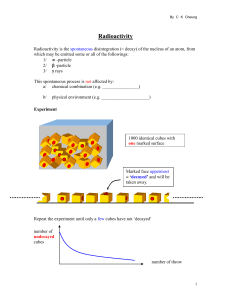
Radioactivity - Miami Beach Senior High School
... • The nucleus splits into two smaller nuclei, which are radioactive. • More neutrons are released. • The additional neutrons released may also hit other uranium nuclei and cause them to split. Even more neutrons are then released, which in turn can split more uranium nuclei. This is called a chain r ...
... • The nucleus splits into two smaller nuclei, which are radioactive. • More neutrons are released. • The additional neutrons released may also hit other uranium nuclei and cause them to split. Even more neutrons are then released, which in turn can split more uranium nuclei. This is called a chain r ...
GRAMMAR: verb tenses
... repel one another. Hence these electrical forces of repulsion must be counteracted by another type of attractive force, a nuclear force, if the nucleus is to bind together. The relative balance between the electrical repulsion of the protons forcing the nucleons apart and the nuclear forces pulling ...
... repel one another. Hence these electrical forces of repulsion must be counteracted by another type of attractive force, a nuclear force, if the nucleus is to bind together. The relative balance between the electrical repulsion of the protons forcing the nucleons apart and the nuclear forces pulling ...
Unit 14 Notes - shscience.net
... reactor fuel captures them to continue the chain reaction Neutron absorption- decreases the number of slow-moving neutrons Control rods- used to absorb neutrons ...
... reactor fuel captures them to continue the chain reaction Neutron absorption- decreases the number of slow-moving neutrons Control rods- used to absorb neutrons ...
Summative Assessment Review!
... 32 days. Do this by seeing how many times 8, the halflife of the substance, can go into 32, the amount of days. 32/8 = 4, so the amount of substance is halved ...
... 32 days. Do this by seeing how many times 8, the halflife of the substance, can go into 32, the amount of days. 32/8 = 4, so the amount of substance is halved ...
Nuclear Chemistry
... • Gamma rays are emitted during nuclear reactions, either alone or with other types of radiation • Gamma rays do NOT change the mass number or atomic number because they are energy not matter. ...
... • Gamma rays are emitted during nuclear reactions, either alone or with other types of radiation • Gamma rays do NOT change the mass number or atomic number because they are energy not matter. ...
Nuclear Chemistry - Xavier High School
... neutrons are rearranged • The opening of the nucleus releases a tremendous amount of energy that holds the nucleus together – called binding ...
... neutrons are rearranged • The opening of the nucleus releases a tremendous amount of energy that holds the nucleus together – called binding ...
Glossary of Key Terms in Chapter Two
... alpha particle (9.1) consists of two protons and two neutrons (the alpha particle is identical to helium nucleus). artificial radioactivity (9.5) radiation that results from the conversion of a stable nucleus to another, unstable nucleus. background radiation (9.6) the radiation that emanates from n ...
... alpha particle (9.1) consists of two protons and two neutrons (the alpha particle is identical to helium nucleus). artificial radioactivity (9.5) radiation that results from the conversion of a stable nucleus to another, unstable nucleus. background radiation (9.6) the radiation that emanates from n ...
Chapter 19 Nuclear Chemistry
... • 42 He nucleus is “synthesized” from 2 protons and two neutrons. m = 4.0026 2me m = 0.0304 amu ...
... • 42 He nucleus is “synthesized” from 2 protons and two neutrons. m = 4.0026 2me m = 0.0304 amu ...
25.3 Fission and Fusion of Atomic Nuclei
... illustration shows the basic components of a nuclear reactor. Energy from the fission process heats the circulating coolant. The heated coolant is used to produce steam that turns a steam-driven turbine. The turbine drives a generator to produce electrical energy. ...
... illustration shows the basic components of a nuclear reactor. Energy from the fission process heats the circulating coolant. The heated coolant is used to produce steam that turns a steam-driven turbine. The turbine drives a generator to produce electrical energy. ...
Radioactivity
... Nuclear Fission When a heavy nuclide is split into 2 large fragments, the process is called fission. Usually, a fission process requires a neutron ( uncharged ) to initiate it. ...
... Nuclear Fission When a heavy nuclide is split into 2 large fragments, the process is called fission. Usually, a fission process requires a neutron ( uncharged ) to initiate it. ...
Problem Set 7 Solutions
... l’s moderating power is a function of how much is there, and its value of A material’s finding a way to increase the α, or how much it can slow neutrons down. Therefore, finding hydrogen density, like by using liquid hydrogen (not very feasible) or using a solid metal-hydride (much more feasible) cou ...
