Writing Chemical Equations KClO3 O2 (g) + KCl (s) Balancing
... In this example we started with 2 atoms of hydrogen and we ended with 2 atoms of hydrogen. But we started with only one atom of oxygen and ended with 2 atoms of oxygen. We cannot change the numbers at the bottom of the symbols because that is how the formulas must be written. To balance the equation ...
... In this example we started with 2 atoms of hydrogen and we ended with 2 atoms of hydrogen. But we started with only one atom of oxygen and ended with 2 atoms of oxygen. We cannot change the numbers at the bottom of the symbols because that is how the formulas must be written. To balance the equation ...
Chapter 6-student notes
... Can you figure out what is missing in the following chemical reactions? 1. Aluminum resists corrosion (rust) because it reacts with a gas in the air to form a protective coating of aluminum oxide. Aluminum + ____________-> aluminum oxide 2. Zinc Metal, used as a coating in galvanized iron, also reac ...
... Can you figure out what is missing in the following chemical reactions? 1. Aluminum resists corrosion (rust) because it reacts with a gas in the air to form a protective coating of aluminum oxide. Aluminum + ____________-> aluminum oxide 2. Zinc Metal, used as a coating in galvanized iron, also reac ...
Date Hour
... Aqueous solution (a metal salt dissolved in water or an acid or base). The word solution is your clue that you are to use the subscript (aq). ...
... Aqueous solution (a metal salt dissolved in water or an acid or base). The word solution is your clue that you are to use the subscript (aq). ...
Chem 1100 Chapter Three Study Guide Outline I. Molar Mass and
... 26. How many moles of CuO can be produced from 0.450 mol of Cu2O in the following reaction? 2 Cu2O (s) + O2 (g) Æ 4 CuO (s) a. 1.80 mol b. 0.225 mol c. 0.900 mol d. 0.450 mol 27. 10 g of nitrogen is reacted with 5.0 g of hydrogen to produce ammonia according to the chemical equation shown below. Whi ...
... 26. How many moles of CuO can be produced from 0.450 mol of Cu2O in the following reaction? 2 Cu2O (s) + O2 (g) Æ 4 CuO (s) a. 1.80 mol b. 0.225 mol c. 0.900 mol d. 0.450 mol 27. 10 g of nitrogen is reacted with 5.0 g of hydrogen to produce ammonia according to the chemical equation shown below. Whi ...
Final Exam Review – Free Response Section Name: 1. A sample of
... If it is a single displacement reaction, use the activity series to determine if it will occur. If it will not occur, write “no reaction.” If it is a double displacement, use the solubility rules to determine if it will occur. If it will not occur, write “no reaction.” If it will occur, write the ba ...
... If it is a single displacement reaction, use the activity series to determine if it will occur. If it will not occur, write “no reaction.” If it is a double displacement, use the solubility rules to determine if it will occur. If it will not occur, write “no reaction.” If it will occur, write the ba ...
Chemistry 221 - Oregon State chemistry
... with water molecules, so making it highly soluble in water. This allows the glucose fuel to be transported easily within biological systems, for example in the bloodstream of animals or the sap of plants. An adult has 5-6 grams of glucose in the blood (about 1 teaspoon), which will supply the body' ...
... with water molecules, so making it highly soluble in water. This allows the glucose fuel to be transported easily within biological systems, for example in the bloodstream of animals or the sap of plants. An adult has 5-6 grams of glucose in the blood (about 1 teaspoon), which will supply the body' ...
Standard B-2
... Enzymatic proteins accelerate the speed of chemical reactions such as digestive enzymes Carbohydrates: sugars and starches; composed of carbon, hydrogen, and oxygen; main source of energy for the cell and used to store energy for short periods of time; caloric value of carbs depend on the number o ...
... Enzymatic proteins accelerate the speed of chemical reactions such as digestive enzymes Carbohydrates: sugars and starches; composed of carbon, hydrogen, and oxygen; main source of energy for the cell and used to store energy for short periods of time; caloric value of carbs depend on the number o ...
File
... Its a molecule made of a metal and one or more non-metals. In this type of compounds, there is a transfer of e-→ The metal loses e- and the non-metals gains e- to form an ionic bond. Physical Properties ...
... Its a molecule made of a metal and one or more non-metals. In this type of compounds, there is a transfer of e-→ The metal loses e- and the non-metals gains e- to form an ionic bond. Physical Properties ...
