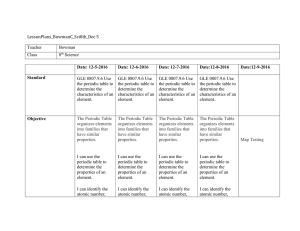
Amount of substance
... Q31.The percentage of copper in a copper(II) salt can be determined by using a thiosulphate titration. 0.305 g of a copper(II) salt was dissolved in water and added to an excess of potassium iodide solution, liberating iodine according to the following equation: 2Cu2+(aq) + 4I−(aq) → 2CuI(s) + I2(aq ...
... Q31.The percentage of copper in a copper(II) salt can be determined by using a thiosulphate titration. 0.305 g of a copper(II) salt was dissolved in water and added to an excess of potassium iodide solution, liberating iodine according to the following equation: 2Cu2+(aq) + 4I−(aq) → 2CuI(s) + I2(aq ...
Concerning Electronegativity as a Basic Elemental Property and
... Fig. 4 Atomic number, Z, increases across (left-to-right) and down the Janet Left-Step formulation. Physical and chemical properties show periodicity down the Groups. There is no general left-to-right periodicity. Consider elements 9, 10, 11, 12 that correspond to the distinctly non-periodic: F, Ne, ...
... Fig. 4 Atomic number, Z, increases across (left-to-right) and down the Janet Left-Step formulation. Physical and chemical properties show periodicity down the Groups. There is no general left-to-right periodicity. Consider elements 9, 10, 11, 12 that correspond to the distinctly non-periodic: F, Ne, ...
QUESTION BANK CHEMISTRY-XII THE SOLID STATE CHAPTER
... (i) Electrolysis of aqueous solution of NaCl gives H2 at cathode and Cl2 gas at anode. (ii) Electrolysis of aqueous solution of CuBr2 gives Cu at cathode and Br2 at the anode. (ii) A Bromide or iodide on electrolysis give Br2 or I2 at the anode whereas of fluoride does no ...
... (i) Electrolysis of aqueous solution of NaCl gives H2 at cathode and Cl2 gas at anode. (ii) Electrolysis of aqueous solution of CuBr2 gives Cu at cathode and Br2 at the anode. (ii) A Bromide or iodide on electrolysis give Br2 or I2 at the anode whereas of fluoride does no ...
virtual lab- Atoms on periodic table student
... _____________________ ____________. He_____________________ that there was a _________________ to the ___________________of the ___________________. This ___________________of _________________ according to ___________________ changes in __________________is called a ________________ _______________ ...
... _____________________ ____________. He_____________________ that there was a _________________ to the ___________________of the ___________________. This ___________________of _________________ according to ___________________ changes in __________________is called a ________________ _______________ ...
periodic table
... – IA are called alkali metals because the react with water to from an alkaline solution – Group IIA are called the alkaline earth metals because they are reactive, but not as reactive as Group IA. • They are also soft metals like Earth. ...
... – IA are called alkali metals because the react with water to from an alkaline solution – Group IIA are called the alkaline earth metals because they are reactive, but not as reactive as Group IA. • They are also soft metals like Earth. ...
Periodic Table - Derry Area School District
... Configurations and the Periodic Table • The following slide illustrates how the ...
... Configurations and the Periodic Table • The following slide illustrates how the ...
CPO Science Link Teacher`s Guide
... neon (Ne), and argon (Ar). These elements do not naturally form chemical bonds with other atoms. They are almost always found in their pure state, not as part of compounds. Elements in Groups 3 through 12 are called the transition metals. These elements are usually good conductors of heat and electr ...
... neon (Ne), and argon (Ar). These elements do not naturally form chemical bonds with other atoms. They are almost always found in their pure state, not as part of compounds. Elements in Groups 3 through 12 are called the transition metals. These elements are usually good conductors of heat and electr ...
Ch. 5 - Periodic Law
... Any process that results in the formation of an ion is referred to as ionization. The energy required to remove one electron from a neutral atom of an element is the ionization energy, IE. ...
