Subject Area Assessment Guides
... electronegativities can form covalent bonds to electrons to form ionic bonds. become molecules. In a covalent bond, therefore, bonding electron pairs are localized in the region between the bonded atoms. In metals valence electrons are not localized to individual atoms but are free to move to tempor ...
... electronegativities can form covalent bonds to electrons to form ionic bonds. become molecules. In a covalent bond, therefore, bonding electron pairs are localized in the region between the bonded atoms. In metals valence electrons are not localized to individual atoms but are free to move to tempor ...
Environmental Analysis - Evergreen State College Archives
... American Society for Testing and Materials (ASTM). These methods are specific for use with certain analytes in certain types of samples. They commonly define both the sample handling requirements and the analytical techniques that are to be used. Some methods require calibration. This involves makin ...
... American Society for Testing and Materials (ASTM). These methods are specific for use with certain analytes in certain types of samples. They commonly define both the sample handling requirements and the analytical techniques that are to be used. Some methods require calibration. This involves makin ...
CHAPTER 2: ATOMS, MOLECULES AND IONS ULES AND IONS
... diffuse cloud of positive charge with the negative electrons embedded randomly in it”. This model is called as palm-pudding model. ...
... diffuse cloud of positive charge with the negative electrons embedded randomly in it”. This model is called as palm-pudding model. ...
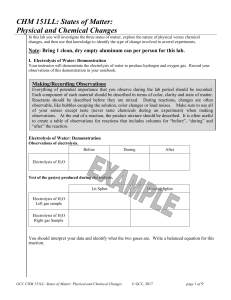
CHM 151LL: States of Matter: Physical and Chemical Changes
... II. Separation Methods A mixture is matter that contains two or more chemicals (atoms, molecules or compounds) that are combined, and that have not reacted with each other to form a pure substance. Mixtures can be separated into their individual compounds by physical methods, that is, by methods th ...
... II. Separation Methods A mixture is matter that contains two or more chemicals (atoms, molecules or compounds) that are combined, and that have not reacted with each other to form a pure substance. Mixtures can be separated into their individual compounds by physical methods, that is, by methods th ...
Introduction to Stoichiometry
... What is Stoichiometry? The proportional relationship between two or more substances during a chemical reaction. In other words, using dimensional analysis to convert one substance to another There are many different types, but they are all similar. So, let’s start small. How small? ...
... What is Stoichiometry? The proportional relationship between two or more substances during a chemical reaction. In other words, using dimensional analysis to convert one substance to another There are many different types, but they are all similar. So, let’s start small. How small? ...
Chemistry II Exams and Keys 2014 Season
... 11. A cylinder of unknown volume contains neon gas, Ne(g), at 4.0 atm and 400 K. The neon gas is then transferred to a 10.0 L gas cylinder containing Ar(g), at 6.0 atm and 400 K. If the final total pressure at 400 K is 9.0 atm, then what is the volume of the cylinder that initially contained the neo ...
... 11. A cylinder of unknown volume contains neon gas, Ne(g), at 4.0 atm and 400 K. The neon gas is then transferred to a 10.0 L gas cylinder containing Ar(g), at 6.0 atm and 400 K. If the final total pressure at 400 K is 9.0 atm, then what is the volume of the cylinder that initially contained the neo ...
GCSE ADDITIONAL CHEMISTRY (C2) REVISION BOOKLET
... 1 a) The centre of an atom is called the nucleus. b) In the centre of the atom there are two types of sub-atomic particles. These particles are the proton and the neutron. The protons are positively charged. The neutrons have no charge. They are neutral. c) The centre of the atom is positively charg ...
... 1 a) The centre of an atom is called the nucleus. b) In the centre of the atom there are two types of sub-atomic particles. These particles are the proton and the neutron. The protons are positively charged. The neutrons have no charge. They are neutral. c) The centre of the atom is positively charg ...
Document
... (a) It is possible to identify the sulfite ion without memorizing all the ions in Table 2.4. If you remember the name and formula of one of the sulfur–oxygen polyatomic anions, you should be able to deduce the names of others. Suppose you remember that sulfate is SO42–. The -ite anion has one fewer ...
... (a) It is possible to identify the sulfite ion without memorizing all the ions in Table 2.4. If you remember the name and formula of one of the sulfur–oxygen polyatomic anions, you should be able to deduce the names of others. Suppose you remember that sulfate is SO42–. The -ite anion has one fewer ...
Plasma Surface Technology - Diener electronic GmbH + Co. KG
... Excited atoms where the electrons have not left the atom, but were excited to a higher energy level ...
