Chapter12
... Chemists use balanced chemical equations as a basis to calculate how much reactant is needed or how much product is formed in a reaction. The calculation of quantities in chemical reactions is a subject of chemistry called stoichiometry. These quantities are typically measured in grams or moles, but ...
... Chemists use balanced chemical equations as a basis to calculate how much reactant is needed or how much product is formed in a reaction. The calculation of quantities in chemical reactions is a subject of chemistry called stoichiometry. These quantities are typically measured in grams or moles, but ...
More Fingerprinting Notes FP Analysis
... Fingerprints are treated with chemicals that would induce fluorescence when exposed to lasers, or high-intensity light sources (“alternate light sources”) such as quartz halogen, xenon arc, or indium arc light sources. ...
... Fingerprints are treated with chemicals that would induce fluorescence when exposed to lasers, or high-intensity light sources (“alternate light sources”) such as quartz halogen, xenon arc, or indium arc light sources. ...
7. A timeline of symbols and signs in chemistry
... This article aims to explore the relationship between symbolic explanation and the phenomena that require explanation. As a major source of evidence, it refers to textbooks use of chemical symbols and equations. The research questions were: 1. What signs and symbols appear in textbook of chemistry a ...
... This article aims to explore the relationship between symbolic explanation and the phenomena that require explanation. As a major source of evidence, it refers to textbooks use of chemical symbols and equations. The research questions were: 1. What signs and symbols appear in textbook of chemistry a ...
Course Map_2011-2012 - Kenwood Academy High School
... increases. Understand how a hydraulic lift (such as the kind used to raise a car for repairs) confers mechanical advantage. 12.11.78 Understand the universal law of gravitation: that gravitation is a force that every mass exerts on every other mass. The strength of the gravitational attractive force ...
... increases. Understand how a hydraulic lift (such as the kind used to raise a car for repairs) confers mechanical advantage. 12.11.78 Understand the universal law of gravitation: that gravitation is a force that every mass exerts on every other mass. The strength of the gravitational attractive force ...
Help us improve Wikipedia by supporting it financially
... The lightest elements are hydrogen and helium, both theoretically created by Big Bang nucleosynthesis during the first 20 minutes of the universe[10] in a ratio of around 3:1 by mass (approximately 12:1 by number of atoms). Almost all other elements found in nature, including some further hydrogen a ...
... The lightest elements are hydrogen and helium, both theoretically created by Big Bang nucleosynthesis during the first 20 minutes of the universe[10] in a ratio of around 3:1 by mass (approximately 12:1 by number of atoms). Almost all other elements found in nature, including some further hydrogen a ...
The Precautionary Principle and chemical risks - Hal-SHS
... (Godard, 1997; European Environmental Agency, 2001). At the same time, informed ignorance, i.e. ignorance that remains after having mobilised existing sources of scientific information- or the simple postulated presupposition that “bad effects could happen” are not sufficient to mobilize the PP. Thi ...
... (Godard, 1997; European Environmental Agency, 2001). At the same time, informed ignorance, i.e. ignorance that remains after having mobilised existing sources of scientific information- or the simple postulated presupposition that “bad effects could happen” are not sufficient to mobilize the PP. Thi ...
Sample pages 2 PDF
... reactions, studies the factors that influence them, designs and prepares new catalysts, and interprets the results at the molecular level. The independent variable of chemical kinetics, from the chemical reaction starting moment when the reactants are mixed to its final moment when equilibrium is rea ...
... reactions, studies the factors that influence them, designs and prepares new catalysts, and interprets the results at the molecular level. The independent variable of chemical kinetics, from the chemical reaction starting moment when the reactants are mixed to its final moment when equilibrium is rea ...
It`s Easy Being a Green Chemist
... (photovoltaic) converts solar energy into electricity and produces no green house gas emissions while providing a sustainable alternative to fossil fuels. On the downside, solar panels are manufactured with silicon-based products and contain heavy metals, such as lead (neurotoxin) and cadmium (carci ...