... Any process that results in the formation of an ion is referred to as ionization. The energy required to remove one electron from a neutral atom of an element is the ionization energy, IE. ...
The science of chemistry is concerned with the composition
... gentleman farmer, economist, and social reformer, as well as the leading chemist of his day. It was Lavoisier’s position as a tax collector, not his chemical research, which led to his death by guillotine in 1794, at the height of the French Revolution. Much of Lavoisier’s work as a chemist was devo ...
... gentleman farmer, economist, and social reformer, as well as the leading chemist of his day. It was Lavoisier’s position as a tax collector, not his chemical research, which led to his death by guillotine in 1794, at the height of the French Revolution. Much of Lavoisier’s work as a chemist was devo ...
The science of chemistry is concerned with the
... gentleman farmer, economist, and social reformer, as well as the leading chemist of his day. It was Lavoisier’s position as a tax collector, not his chemical research, which led to his death by guillotine in 1794, at the height of the French Revolution. Much of Lavoisier’s work as a chemist was devo ...
... gentleman farmer, economist, and social reformer, as well as the leading chemist of his day. It was Lavoisier’s position as a tax collector, not his chemical research, which led to his death by guillotine in 1794, at the height of the French Revolution. Much of Lavoisier’s work as a chemist was devo ...
Chem 2A Final Review
... 9. The answer closest to the number of grams of NaH2PO4 needed to react with 38.74 mL of 0.275 M NaOH, according to the following balanced equation is: (NaH2PO4 = 119.98 g/mol) NaH2PO4 (s) + 2NaOH (aq) Na3POH4 (aq) + 3H2O ...
... 9. The answer closest to the number of grams of NaH2PO4 needed to react with 38.74 mL of 0.275 M NaOH, according to the following balanced equation is: (NaH2PO4 = 119.98 g/mol) NaH2PO4 (s) + 2NaOH (aq) Na3POH4 (aq) + 3H2O ...
1aUnit Two Handouts - Dunmore High School
... Is it a strong base: NaOH, LiOH, KOH, Ba(OH)2, or Ca(OH)2? If yes, write it as ions. Example: NaOH becomes Na+ + OHIf no, do not write it as ions. Example: Fe(OH)3 stays Fe(OH)3 (Note: Most hydroxides not listed above are weak or nonelectrolytes because they are insoluble in water—always check this ...
... Is it a strong base: NaOH, LiOH, KOH, Ba(OH)2, or Ca(OH)2? If yes, write it as ions. Example: NaOH becomes Na+ + OHIf no, do not write it as ions. Example: Fe(OH)3 stays Fe(OH)3 (Note: Most hydroxides not listed above are weak or nonelectrolytes because they are insoluble in water—always check this ...
Collins CSEC® Chemistry Workbook answers A1 States of matter
... on a balance, transfer it to a beaker and add enough distilled water to dissolve the solid. She would pour the solution into a clean, 1 dm3 volumetric flask and rinse the beaker over the flask, transferring the washings to the flask. She would then fill the flask with distilled water so the meniscus ...
... on a balance, transfer it to a beaker and add enough distilled water to dissolve the solid. She would pour the solution into a clean, 1 dm3 volumetric flask and rinse the beaker over the flask, transferring the washings to the flask. She would then fill the flask with distilled water so the meniscus ...
2(g)
... for the products and the reactants. Be sure to include all states of matter! 2) Balance the atoms or ion present in the greatest number. You may do this by finding the lowest common multiple of the two. 3) Continue to systematically balance the rest of the atoms or ions. 4) Check the final equation. ...
... for the products and the reactants. Be sure to include all states of matter! 2) Balance the atoms or ion present in the greatest number. You may do this by finding the lowest common multiple of the two. 3) Continue to systematically balance the rest of the atoms or ions. 4) Check the final equation. ...
