5 Steps to a 5 AP Chemistry, 2008-2009 Edition
... Not too long ago, you enrolled in AP Chemistry. A curiosity about chemistry, encouragement from a respected teacher, or the simple fact that it was a requirement may have been your motivation. No matter what the reason, you find yourself flipping through a book, which promises to help you culminate ...
... Not too long ago, you enrolled in AP Chemistry. A curiosity about chemistry, encouragement from a respected teacher, or the simple fact that it was a requirement may have been your motivation. No matter what the reason, you find yourself flipping through a book, which promises to help you culminate ...
Stoichiometric Calculations
... Limiting reagent problems are worded differently because the quantities of both reactants are given. 10 moles of H2 and 20 moles of Cl2 react to produce HCl. Which quantity is the limiting reagent? It is your job to figure out which reactant is limiting because that will determine the maximum amount ...
... Limiting reagent problems are worded differently because the quantities of both reactants are given. 10 moles of H2 and 20 moles of Cl2 react to produce HCl. Which quantity is the limiting reagent? It is your job to figure out which reactant is limiting because that will determine the maximum amount ...
Stoichiometric Calculations
... The limiting reactant, or limiting reagent, is the reactant present in the smallest stoichiometric amount. This is not necessarily the one with the smallest mass. The limiting reactant is the reactant you’ll run out of first, and it is the one that determines the maximum amount of product that can b ...
... The limiting reactant, or limiting reagent, is the reactant present in the smallest stoichiometric amount. This is not necessarily the one with the smallest mass. The limiting reactant is the reactant you’ll run out of first, and it is the one that determines the maximum amount of product that can b ...
Chapter 1 - Solutions
... reactant in predicting the amount of product obtained in a reaction? Can there be a limiting reactant if only one reactant is present? The limiting reactant is the reactant that first runs out in a chemical reaction, therefore limiting the amount of products that can be formed. Excess reactants refe ...
... reactant in predicting the amount of product obtained in a reaction? Can there be a limiting reactant if only one reactant is present? The limiting reactant is the reactant that first runs out in a chemical reaction, therefore limiting the amount of products that can be formed. Excess reactants refe ...
Part 1-ICHO-21-25
... atmospheric liquid water pool of 5000 m and fully returned on earth as rain, what is the expected pH of the condensed water? ...
... atmospheric liquid water pool of 5000 m and fully returned on earth as rain, what is the expected pH of the condensed water? ...
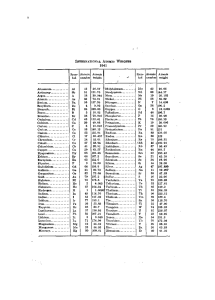
INTEKNATIONAL ATOMIC WEIGHTS Aluminum... Antimony..., Argon
... find interesting things to do under "Suggestions for Further Work"; when it is too long, the instructor will select parts of the "Procedure" which may be omitted. Several of the procedures have been expanded and the directions made more specific without sacrificing the development of self-reUance on ...
... find interesting things to do under "Suggestions for Further Work"; when it is too long, the instructor will select parts of the "Procedure" which may be omitted. Several of the procedures have been expanded and the directions made more specific without sacrificing the development of self-reUance on ...
IB Chemistry Online SAQ_Ans
... between 0 and n − 1. If n = 3, for example, l can be either 0, 1, or 2. The magnetic quantum number (m) can be any integer between −l and +l. If l = 2, m can be either −2, −1, 0, +1, or +2. ...
... between 0 and n − 1. If n = 3, for example, l can be either 0, 1, or 2. The magnetic quantum number (m) can be any integer between −l and +l. If l = 2, m can be either −2, −1, 0, +1, or +2. ...
CHAPTER 4 REACTIONS IN AQUEOUS SOLUTIONS
... and weak bases are weak electrolytes. They only ionize to a small extent in solution. Weak acids and weak bases are shown as molecules in ionic and net ionic equations. A net ionic equation shows only the species that actually take part in the reaction. ...
... and weak bases are weak electrolytes. They only ionize to a small extent in solution. Weak acids and weak bases are shown as molecules in ionic and net ionic equations. A net ionic equation shows only the species that actually take part in the reaction. ...
chapter 5 gases
... number of ions and therefore the solution conducts electricity. A weak electrolyte solution contains a small number of ions, so the solution conducts electricity, but not as strongly as a strong electrolyte. ...