... (photovoltaic) converts solar energy into electricity and produces no green house gas emissions while providing a sustainable alternative to fossil fuels. On the downside, solar panels are manufactured with silicon-based products and contain heavy metals, such as lead (neurotoxin) and cadmium (carci ...
Chemical Dynamics at Surfaces
... The Haber process now produces 100 million tons of nitrogen fertilizer per year, mostly in the form of anhydrous ammonia, ammonium nitrate, and urea. ...
... The Haber process now produces 100 million tons of nitrogen fertilizer per year, mostly in the form of anhydrous ammonia, ammonium nitrate, and urea. ...
Shielding vs. Deshielding
... the electron withdrawing or releasing properties of the substituents based on relevant resonance structures and is symbolized by the letter M. The mesomeric effect is negative (-M) when the substituent is an electron-withdrawing group and the effect is positive (+M) when based on resonance the subst ...
... the electron withdrawing or releasing properties of the substituents based on relevant resonance structures and is symbolized by the letter M. The mesomeric effect is negative (-M) when the substituent is an electron-withdrawing group and the effect is positive (+M) when based on resonance the subst ...
Chemical Equilibrium
... o Ch 18 practice problems (due Thursday) o Ch 18 Review – due with the 21 May o The sub has permission to separate groups if you are not using your time well ...
... o Ch 18 practice problems (due Thursday) o Ch 18 Review – due with the 21 May o The sub has permission to separate groups if you are not using your time well ...
Chemical Compounds
... 4. The oxidation state of hydrogen is generally +1 except when it is bonded to metals such as sodium (NaH) in which case it's oxidation number is -1. 5. Fluorine has an oxidation number of -1 in its compounds … always. Group 1 elements have an oxidation number of +1 in their compounds … always. Grou ...
... 4. The oxidation state of hydrogen is generally +1 except when it is bonded to metals such as sodium (NaH) in which case it's oxidation number is -1. 5. Fluorine has an oxidation number of -1 in its compounds … always. Group 1 elements have an oxidation number of +1 in their compounds … always. Grou ...
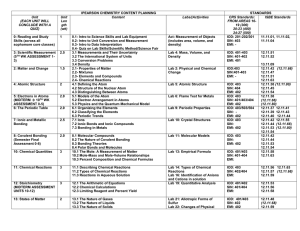
CHEMISTRY Academic Standards Statement
... substances or to understand how substances are formed and removed in the environment. Chemistry is the science of analysing, transforming or manipulating substances and the molecular interpretation of the world around us. It is at the molecular level that major advances are made in many diverse area ...
... substances or to understand how substances are formed and removed in the environment. Chemistry is the science of analysing, transforming or manipulating substances and the molecular interpretation of the world around us. It is at the molecular level that major advances are made in many diverse area ...
ch 7.1 - PickIntSci
... How many shoes do you own? Because shoes come in twos, you would most likely count them by the pair rather than individually. The counting units you use depend on what you are counting. For example, you might count eggs by the dozen or paper by the ream. Chemists also need practical units for counti ...
... How many shoes do you own? Because shoes come in twos, you would most likely count them by the pair rather than individually. The counting units you use depend on what you are counting. For example, you might count eggs by the dozen or paper by the ream. Chemists also need practical units for counti ...
aq - HCC Learning Web
... • Formulas and symbols are used to describe a chemical reaction as a chemical equation A+BC+D • A and B are reactants and C and D are products • Physical state is abbreviated by either (s), (l), (g), or (aq) • A catalyst is a substance that speeds up a reaction without being consumed or permanently ...
... • Formulas and symbols are used to describe a chemical reaction as a chemical equation A+BC+D • A and B are reactants and C and D are products • Physical state is abbreviated by either (s), (l), (g), or (aq) • A catalyst is a substance that speeds up a reaction without being consumed or permanently ...
Chapter 6 - Sites @ Suffolk University
... with the masses of the elements and their compounds by expressing the atomic masses in grams. The gram atomic mass of hydrogen, for instance, is about one gram, and the gram atomic mass of oxygen is about 16 grams. Any other unit of mass would work as well, of course, as long as oxygen is sixteen ti ...
