten neutrons, so A = 18. These three forms of oxygen are called isotopes. Isotopes are atoms of an element that have the same number of protons but different numbers of neutrons. The three isotopes of oxygen are called oxygen-16, oxygen-17, and oxygen-18. Oxygen-16 has the same meaning as 168O. Simi ...
... ten neutrons, so A = 18. These three forms of oxygen are called isotopes. Isotopes are atoms of an element that have the same number of protons but different numbers of neutrons. The three isotopes of oxygen are called oxygen-16, oxygen-17, and oxygen-18. Oxygen-16 has the same meaning as 168O. Simi ...
Holt Modern Chemistry Workbook: intro - ch 5
... balance. The mass of matter will always measure at greater than zero, because mass is a necessary property of matter. The second property of matter is that it takes up space. Another way to describe this property is to say that all matter has volume. An object such as a book or a tree takes up space ...
... balance. The mass of matter will always measure at greater than zero, because mass is a necessary property of matter. The second property of matter is that it takes up space. Another way to describe this property is to say that all matter has volume. An object such as a book or a tree takes up space ...
General chemistry laboratory activities, Lorentz
... accompanied by their necks, at the ends of which are ground glass joints to quickly and tightly connect to the rest of the apparatus (such as a reflux condenser or dropping funnel). The reaction flask is usually made of thick glass and they can tolerate large pressure differences, with the result th ...
... accompanied by their necks, at the ends of which are ground glass joints to quickly and tightly connect to the rest of the apparatus (such as a reflux condenser or dropping funnel). The reaction flask is usually made of thick glass and they can tolerate large pressure differences, with the result th ...
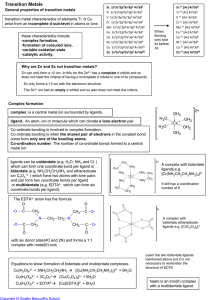
Transition Metals
... CO is toxic to humans as CO can from a strong coordinate bond with haemoglobin. This is a stronger bond than that made with oxygen and so it prevents the oxygen attaching to the haemoglobin.. The Pt(II) complex cisplatin is used as an anticancer drug. The cisplatin version only works as two chloride ...
... CO is toxic to humans as CO can from a strong coordinate bond with haemoglobin. This is a stronger bond than that made with oxygen and so it prevents the oxygen attaching to the haemoglobin.. The Pt(II) complex cisplatin is used as an anticancer drug. The cisplatin version only works as two chloride ...
For metals
... valence electron from an atom in the gas phase. High electronegativity means high ionization energy because if an atom is more attracted to electrons, it will take more energy to remove those electrons. Metals have low ionization energy. They lose electrons easily to form (+) charged ions. Nonmetals ...
... valence electron from an atom in the gas phase. High electronegativity means high ionization energy because if an atom is more attracted to electrons, it will take more energy to remove those electrons. Metals have low ionization energy. They lose electrons easily to form (+) charged ions. Nonmetals ...
Discussion Questions
... amount of titrant has been added to exactly react with the substance being analyzed n Endpoint: the point at which a chemical indicator changes color Oxidation–reduction reactions n Oxidation states are assigned using a set of rules to keep track of electron flow n Oxidation: increase in oxidatio ...
... amount of titrant has been added to exactly react with the substance being analyzed n Endpoint: the point at which a chemical indicator changes color Oxidation–reduction reactions n Oxidation states are assigned using a set of rules to keep track of electron flow n Oxidation: increase in oxidatio ...
Vinnitsa National Pirogov Memorial Medical University Biological
... The formation of complexes in biological systems……………………………………...13 Methods of expressing concentration of solution ……………………....………………16 Acid-base equilibrium in the organism. pH scale of biological liquids ………………...19 Volumetric analysis.Neutralization methods. Titration. Alkalimetry. Acidimetry……. ...
... The formation of complexes in biological systems……………………………………...13 Methods of expressing concentration of solution ……………………....………………16 Acid-base equilibrium in the organism. pH scale of biological liquids ………………...19 Volumetric analysis.Neutralization methods. Titration. Alkalimetry. Acidimetry……. ...
CHAPTER 19 TRANSITION METALS AND COORDINATION
... Sc3+ has no electrons in d orbitals. Ti3+ and V3+ have d electrons present. The color of transition metal complexes results from electron transfer between split d orbitals. If no d electrons are present, no electron transfer can occur, and the compounds are not colored. ...
... Sc3+ has no electrons in d orbitals. Ti3+ and V3+ have d electrons present. The color of transition metal complexes results from electron transfer between split d orbitals. If no d electrons are present, no electron transfer can occur, and the compounds are not colored. ...
AP Chemistry - Notes
... a. Atomic masses are based on 12C ("carbon twelve"), which is assigned a value of exactly 12 atomic mass units. b. The atomic masses of other elements are determined by comparison to 12C. c. Mass spectrometer - currently best method for determination of atomic masses of atoms - Procedure : - atoms o ...
