Year Review Booklet (optional)
... an atom of Sr will __________ ___ electrons and become the ion ________ an atom of As will __________ ___ electrons and become the ion ________ an atom of Al will __________ ___ electrons and become the ion ________ an atom of Se will __________ ___ electrons and become the ion ________ an atom of N ...
... an atom of Sr will __________ ___ electrons and become the ion ________ an atom of As will __________ ___ electrons and become the ion ________ an atom of Al will __________ ___ electrons and become the ion ________ an atom of Se will __________ ___ electrons and become the ion ________ an atom of N ...
Chemistry 11th
... (iii) Discrimination will be removed from our society, all the people would have same status. It will be an ideal situation but then nobody will try to work of lower levels. 24. Distillation means the conversion of a liquid into vapours, which on condensation by cooling gives ...
... (iii) Discrimination will be removed from our society, all the people would have same status. It will be an ideal situation but then nobody will try to work of lower levels. 24. Distillation means the conversion of a liquid into vapours, which on condensation by cooling gives ...
CHAPTER 2 ATOMS, MOLECULES, AND IONS Questions
... nucleus itself is composed of neutrons and protons. Neutrons have a mass slightly larger than that of a proton and have no charge. Protons, on the other hand, have a 1+ relative charge as compared to the 1– charged electrons; the electrons move about the nucleus at relatively large distances. The vo ...
... nucleus itself is composed of neutrons and protons. Neutrons have a mass slightly larger than that of a proton and have no charge. Protons, on the other hand, have a 1+ relative charge as compared to the 1– charged electrons; the electrons move about the nucleus at relatively large distances. The vo ...
CHAPTER 2 ATOMS, MOLECULES, AND IONS Questions
... nucleus itself is composed of neutrons and protons. Neutrons have a mass slightly larger than that of a proton and have no charge. Protons, on the other hand, have a 1+ relative charge as compared to the 1– charged electrons; the electrons move about the nucleus at relatively large distances. The vo ...
... nucleus itself is composed of neutrons and protons. Neutrons have a mass slightly larger than that of a proton and have no charge. Protons, on the other hand, have a 1+ relative charge as compared to the 1– charged electrons; the electrons move about the nucleus at relatively large distances. The vo ...
CHAPTER 4: AQUEOUS REACTIONS AND SOLUTION
... called the solvent. Water is considered the universal solvent because of its ability to dissolve many substances. The other dissolved substances are called the solutes. A solvent dissolves a solute. ...
... called the solvent. Water is considered the universal solvent because of its ability to dissolve many substances. The other dissolved substances are called the solutes. A solvent dissolves a solute. ...
Congratulations! You have signed up for AP Chemistry for this year
... AP stands for “Advanced Placement” and AP Chemistry is the equivalent of a college chemistry class. There are some chemistry principles that are important that you remember from Pre-AP Chemistry, so I have compiled some practice notes and assignments that will equip you for the beginning of the year ...
... AP stands for “Advanced Placement” and AP Chemistry is the equivalent of a college chemistry class. There are some chemistry principles that are important that you remember from Pre-AP Chemistry, so I have compiled some practice notes and assignments that will equip you for the beginning of the year ...
Chemistry 11 – Course Review
... Chemistry 11 – Course Review Note: This review is meant for practice purposes. There are multiple outcomes that have not been covered in this booklet, which should be part of your personal review. ...
... Chemistry 11 – Course Review Note: This review is meant for practice purposes. There are multiple outcomes that have not been covered in this booklet, which should be part of your personal review. ...
Ch 8 Lecture Notes
... Microorganisms called denitrifying bacteria that grow in waterlogged soil convert NO3- ions into N2O gas as they feed on dead plant tissue (empirical formula = CH2O), converting it into CO2 and H2O. Write a balanced net ionic equation describing this conversion of dissolved nitrates to N 2O gas. Ass ...
... Microorganisms called denitrifying bacteria that grow in waterlogged soil convert NO3- ions into N2O gas as they feed on dead plant tissue (empirical formula = CH2O), converting it into CO2 and H2O. Write a balanced net ionic equation describing this conversion of dissolved nitrates to N 2O gas. Ass ...
Chapter 7 – Chemical Formulas and Chemical
... Before we start talking about chemical formulas we need to talk about polyatomic ions. These are groups of atoms bonded together covalently. What makes them different is that they carry a charge as a group. Ex: OH-, SO4-2, NH4+ These polyatomic atoms like many other formulas have common names that d ...
... Before we start talking about chemical formulas we need to talk about polyatomic ions. These are groups of atoms bonded together covalently. What makes them different is that they carry a charge as a group. Ex: OH-, SO4-2, NH4+ These polyatomic atoms like many other formulas have common names that d ...