... with the masses of the elements and their compounds by expressing the atomic masses in grams. The gram atomic mass of hydrogen, for instance, is about one gram, and the gram atomic mass of oxygen is about 16 grams. Any other unit of mass would work as well, of course, as long as oxygen is sixteen ti ...
eprint_5_24935_775
... Polymers are long chain molecules with a wide range of physical and chemical properties. One of the main advantages of the polymer materials is the ease of fabrication to produce various shapes (rod, film, fiber, sheet, etc.). The advances in polymer chemistry have made it possible to tailor the pro ...
... Polymers are long chain molecules with a wide range of physical and chemical properties. One of the main advantages of the polymer materials is the ease of fabrication to produce various shapes (rod, film, fiber, sheet, etc.). The advances in polymer chemistry have made it possible to tailor the pro ...
The Logical Structure of Organic Chemistry and the Empirical
... chemists, especially for organic chemists, than calculating abstract state functions by using higher mathematics. It was fortunate for chemists that most of the chemical reactions investigated thus far are not concerted, but ionic in character, and associated with the total energy of the molecule. T ...
... chemists, especially for organic chemists, than calculating abstract state functions by using higher mathematics. It was fortunate for chemists that most of the chemical reactions investigated thus far are not concerted, but ionic in character, and associated with the total energy of the molecule. T ...
1A. Growing Plants - The Royal Society of Chemistry
... required for growth from the supply within the seed. Once this supply is exhausted the plant takes up the major chemicals required (ie those based on nitrogen, phosphorus and potassium) from the soil. Leaf production requires a great deal of nitrogen, root formation demands phosphorus while during t ...
... required for growth from the supply within the seed. Once this supply is exhausted the plant takes up the major chemicals required (ie those based on nitrogen, phosphorus and potassium) from the soil. Leaf production requires a great deal of nitrogen, root formation demands phosphorus while during t ...
LN_ch06
... Balanced Chemical Equations Chemical Equations must be balanced There must be equal numbers of atoms of each element on both sides of the equation (both sides of the arrow) ►1. Write the correct symbols and formulas for all of the _______ and ____________. ►2. Count the number of each type of __ ...
... Balanced Chemical Equations Chemical Equations must be balanced There must be equal numbers of atoms of each element on both sides of the equation (both sides of the arrow) ►1. Write the correct symbols and formulas for all of the _______ and ____________. ►2. Count the number of each type of __ ...
4.2- Reaction Stoichiometry Reaction Stoichiometry
... (+) ion from one reactant with (–) ion from other Balance charges of combined ions to get formula of each product Determine solubility of each product in water Use the solubility rules If product is insoluble or slightly soluble, it will precipitate If neither product will precipitate, write no reac ...
... (+) ion from one reactant with (–) ion from other Balance charges of combined ions to get formula of each product Determine solubility of each product in water Use the solubility rules If product is insoluble or slightly soluble, it will precipitate If neither product will precipitate, write no reac ...
Eperimental studies of V.Ostwald and J.van Hoff
... chemistry and became interested in explaining why various chemical reactions occur at widely different rates. In 1884 he published the innovative book Études de dynamique chimique (“Studies in Chemical Dynamics”), in which he used the principles of thermodynamics to provide a mathematical model for ...
... chemistry and became interested in explaining why various chemical reactions occur at widely different rates. In 1884 he published the innovative book Études de dynamique chimique (“Studies in Chemical Dynamics”), in which he used the principles of thermodynamics to provide a mathematical model for ...
Chemistry
... aspects underlying global and local issues; to be sceptical and questioning of claims made by others about scientific matters; to be able to identify questions, draw evidence-based conclusions and discuss their validity; and to form opinions, that are reasoned and informed, about the environment, ab ...
... aspects underlying global and local issues; to be sceptical and questioning of claims made by others about scientific matters; to be able to identify questions, draw evidence-based conclusions and discuss their validity; and to form opinions, that are reasoned and informed, about the environment, ab ...
Chemical industry

The chemical industry comprises the companies that produce industrial chemicals. Central to the modern world economy, it converts raw materials (oil, natural gas, air, water, metals, and minerals) into more than 70,000 different products.