... a. Atomic masses are based on 12C ("carbon twelve"), which is assigned a value of exactly 12 atomic mass units. b. The atomic masses of other elements are determined by comparison to 12C. c. Mass spectrometer - currently best method for determination of atomic masses of atoms - Procedure : - atoms o ...
Lactose/D-Galactose
... The reagents used in the determination of lactose and D-galactose are not hazardous materials in the sense of the Hazardous Substances Regulations, the Chemicals Law or EC Regulation 67/548/EEC and subsequent alteration, supplementation and adaptation guidelines. However, the general safety measures ...
... The reagents used in the determination of lactose and D-galactose are not hazardous materials in the sense of the Hazardous Substances Regulations, the Chemicals Law or EC Regulation 67/548/EEC and subsequent alteration, supplementation and adaptation guidelines. However, the general safety measures ...
Chemistry_Stoichiome..
... 80. 100 mL of 10 % NaOH (w/V) is added to 100 mL of 10 % HCl (w/V). The resultant solution becomes: a) alkaline b) strongly alkaline c) acidic d) neutral 81. Calculate the molality of 1 L solution of 80 % H2SO4 (w/V), given that the density of the solution is 1.80 g mL−1 . a) 9.18 b) 8.6 c) 1.02 d) ...
... 80. 100 mL of 10 % NaOH (w/V) is added to 100 mL of 10 % HCl (w/V). The resultant solution becomes: a) alkaline b) strongly alkaline c) acidic d) neutral 81. Calculate the molality of 1 L solution of 80 % H2SO4 (w/V), given that the density of the solution is 1.80 g mL−1 . a) 9.18 b) 8.6 c) 1.02 d) ...
Lactose/D-Galactose
... The reagents used in the determination of lactose and D-galactose are not hazardous materials in the sense of the Hazardous Substances Regulations, the Chemicals Law or EC Regulation 67/548/EEC and subsequent alteration, supplementation and adaptation guidelines. However, the general safety measures ...
... The reagents used in the determination of lactose and D-galactose are not hazardous materials in the sense of the Hazardous Substances Regulations, the Chemicals Law or EC Regulation 67/548/EEC and subsequent alteration, supplementation and adaptation guidelines. However, the general safety measures ...
Section 10.2
... In 1811, Amedeo Avogadro proposed a groundbreaking explanation. Avogadro’s hypothesis states that equal volumes of gases at the same temperature and pressure contain equal numbers of particles. The particles that make up different gases are not the same size. But the particles in all gases are so fa ...
... In 1811, Amedeo Avogadro proposed a groundbreaking explanation. Avogadro’s hypothesis states that equal volumes of gases at the same temperature and pressure contain equal numbers of particles. The particles that make up different gases are not the same size. But the particles in all gases are so fa ...
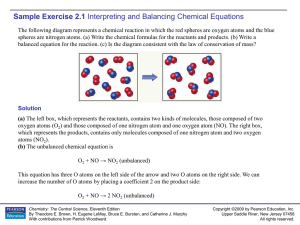
Sample Exercise 2.1
... Now there are two N atoms and four O atoms on the right. Placing the coefficient 2 in front of NO balances both the number of N atoms and O atoms: O2 + 2 NO → 2 NO2 (balanced) (c) The left box (reactants) contains four O2 molecules and eight NO molecules. Thus, the molecular ratio is one O2 for each ...
... Now there are two N atoms and four O atoms on the right. Placing the coefficient 2 in front of NO balances both the number of N atoms and O atoms: O2 + 2 NO → 2 NO2 (balanced) (c) The left box (reactants) contains four O2 molecules and eight NO molecules. Thus, the molecular ratio is one O2 for each ...
Inductively coupled plasma mass spectrometry

Inductively coupled plasma mass spectrometry (ICP-MS) is a type of mass spectrometry which is capable of detecting metals and several non-metals at concentrations as low as one part in 1015 (part per quadrillion, ppq) on non-interfered low-background isotopes. This is achieved by ionizing the sample with inductively coupled plasma and then using a mass spectrometer to separate and quantify those ions.Compared to atomic absorption techniques, ICP-MS has greater speed, precision, and sensitivity. However, compared with other types of mass spectrometry, such as Thermal ionization mass spectrometry (TIMS) and Glow Discharge Mass Spectrometry (GD-MS), ICP-MS introduces a lot of interfering species: argon from the plasma, component gasses of air that leak through the cone orifices, and contamination from glassware and the cones.The variety of applications exceeds that of inductively coupled plasma atomic emission spectroscopy and includes isotopic speciation. Due to possible applications in nuclear technologies, ICP-MS hardware is a subject for special exporting regulations.