Unit 3: Bonding and Nomenclature Content Outline: Chemical
... A. This type of chemical formula shows the kinds of atoms and their relative numbers in a substance in the smallest whole number ratios. For example: H2O2 HO OR C6H12O6 CH2O (Take the lowest subscript number and divide it into the other subscripts.) VII. Energy and Molecules A. The natural tende ...
... A. This type of chemical formula shows the kinds of atoms and their relative numbers in a substance in the smallest whole number ratios. For example: H2O2 HO OR C6H12O6 CH2O (Take the lowest subscript number and divide it into the other subscripts.) VII. Energy and Molecules A. The natural tende ...
vsepr_lite_oct_2011 - chemistry11crescentsummer
... fig 5. The Lewis structure of HOH. How many lone pair(s) of electrons are on the central atom, O? Adapt the molecular model of NH3 to get a model of HOH. How would you describe the shape of a water molecule? No fancy name required. Use the bond angles in CH4 and NH3 to predict the H–O–H bond angle i ...
... fig 5. The Lewis structure of HOH. How many lone pair(s) of electrons are on the central atom, O? Adapt the molecular model of NH3 to get a model of HOH. How would you describe the shape of a water molecule? No fancy name required. Use the bond angles in CH4 and NH3 to predict the H–O–H bond angle i ...
Document
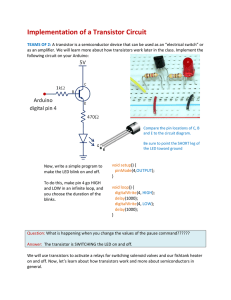
... (electrons in an NPN transistor) in the base is linear. • The amplifying action is produced by transferring a current from a low-resistance circuit to a high resistance circuit. • transfer + resistor → transistor ...
... (electrons in an NPN transistor) in the base is linear. • The amplifying action is produced by transferring a current from a low-resistance circuit to a high resistance circuit. • transfer + resistor → transistor ...
CHEMONE Directions: Select the letter of the best
... a. Ionic bonding results from the transfer of electrons from one atom to another. b. Dipole moments result from the unequal distribution of electrons in a molecule. c. The electrons in a polar bond are found nearer to the more electronegative element. d. A molecule with very nonpolar bonds can be no ...
... a. Ionic bonding results from the transfer of electrons from one atom to another. b. Dipole moments result from the unequal distribution of electrons in a molecule. c. The electrons in a polar bond are found nearer to the more electronegative element. d. A molecule with very nonpolar bonds can be no ...
The chemical building blocks of life Carbon
... medium for life For instance other polar solvents, such as HF, are interesting in principle, but are by far less abundant than water in the cosmos. Here the index in the row (g) represents the product of the cosmic abundances of the atoms that compose the molecule. Water also has a relatively high s ...
... medium for life For instance other polar solvents, such as HF, are interesting in principle, but are by far less abundant than water in the cosmos. Here the index in the row (g) represents the product of the cosmic abundances of the atoms that compose the molecule. Water also has a relatively high s ...