College Grossmont 115
... or numbers obtained by definition. For example, we can count the fingers on our hand and get an exact number (most people have 5). There is no uncertainty in this result, but we cannot count large groups of objects without some degree of uncertainty. For example, the number of stars in our galaxy is ...
... or numbers obtained by definition. For example, we can count the fingers on our hand and get an exact number (most people have 5). There is no uncertainty in this result, but we cannot count large groups of objects without some degree of uncertainty. For example, the number of stars in our galaxy is ...
OCR Gateway Science
... (a) How does the volume of hydrogen produced depend on the mass of magnesium used? (b) Which reactant is limiting? Explain your answer. (Mg = 24) (c) Calculate the initial rate of reaction when 0.08 g of magnesium reacts with 20 cm3 of 2 mol/dm3 hydrochloric acid. (d) 1 mole of gas occupies 24 dm3 a ...
... (a) How does the volume of hydrogen produced depend on the mass of magnesium used? (b) Which reactant is limiting? Explain your answer. (Mg = 24) (c) Calculate the initial rate of reaction when 0.08 g of magnesium reacts with 20 cm3 of 2 mol/dm3 hydrochloric acid. (d) 1 mole of gas occupies 24 dm3 a ...
chemistry
... studies in chemistry, we can only look at the general; more specific knowledge will come in time as you continue to study. Figure 1.2 illustrates the relationships of the components of matter and how they can be classified; some of the terms will be defined in the text below, some will already be fa ...
... studies in chemistry, we can only look at the general; more specific knowledge will come in time as you continue to study. Figure 1.2 illustrates the relationships of the components of matter and how they can be classified; some of the terms will be defined in the text below, some will already be fa ...
KHOA: HÓA HỌC - CCS - Trường Đại học Sư phạm Hà Nội
... element is a substance comprised of a single type of atom. The elements are the building blocks of our nature. An element is either discovered in nature or synthesized in the laboratory in pure form that cannot be separated into simpler substances by chemical methods. Currently, there are about 118 ...
... element is a substance comprised of a single type of atom. The elements are the building blocks of our nature. An element is either discovered in nature or synthesized in the laboratory in pure form that cannot be separated into simpler substances by chemical methods. Currently, there are about 118 ...
The Evolution of the Periodic System
... by what scientists call quantum numbers. The lengths of the various periods emerge from experimental evidence about the order of electron-shell filling and from the quantum-mechanical restrictions on the four quantum numbers that electrons can adopt. The modifications to quantum theory made by Werne ...
... by what scientists call quantum numbers. The lengths of the various periods emerge from experimental evidence about the order of electron-shell filling and from the quantum-mechanical restrictions on the four quantum numbers that electrons can adopt. The modifications to quantum theory made by Werne ...
The Evolution of the Periodic System - Science
... by what scientists call quantum numbers. The lengths of the various periods emerge from experimental evidence about the order of electron-shell filling and from the quantum-mechanical restrictions on the four quantum numbers that electrons can adopt. The modifications to quantum theory made by Werne ...
... by what scientists call quantum numbers. The lengths of the various periods emerge from experimental evidence about the order of electron-shell filling and from the quantum-mechanical restrictions on the four quantum numbers that electrons can adopt. The modifications to quantum theory made by Werne ...
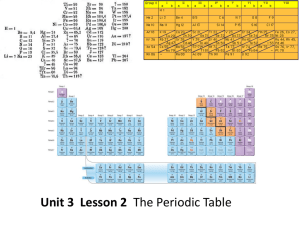
periodic table
... How are the elements arranged on the periodic table? • Each vertical column of elements on the periodic table is called a group, or family. There are 18 groups. • Elements in a group are similar because their atoms have the same number of valence electrons. • Valence electrons are the electrons foun ...
... How are the elements arranged on the periodic table? • Each vertical column of elements on the periodic table is called a group, or family. There are 18 groups. • Elements in a group are similar because their atoms have the same number of valence electrons. • Valence electrons are the electrons foun ...