... Excited atoms where the electrons have not left the atom, but were excited to a higher energy level ...
Introduction to Chemical Bonding
... The bond of Sodium and Fluorine is an example of Ionic bonding: electrons have been transferred in order for the atoms to have a full outer level. When an atom loses or gains electrons, it becomes what is called an ion. An ion is no longer neutrally charged because it has different numbers of proton ...
... The bond of Sodium and Fluorine is an example of Ionic bonding: electrons have been transferred in order for the atoms to have a full outer level. When an atom loses or gains electrons, it becomes what is called an ion. An ion is no longer neutrally charged because it has different numbers of proton ...
Note Sheets and Sample Problems
... Chemical Foundations Chemistry: An Overview • Matter – takes up space, has mass, exhibits inertia - composed of atoms only 100 or so different types - Water made up of one oxygen and two hydrogen atoms - Pass an electric current through it to separate the two types of atoms and they rearrange to bec ...
... Chemical Foundations Chemistry: An Overview • Matter – takes up space, has mass, exhibits inertia - composed of atoms only 100 or so different types - Water made up of one oxygen and two hydrogen atoms - Pass an electric current through it to separate the two types of atoms and they rearrange to bec ...
atom - Zanichelli online per la scuola
... Atoms have a tiny, dense nucleus with a positive charge. The nucleus is made up of protons and neutrons and is surrounded by an empty space in which electrons move. Nuclear force keeps protons and neutrons together inside the nucleus, overcoming the mutual repulsion caused by the positive charges. ...
... Atoms have a tiny, dense nucleus with a positive charge. The nucleus is made up of protons and neutrons and is surrounded by an empty space in which electrons move. Nuclear force keeps protons and neutrons together inside the nucleus, overcoming the mutual repulsion caused by the positive charges. ...
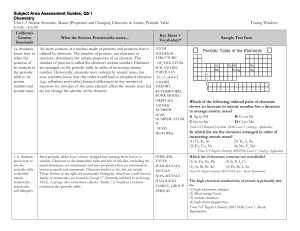
Subject Area Assessment Guides
... element from Group 2 will most often combine with two atoms of an element from Group 17 (e.g., MgCl2) because Group 2 elements have two electrons available for bonding, and Group 17 elements have only one electron position open in the outermost energy level. (Note that some periodic tables indicate ...
... element from Group 2 will most often combine with two atoms of an element from Group 17 (e.g., MgCl2) because Group 2 elements have two electrons available for bonding, and Group 17 elements have only one electron position open in the outermost energy level. (Note that some periodic tables indicate ...
eBook AQA GCSE Chemistry Unit C2 Part 1
... a noble gas, with eight electrons in its highest energy level. A hydrogen atom has just one electron. It needs one more electron to achieve the stable electronic structure of the noble gas helium. When nitrogen and hydrogen react to form ammonia, the two types of atom join together by sharing pairs ...
... a noble gas, with eight electrons in its highest energy level. A hydrogen atom has just one electron. It needs one more electron to achieve the stable electronic structure of the noble gas helium. When nitrogen and hydrogen react to form ammonia, the two types of atom join together by sharing pairs ...
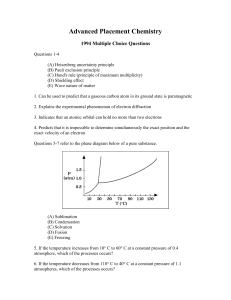
Advanced Placement Chemistry
... (C) Hund's rule (principle of maximum multiplicity) (D) Shielding effect (E) Wave nature of matter 1. Can be used to predict that a gaseous carbon atom in its ground state is paramagnetic 2. Explains the experimental phenomenon of electron diffraction 3. Indicates that an atomic orbital can hold no ...
... (C) Hund's rule (principle of maximum multiplicity) (D) Shielding effect (E) Wave nature of matter 1. Can be used to predict that a gaseous carbon atom in its ground state is paramagnetic 2. Explains the experimental phenomenon of electron diffraction 3. Indicates that an atomic orbital can hold no ...
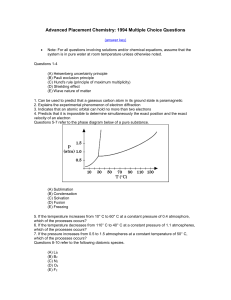
MC94 - Southchemistry.com
... Advanced Placement Chemistry: 1994 Multiple Choice Questions (answer key) ...
... Advanced Placement Chemistry: 1994 Multiple Choice Questions (answer key) ...
Plasma Clean to Reduce Wire Bond Failures
... present in the molding compound used in the device package. By a somewhat complicated mechanism known as the Horsting Effect and, like Kirkendall voiding, a consequence of the diffusion of one metal into the other, the halogens become ...