... l’s moderating power is a function of how much is there, and its value of A material’s finding a way to increase the α, or how much it can slow neutrons down. Therefore, finding hydrogen density, like by using liquid hydrogen (not very feasible) or using a solid metal-hydride (much more feasible) cou ...
Chemistry 1 CP Concept 4 Nuclear Chemistry Study Guide
... decay? _______________, for ½ _________________, for ¾ _________________ 19. Which radioactive decay process does not reduce the atomic number of a nuclide? ____________________________ 20. What is the half-life of an isotope if 100 g of an 800 g sample of the isotope remain after 6 years? _________ ...
... decay? _______________, for ½ _________________, for ¾ _________________ 19. Which radioactive decay process does not reduce the atomic number of a nuclide? ____________________________ 20. What is the half-life of an isotope if 100 g of an 800 g sample of the isotope remain after 6 years? _________ ...
Nuclear Weapons (and Energy) Each element has different number
... goes down a bit but still have strong nuclear binding forces. Like putting a bunch of neutral people between one’s that find each other repulsive in same room… MORE STABLE SITUATION ...
... goes down a bit but still have strong nuclear binding forces. Like putting a bunch of neutral people between one’s that find each other repulsive in same room… MORE STABLE SITUATION ...
Nuclear Decay
... The energy released by fusion is three to four times than that released in chemical reactions, but lower than greater than the energy released by fission. the energy released by nuclear fusion. ...
... The energy released by fusion is three to four times than that released in chemical reactions, but lower than greater than the energy released by fission. the energy released by nuclear fusion. ...
Nuclear Reactions
... The missing mass (mass defect) has been stored as energy in the nucleus. It is called the binding energy of the nucleus. ...
... The missing mass (mass defect) has been stored as energy in the nucleus. It is called the binding energy of the nucleus. ...
Article 2: Key Concepts and Vocabulary
... two products have two protons and three neutrons, just as the reactants do. Also, 17.6 million electron volts (MeV) are released, initially in the form of the kinetic energy of the two products. Of all the nuclear reactions, this “deuterium-tritium” or “D-T” reaction is the least difficult to produc ...
... two products have two protons and three neutrons, just as the reactants do. Also, 17.6 million electron volts (MeV) are released, initially in the form of the kinetic energy of the two products. Of all the nuclear reactions, this “deuterium-tritium” or “D-T” reaction is the least difficult to produc ...
Nuclear Reactions Review
... 3.Nuclei with too many or too few neutrons are A.never found. C.unnatural. B.unstable. D.stable. ...
... 3.Nuclei with too many or too few neutrons are A.never found. C.unnatural. B.unstable. D.stable. ...
Nuclear Reactions Review powerpt
... 3.Nuclei with too many or too few neutrons are A.never found. C.unnatural. B.unstable. D.stable. ...
... 3.Nuclei with too many or too few neutrons are A.never found. C.unnatural. B.unstable. D.stable. ...
120 min This paper - University of Southampton
... paired, so spin and parity are defined by unpaired neutron. Neutrons fill shells as follows ( with j = l + 1/2 fills before j = l − 1/2 due to specific features of the spinorbit interactions): (1s1/2)2 (1p3/2)4 (1p1/2)1 Therefore there is one unpaired neutron in 1p1/2 state forming state with parity ...
... paired, so spin and parity are defined by unpaired neutron. Neutrons fill shells as follows ( with j = l + 1/2 fills before j = l − 1/2 due to specific features of the spinorbit interactions): (1s1/2)2 (1p3/2)4 (1p1/2)1 Therefore there is one unpaired neutron in 1p1/2 state forming state with parity ...
Physical Science: Nuclear Chemistry Study Guide
... 35. What is one example of a place where naturally occurring extreme temperatures provide the energy needed to bring hydrogen nuclei together in a fusion reaction? 36. Name one good use of the energy produced in a controlled chain reaction. 37. What is nuclear radiation that is emitted from natural ...
... 35. What is one example of a place where naturally occurring extreme temperatures provide the energy needed to bring hydrogen nuclei together in a fusion reaction? 36. Name one good use of the energy produced in a controlled chain reaction. 37. What is nuclear radiation that is emitted from natural ...
File
... nuclear fission. This releases lots of energy Heavy atoms (mass number>60) tend to break into smaller atoms, thereby increasing their stability. ...
... nuclear fission. This releases lots of energy Heavy atoms (mass number>60) tend to break into smaller atoms, thereby increasing their stability. ...
Nuclear Chemistry
... • All nuclei with atomic numbers greater than 83 are radioactive • These nuclei have both too many neutrons and too many protons to be stable – So most undergo decay ...
... • All nuclei with atomic numbers greater than 83 are radioactive • These nuclei have both too many neutrons and too many protons to be stable – So most undergo decay ...