... number of ions and therefore the solution conducts electricity. A weak electrolyte solution contains a small number of ions, so the solution conducts electricity, but not as strongly as a strong electrolyte. ...
10. Solution Guide to Supplementary Exercises
... Each question (Questions 56 – 60) consists of two separate statements. Decide whether each of the two statements is true or false; if both are true, then decide whether or not the second statement is a correct explanation of the first statement. Then select one option from A to D according to the fo ...
... Each question (Questions 56 – 60) consists of two separate statements. Decide whether each of the two statements is true or false; if both are true, then decide whether or not the second statement is a correct explanation of the first statement. Then select one option from A to D according to the fo ...
Cliffs Notes
... Technical Editor: Christopher Bushee Production Proofreader: Joel K. Draper Hungry Minds Indianapolis Production Services ...
... Technical Editor: Christopher Bushee Production Proofreader: Joel K. Draper Hungry Minds Indianapolis Production Services ...
Stoichiometry
... 1. Write the skeletal equation 2. Balance one element at a time using coefficients – Start with the elements in the most complex substance and finish with those in the least complex one – Alternatively, start with the element present in the fewest number of formulas and finish with the element prese ...
... 1. Write the skeletal equation 2. Balance one element at a time using coefficients – Start with the elements in the most complex substance and finish with those in the least complex one – Alternatively, start with the element present in the fewest number of formulas and finish with the element prese ...
Section – B - About iTutoring
... becomes insoluble and separates as precipitates due to the common ino effect of Cl- ions. It is necessary to note that under certain situations the solubility increases instead of decreasing. The solubility of salt like phosphates increase when acid is added to their solutions or pH of the ...
... becomes insoluble and separates as precipitates due to the common ino effect of Cl- ions. It is necessary to note that under certain situations the solubility increases instead of decreasing. The solubility of salt like phosphates increase when acid is added to their solutions or pH of the ...
Stoichiometric Calculations
... moles of each substance. For instance, this formula 2 H2 + O2 → 2 H2O can be read as: 2 moles of H2 plus 1 mole of O2 yields 2 moles of H2O ...
... moles of each substance. For instance, this formula 2 H2 + O2 → 2 H2O can be read as: 2 moles of H2 plus 1 mole of O2 yields 2 moles of H2O ...
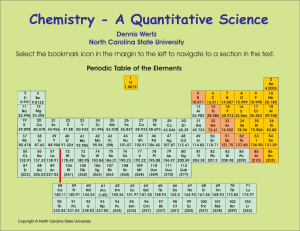
Chemistry - A Quantitative Science
... 6.0221x1023 = NA is Avogadro's number. A mole is used to indicate a number of atoms just as a dozen is used to indicate a number of eggs. Converting from moles to atoms is done the same as converting dozens to items. 1.5 doz = (1.5 doz)(12 items.doz-1) = 18 items and 1.5 mol = (1.5 mol)( 6.0x1023 at ...
... 6.0221x1023 = NA is Avogadro's number. A mole is used to indicate a number of atoms just as a dozen is used to indicate a number of eggs. Converting from moles to atoms is done the same as converting dozens to items. 1.5 doz = (1.5 doz)(12 items.doz-1) = 18 items and 1.5 mol = (1.5 mol)( 6.0x1023 at ...
CHAPTER 9
... Antoine Laurent Lavoisier was a meticulous scientist. He realized that Rutherford and Priestley had carefully observed and described their experiments but had not measured the mass of anything. Unlike his colleagues, Lavoisier knew the importance of using a balance. He measured the masses of reactan ...
... Antoine Laurent Lavoisier was a meticulous scientist. He realized that Rutherford and Priestley had carefully observed and described their experiments but had not measured the mass of anything. Unlike his colleagues, Lavoisier knew the importance of using a balance. He measured the masses of reactan ...
Chapter 4
... and weak bases are weak electrolytes. They only ionize to a small extent in solution. Weak acids and weak bases are shown as molecules in ionic and net ionic equations. A net ionic equation shows only the species that actually take part in the reaction. (a) Solution: CH3COOH is a weak acid. It will ...
... and weak bases are weak electrolytes. They only ionize to a small extent in solution. Weak acids and weak bases are shown as molecules in ionic and net ionic equations. A net ionic equation shows only the species that actually take part in the reaction. (a) Solution: CH3COOH is a weak acid. It will ...