CHAPTER 8 PERIODIC RELATIONSHIPS AMONG THE ELEMENTS
... Hence, when a cation is formed from an atom of a transition metal, electrons are always removed first from the ns orbital and then from the (n−1)d orbitals if necessary. Since the metal ion has a +3 charge, three electrons have been removed. Since the 4s subshell is less stable than the 3d, two elec ...
... Hence, when a cation is formed from an atom of a transition metal, electrons are always removed first from the ns orbital and then from the (n−1)d orbitals if necessary. Since the metal ion has a +3 charge, three electrons have been removed. Since the 4s subshell is less stable than the 3d, two elec ...
Chemistry 11 – Course Review
... an atom of Sr will __________ ___ electrons and become the ion ________ an atom of As will __________ ___ electrons and become the ion ________ an atom of Al will __________ ___ electrons and become the ion ________ an atom of Se will __________ ___ electrons and become the ion ________ an atom of N ...
... an atom of Sr will __________ ___ electrons and become the ion ________ an atom of As will __________ ___ electrons and become the ion ________ an atom of Al will __________ ___ electrons and become the ion ________ an atom of Se will __________ ___ electrons and become the ion ________ an atom of N ...
File
... All metals (on the left side of the periodic table) form cations and nonmetals (on the left side of the periodic table) form anions primarily. In order to determine the formula of the compound they create you must make sure their ions sum to zero. For example, table salt is sodium chloride. Using th ...
... All metals (on the left side of the periodic table) form cations and nonmetals (on the left side of the periodic table) form anions primarily. In order to determine the formula of the compound they create you must make sure their ions sum to zero. For example, table salt is sodium chloride. Using th ...
Solution Preparation Final Goueth
... 33. Which Group III element is expected to have physical and chemical properties that are the least similar to the other elements in that family? (A) B ...
... 33. Which Group III element is expected to have physical and chemical properties that are the least similar to the other elements in that family? (A) B ...
Complex forming reactions and complexometry Complex forming
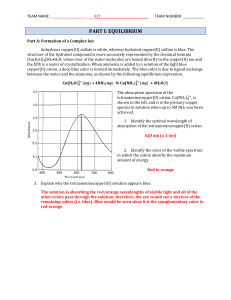
... The following conditions need to be met for the successful use of a complex reaction in titrimetry: • the complex needs to be very stable, with a high formation constant so the reaction is stochiometric (chelates) • the solution needs to be buffered • the complex needs to be formed in 1:1 ratio (for ...
... The following conditions need to be met for the successful use of a complex reaction in titrimetry: • the complex needs to be very stable, with a high formation constant so the reaction is stochiometric (chelates) • the solution needs to be buffered • the complex needs to be formed in 1:1 ratio (for ...
First Semester Final Review
... a. Mg(OH)2(s) b. (NH4)2CO3(s) c. CuSO4(s) d. (NH4)2SO4(s) e. Sr(NO3)2(s) 45. In which of the following processes are covalent bonds broken? a. I2(s) I2(g) b. CO2(s) CO2(g) c. NaCl(s) NaCl(l) d. C(diamond) C(g) e. Fe(s) Fe(l) 46. What is the final concentration of barium ions, [Ba2+], in so ...
... a. Mg(OH)2(s) b. (NH4)2CO3(s) c. CuSO4(s) d. (NH4)2SO4(s) e. Sr(NO3)2(s) 45. In which of the following processes are covalent bonds broken? a. I2(s) I2(g) b. CO2(s) CO2(g) c. NaCl(s) NaCl(l) d. C(diamond) C(g) e. Fe(s) Fe(l) 46. What is the final concentration of barium ions, [Ba2+], in so ...
Chapter 2: Atoms, Molecules and Ions
... • Understand the atomic theory of matter • Understand and be able to discuss/describe the history of the discovery of subatomic particles • Understand the structure of the atom, atomic number and mass number • Understand atomic mass, isotopes and be able to calculate average atomic mass. • Become mo ...
... • Understand the atomic theory of matter • Understand and be able to discuss/describe the history of the discovery of subatomic particles • Understand the structure of the atom, atomic number and mass number • Understand atomic mass, isotopes and be able to calculate average atomic mass. • Become mo ...
Lab 1
... look unlike anything else. In this experiment, you will describe the physical properties of elements in a laboratory display and determine the location of elements on a blank periodic table. A. Physical Properties of Elements Metals are elements that are usually shiny or have a metallic luster. They ...
... look unlike anything else. In this experiment, you will describe the physical properties of elements in a laboratory display and determine the location of elements on a blank periodic table. A. Physical Properties of Elements Metals are elements that are usually shiny or have a metallic luster. They ...
1 of 52
... The following questions represent the baseline knowledge you will need to succeed in AP Chemistry. The goal of this assignment is twofold. First, it will tell you what you are up against in the upcoming school year. Second, this assignment covers curriculum that is basic and will allow us the time t ...