... present in the molding compound used in the device package. By a somewhat complicated mechanism known as the Horsting Effect and, like Kirkendall voiding, a consequence of the diffusion of one metal into the other, the halogens become ...
FE Exam review for Chemistry
... How are they affected by changing temperature & pressure? As temperature increases: solid liquid gas As pressure increases: gas liquid solid How do they differ in terms of: Solids > liquids > gases • density (abundance) Gas > liquid > solids • energy / movement • shape & compressibility Only ...
... How are they affected by changing temperature & pressure? As temperature increases: solid liquid gas As pressure increases: gas liquid solid How do they differ in terms of: Solids > liquids > gases • density (abundance) Gas > liquid > solids • energy / movement • shape & compressibility Only ...
1. formulae equations and amount
... Add solid base to acid (gently heat to speed up reaction Filter off excess solid base Heat filtrate solution until volume reduced by half Leave solution to cool and allow remaining water to evaporate Slowly and crystals to form Filter or pick out crystals Leave to dry and put crystals between filter ...
... Add solid base to acid (gently heat to speed up reaction Filter off excess solid base Heat filtrate solution until volume reduced by half Leave solution to cool and allow remaining water to evaporate Slowly and crystals to form Filter or pick out crystals Leave to dry and put crystals between filter ...
Stoichiometry We compare all other elements to the known mass of
... Calculating empirical and molecular formulas: empirical formulas represent the simplest or smallest m o l a r ratio of elements within a compound while molecular formulas represent the actual numbers of elements within a compound. The empirical mass is the least common multiple of the molar mass. Ex ...
... Calculating empirical and molecular formulas: empirical formulas represent the simplest or smallest m o l a r ratio of elements within a compound while molecular formulas represent the actual numbers of elements within a compound. The empirical mass is the least common multiple of the molar mass. Ex ...
111 Exam I Outline
... Key: You must have a balanced equation!! How many grams of chromic chloride reacts with 6.0 mole Cr? ...
... Key: You must have a balanced equation!! How many grams of chromic chloride reacts with 6.0 mole Cr? ...
111 Exam I Outline
... Key: You must have a balanced equation!! How many grams of chromic chloride reacts with 6.0 mole Cr? ...
... Key: You must have a balanced equation!! How many grams of chromic chloride reacts with 6.0 mole Cr? ...
AQA Additional Sci C2 Revision Guide
... outer shell of its atoms. Elements in groups 1 and 2 of the periodic table only have 1 or 2 electrons in their outer shells so these form positive ions by losing their outer electrons. Elements in groups 6 and 7 of the periodic table only need 1 or 2 electrons to fill up their outer shells so these ...
... outer shell of its atoms. Elements in groups 1 and 2 of the periodic table only have 1 or 2 electrons in their outer shells so these form positive ions by losing their outer electrons. Elements in groups 6 and 7 of the periodic table only need 1 or 2 electrons to fill up their outer shells so these ...
Unit 3 Notes
... Subsection (c) Calculations in reactions that involve solutions. QUESTIONS ON MOLES OF IONS IN SOLUTIONS In these questions you do not have to work out how many ions there are. You just need to understand the idea of mole ratios in formulae. e.g. A solution contains a mixture of calcium nitrate and ...
... Subsection (c) Calculations in reactions that involve solutions. QUESTIONS ON MOLES OF IONS IN SOLUTIONS In these questions you do not have to work out how many ions there are. You just need to understand the idea of mole ratios in formulae. e.g. A solution contains a mixture of calcium nitrate and ...
Inductively coupled plasma mass spectrometry

Inductively coupled plasma mass spectrometry (ICP-MS) is a type of mass spectrometry which is capable of detecting metals and several non-metals at concentrations as low as one part in 1015 (part per quadrillion, ppq) on non-interfered low-background isotopes. This is achieved by ionizing the sample with inductively coupled plasma and then using a mass spectrometer to separate and quantify those ions.Compared to atomic absorption techniques, ICP-MS has greater speed, precision, and sensitivity. However, compared with other types of mass spectrometry, such as Thermal ionization mass spectrometry (TIMS) and Glow Discharge Mass Spectrometry (GD-MS), ICP-MS introduces a lot of interfering species: argon from the plasma, component gasses of air that leak through the cone orifices, and contamination from glassware and the cones.The variety of applications exceeds that of inductively coupled plasma atomic emission spectroscopy and includes isotopic speciation. Due to possible applications in nuclear technologies, ICP-MS hardware is a subject for special exporting regulations.