File
... 31 The definitions of many chemical terms can be illustrated by chemical equations. Which terms can be illustrated by an equation that shows the formation of a positive ion? ...
... 31 The definitions of many chemical terms can be illustrated by chemical equations. Which terms can be illustrated by an equation that shows the formation of a positive ion? ...
Calculations and the Chemical Equation
... Relating Avogadro's number to molar mass: calculation of the mass of Avogadro's number of sodium atoms Converting moles to atoms. Converting atoms to moles. Converting moles of a substance to mass in grams. Converting kilograms to moles. Converting grams to number of atoms. Calculating formula weigh ...
... Relating Avogadro's number to molar mass: calculation of the mass of Avogadro's number of sodium atoms Converting moles to atoms. Converting atoms to moles. Converting moles of a substance to mass in grams. Converting kilograms to moles. Converting grams to number of atoms. Calculating formula weigh ...
The Acidic Environment #2
... pollution. This led to regulations to control emissions from factories, power stations and motor cars. The annual average concentration of SO2 and NO2 in most large cities around the world is 0.01 ppm for each gas. This is about 10 times the value for clean air, though a concentration of 0.01 ppm ...
... pollution. This led to regulations to control emissions from factories, power stations and motor cars. The annual average concentration of SO2 and NO2 in most large cities around the world is 0.01 ppm for each gas. This is about 10 times the value for clean air, though a concentration of 0.01 ppm ...
Chemistry – A Molecular Sciences Appendices
... Comment: Note that the two calculations are almost identical, the only difference being the term that converts moles of H2O to moles of H in part b. This extra step was necessary because we are “counting” hydrogen atoms, not water molecules. The conversion factor comes directly from the chemical for ...
... Comment: Note that the two calculations are almost identical, the only difference being the term that converts moles of H2O to moles of H in part b. This extra step was necessary because we are “counting” hydrogen atoms, not water molecules. The conversion factor comes directly from the chemical for ...
Chemical Quantities
... equation. Reactions are what chemistry is really all about. Recall from Chapter 6 that chemical changes are actually rearrangements of atom groupings that can be described by chemical equations. These chemical equations tell us the identities (formulas) of the reactants and products and also show ho ...
... equation. Reactions are what chemistry is really all about. Recall from Chapter 6 that chemical changes are actually rearrangements of atom groupings that can be described by chemical equations. These chemical equations tell us the identities (formulas) of the reactants and products and also show ho ...
PART 6-ICHO-26-30
... 1.5 In a saturated aqueous solution of CaCO3(s) pH is measured to be 9.95. Calculate the solubility of calcium carbonate in water and show that the calculated value for the solubility product constant Ksp is 5×10 . ...
... 1.5 In a saturated aqueous solution of CaCO3(s) pH is measured to be 9.95. Calculate the solubility of calcium carbonate in water and show that the calculated value for the solubility product constant Ksp is 5×10 . ...
mcdonald (pam78654) – HW 1: High School Concepts – laude
... numbers or are close enough for rounding to a whole number. The empirical formula is C3 H3 O. Next we find the molecular formula. The molecular formula gives the actual number of atoms of each element present in a molecule of the compound. We were given the formula weight of the compound, 110. We ca ...
... numbers or are close enough for rounding to a whole number. The empirical formula is C3 H3 O. Next we find the molecular formula. The molecular formula gives the actual number of atoms of each element present in a molecule of the compound. We were given the formula weight of the compound, 110. We ca ...
Thermometric titration

A thermometric titration is one of a number of instrumental titration techniques where endpoints can be located accurately and precisely without a subjective interpretation on the part of the analyst as to their location. Enthalpy change is arguably the most fundamental and universal property of chemical reactions, so the observation of temperature change is a natural choice in monitoring their progress. It is not a new technique, with possibly the first recognizable thermometric titration method reported early in the 20th century (Bell and Cowell, 1913). In spite of its attractive features, and in spite of the considerable research that has been conducted in the field and a large body of applications that have been developed; it has been until now an under-utilized technique in the critical area of industrial process and quality control. Automated potentiometric titration systems have pre-dominated in this area since the 1970s. With the advent of cheap computers able to handle the powerful thermometric titration software, development has now reached the stage where easy to use automated thermometric titration systems can in many cases offer a superior alternative to potentiometric titrimetry.The applications of thermometric titrimetry discussed on this page are by no means exhaustive. The reader is referred to the bibliography for further reading on the subject.