Chemistry of Life
... • Oxygen and Hydrogen are bonded together by ________ electrons, but the Oxygen atom _________the shared electrons closer to it, creating negative and positive sides of the water molecules. Water has a partial negative charge due to the extra unshared e- that Oxygen and a partial + charge near the h ...
... • Oxygen and Hydrogen are bonded together by ________ electrons, but the Oxygen atom _________the shared electrons closer to it, creating negative and positive sides of the water molecules. Water has a partial negative charge due to the extra unshared e- that Oxygen and a partial + charge near the h ...
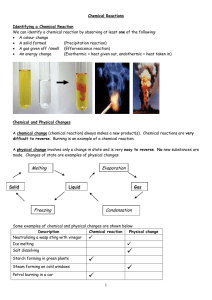
Chemical Reactions
... The reactants are separated from each other by a plus sign and the products are separated from each other by a plus sign. There should be an arrow in the middle. Examples: When sodium is mixed with water, a purple alkaline solution of sodium hydroxide is produced and hydrogen gas is evolved. Sodium ...
... The reactants are separated from each other by a plus sign and the products are separated from each other by a plus sign. There should be an arrow in the middle. Examples: When sodium is mixed with water, a purple alkaline solution of sodium hydroxide is produced and hydrogen gas is evolved. Sodium ...
Chapter 6: Chemistry in Biology
... Substances that release hydrogen ions ( H ) when dissolved in water are called __________. Substances that release hydroxide ions ( OH ) when dissolved in water are called __________. pH and Buffers: The measure of concentration of H in a solution is called __________. ...
... Substances that release hydrogen ions ( H ) when dissolved in water are called __________. Substances that release hydroxide ions ( OH ) when dissolved in water are called __________. pH and Buffers: The measure of concentration of H in a solution is called __________. ...
1) COMBINATION REACTION
... REACTING WITH LEAD (II) NITRATE TO FORM LEAD (II) IODIDE AND POTASSIUM NITRATE: 2 KI + Pb(NO3)2 2 KNO3 ...
... REACTING WITH LEAD (II) NITRATE TO FORM LEAD (II) IODIDE AND POTASSIUM NITRATE: 2 KI + Pb(NO3)2 2 KNO3 ...
HYDROGEN PEROXIDE 20%-50%
... Special fire fighting procedures: Evacuate all non-essential personnel. Intervention only by capable personnel who are trained and aware of hazards of the product. Wear self-contained breathing apparatus when in close proximity or in confined spaces. When intervention in close proximity, wear acid ...
... Special fire fighting procedures: Evacuate all non-essential personnel. Intervention only by capable personnel who are trained and aware of hazards of the product. Wear self-contained breathing apparatus when in close proximity or in confined spaces. When intervention in close proximity, wear acid ...
What are reactions?
... __________. This can be a sign that a chemical __________ has happened. 2. Other signs of a chemical reaction might be an increase in temperature if _____ is released or a change in ________. 3. Physical changes like _________ do not make new materials and are easily _________ eg, by cooling which c ...
... __________. This can be a sign that a chemical __________ has happened. 2. Other signs of a chemical reaction might be an increase in temperature if _____ is released or a change in ________. 3. Physical changes like _________ do not make new materials and are easily _________ eg, by cooling which c ...
What are reactions? - UTLNET Secure Site
... __________. This can be a sign that a chemical __________ has happened. 2. Other signs of a chemical reaction might be an increase in temperature if _____ is released or a change in ________. 3. Physical changes like _________ do not make new materials and are easily _________ eg, by cooling which c ...
... __________. This can be a sign that a chemical __________ has happened. 2. Other signs of a chemical reaction might be an increase in temperature if _____ is released or a change in ________. 3. Physical changes like _________ do not make new materials and are easily _________ eg, by cooling which c ...
Chemical Equations
... Synthesis Reaction in which 2 or more reactants combine to form a new compound. A+B ...
... Synthesis Reaction in which 2 or more reactants combine to form a new compound. A+B ...
Water splitting

Water splitting is the general term for a chemical reaction in which water is separated into oxygen and hydrogen. Efficient and economical water splitting would be a key technology component of a hydrogen economy. Various techniques for water splitting have been issued in water splitting patents in the United States. In photosynthesis, water splitting donates electrons to power the electron transport chain in photosystem II.