... The following questions represent the baseline knowledge you will need to succeed in AP Chemistry. The goal of this assignment is twofold. First, it will tell you what you are up against in the upcoming school year. Second, this assignment covers curriculum that is basic and will allow us the time t ...
chapter 7-Chemical Bonding
... – Examples: • Na+ sodium ion, Ca2+, Al3+ -- cations • Cl- chloride ion, O2-, N3- -- anions ...
... – Examples: • Na+ sodium ion, Ca2+, Al3+ -- cations • Cl- chloride ion, O2-, N3- -- anions ...
Chemistry 11 – Course Review
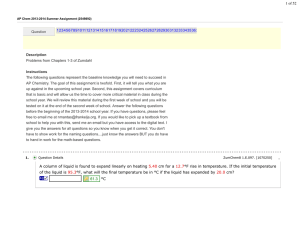
... Unit 9 – Solution Chemistry 1. What is the concentration of NO3- in a solution of 0.325M Al(NO3)3 (aq)? ...
... Unit 9 – Solution Chemistry 1. What is the concentration of NO3- in a solution of 0.325M Al(NO3)3 (aq)? ...
File
... • Add solid base to acid (gently heat to speed up metal/carbonate to ensure all acid reaction reacts/neutralises and that the • Filter off excess solid base product is neutral • Heat filtrate solution until volume reduced by half • Leave solution to cool and allow remaining The percentage yield of c ...
... • Add solid base to acid (gently heat to speed up metal/carbonate to ensure all acid reaction reacts/neutralises and that the • Filter off excess solid base product is neutral • Heat filtrate solution until volume reduced by half • Leave solution to cool and allow remaining The percentage yield of c ...
Chemistry Review 1 Answer Key
... Carbon and oxygen are examples of elements that exist in more than one form in the same phase. Graphite and diamond are two crystalline arrangements for carbon. The crystal structure of graphite is organized in layers. The bonds between carbon atoms within each layer of graphite are strong. The bond ...
... Carbon and oxygen are examples of elements that exist in more than one form in the same phase. Graphite and diamond are two crystalline arrangements for carbon. The crystal structure of graphite is organized in layers. The bonds between carbon atoms within each layer of graphite are strong. The bond ...
Topic_4
... in scientific notation, a mole is 6.02 x 1023 particles. Scientific notation is used to express very small or very large measurements in powers of ten. It expresses quantities by using a number between one and ten, which is then multiplied by ten to a power to give the quantity its proper magnitude. ...
... in scientific notation, a mole is 6.02 x 1023 particles. Scientific notation is used to express very small or very large measurements in powers of ten. It expresses quantities by using a number between one and ten, which is then multiplied by ten to a power to give the quantity its proper magnitude. ...
Answers - Scioly.org
... 20. The student concludes that she has synthesized ethyl butanoate. Use evidence from the two experiments to support or to refute her claim. The peak of highest mass to charge ratio is approximately 116; therefore, the unknown molecule would have a molecular mass of 116. Ethyl butanoate has the chem ...
... 20. The student concludes that she has synthesized ethyl butanoate. Use evidence from the two experiments to support or to refute her claim. The peak of highest mass to charge ratio is approximately 116; therefore, the unknown molecule would have a molecular mass of 116. Ethyl butanoate has the chem ...
Tutorial 4 (PowerPoint)
... • Tip can be operated in “dynamic mode” • The tip and cantilever (beam with the tip on it) have a mechanical natural resonance • The resonance will change as external forces from the sample are exerted on it • The tip’s vibration amplitude must be much less than the distance between it and the sampl ...
... • Tip can be operated in “dynamic mode” • The tip and cantilever (beam with the tip on it) have a mechanical natural resonance • The resonance will change as external forces from the sample are exerted on it • The tip’s vibration amplitude must be much less than the distance between it and the sampl ...
Inductively coupled plasma mass spectrometry

Inductively coupled plasma mass spectrometry (ICP-MS) is a type of mass spectrometry which is capable of detecting metals and several non-metals at concentrations as low as one part in 1015 (part per quadrillion, ppq) on non-interfered low-background isotopes. This is achieved by ionizing the sample with inductively coupled plasma and then using a mass spectrometer to separate and quantify those ions.Compared to atomic absorption techniques, ICP-MS has greater speed, precision, and sensitivity. However, compared with other types of mass spectrometry, such as Thermal ionization mass spectrometry (TIMS) and Glow Discharge Mass Spectrometry (GD-MS), ICP-MS introduces a lot of interfering species: argon from the plasma, component gasses of air that leak through the cone orifices, and contamination from glassware and the cones.The variety of applications exceeds that of inductively coupled plasma atomic emission spectroscopy and includes isotopic speciation. Due to possible applications in nuclear technologies, ICP-MS hardware is a subject for special exporting regulations.