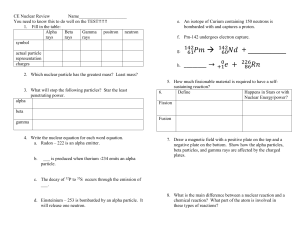
Independent Study: Nuclear Chemistry
... 20. Unstable isotopes of elements are called radioisotopes. 21. The symbol represents tritium. 22. Gamma rays can be stopped by an aluminum sheet. 23. The change of an atom into a new element is called a chemical change. 24. The first artificial transmutation was performed by Albert Einstein. 25. Th ...
... 20. Unstable isotopes of elements are called radioisotopes. 21. The symbol represents tritium. 22. Gamma rays can be stopped by an aluminum sheet. 23. The change of an atom into a new element is called a chemical change. 24. The first artificial transmutation was performed by Albert Einstein. 25. Th ...
Radioactivity - Mrs. Sjuts` Science Site
... ! An atom of C-‐14 eventually will decay into N-‐14 with a half-‐life of 5,730 years ! By measuring the amount of C-‐14 in a sample and comparing it to the amount of C-‐12, scientists can ...
... ! An atom of C-‐14 eventually will decay into N-‐14 with a half-‐life of 5,730 years ! By measuring the amount of C-‐14 in a sample and comparing it to the amount of C-‐12, scientists can ...
Independent Study: Nuclear Chemistry
... 20. Unstable isotopes of elements are called radioisotopes. 21. The symbol represents tritium. 22. Gamma rays can be stopped by an aluminum sheet. 23. The change of an atom into a new element is called a chemical change. 24. The first artificial transmutation was performed by Albert Einstein. 25. Th ...
... 20. Unstable isotopes of elements are called radioisotopes. 21. The symbol represents tritium. 22. Gamma rays can be stopped by an aluminum sheet. 23. The change of an atom into a new element is called a chemical change. 24. The first artificial transmutation was performed by Albert Einstein. 25. Th ...
Chapter 21 - Richsingiser.com
... • Nuclear fission forms two nuclei of roughly similar size from a single heavy nucleus. • Fission reactions release very large quantities of energy (“exergonic”) and produce several neutrons in addition to two nuclides. ...
... • Nuclear fission forms two nuclei of roughly similar size from a single heavy nucleus. • Fission reactions release very large quantities of energy (“exergonic”) and produce several neutrons in addition to two nuclides. ...
File
... • Mechanism: Radiation beams knock off electrons from atoms making up DNA chains and turn them into free radicals (damaging them!) • Polonium, Cobalt, Cesium & Radium are all elements used in radiation therapy. ...
... • Mechanism: Radiation beams knock off electrons from atoms making up DNA chains and turn them into free radicals (damaging them!) • Polonium, Cobalt, Cesium & Radium are all elements used in radiation therapy. ...
nuclear physics - rct study guide
... the observation that fission occurs in these fissionable nuclei only when the neutron has approximately 1 MeV of kinetic energy. The situation is quite different for U-235, U-233, and Pu-239. In these cases, the neutron binding energy exceeds the critical energy for fission. Thus, these nuclei may b ...
... the observation that fission occurs in these fissionable nuclei only when the neutron has approximately 1 MeV of kinetic energy. The situation is quite different for U-235, U-233, and Pu-239. In these cases, the neutron binding energy exceeds the critical energy for fission. Thus, these nuclei may b ...
Nuclear Reactions - Kelso High School
... • The fact that most of the particles passed straight through the foil, which was at least 100 atoms thick, implied that the atom must be mostly empty space. • In order to produce the large deflections at C and D, the alpha particles must be colliding with something very small, of large mass and pos ...
... • The fact that most of the particles passed straight through the foil, which was at least 100 atoms thick, implied that the atom must be mostly empty space. • In order to produce the large deflections at C and D, the alpha particles must be colliding with something very small, of large mass and pos ...
Content Domain III: Chemistry—Atomic Theory and
... Sometimes two low-mass nuclei combine to form one nucleus in a process called fusion. A tremendous amount of energy is released in the process. Extremely high temperatures, like those found in stars, are needed for nuclear fusion to take place. Fusion, as a future energy source, might provide all th ...
... Sometimes two low-mass nuclei combine to form one nucleus in a process called fusion. A tremendous amount of energy is released in the process. Extremely high temperatures, like those found in stars, are needed for nuclear fusion to take place. Fusion, as a future energy source, might provide all th ...
Ch 10 Nuclear Chemistry
... when the strong nuclear force can no longer overcome the repulsive electric forces among protons. • All nuclei with more than 83 protons are radioactive ...
... when the strong nuclear force can no longer overcome the repulsive electric forces among protons. • All nuclei with more than 83 protons are radioactive ...
Nuclear Chemistry - Mona Shores Blogs
... uncontrolled chain reactions • In order for a chain reaction to keep going, the nucleus that splits needs to produce one neutron that causes the fission of another nucleus – The material reacting uncontrolled needs to have a critical mass. – CRITICAL MASS: the smallest possible mass of a fissionable ...
... uncontrolled chain reactions • In order for a chain reaction to keep going, the nucleus that splits needs to produce one neutron that causes the fission of another nucleus – The material reacting uncontrolled needs to have a critical mass. – CRITICAL MASS: the smallest possible mass of a fissionable ...
Nuclear For Forensics
... detonated, gamma rays and Xrays emitted first compress the fusion fuel, then heat it to thermonuclear temperatures ...
... detonated, gamma rays and Xrays emitted first compress the fusion fuel, then heat it to thermonuclear temperatures ...
NUCLEAR CHEMISTRY
... 1. Gamma rays () are high-energy electromagnetic waves emitted from a nucleus as it changes from an excited state to a ground energy state 2. Gamma rays are produced when nuclear particles undergo transitions in energy levels ...
... 1. Gamma rays () are high-energy electromagnetic waves emitted from a nucleus as it changes from an excited state to a ground energy state 2. Gamma rays are produced when nuclear particles undergo transitions in energy levels ...
Atomic/Nuclear
... If the neutrons set free are absorbed by other nuclei, then a chain reaction can occur. If the sample is less than the critical size, it has a small mass to surface area ratio and too many neutrons lost from the sample to sustain a chain reaction. Detonation of a bomb is achieved by ramming together ...
... If the neutrons set free are absorbed by other nuclei, then a chain reaction can occur. If the sample is less than the critical size, it has a small mass to surface area ratio and too many neutrons lost from the sample to sustain a chain reaction. Detonation of a bomb is achieved by ramming together ...
Glossary of Technical Terms - Institute for Energy and Environmental
... Any isotope created by the fission of a heavy element. Fission products are usually radioactive. fission The splitting of the nucleus of an element into fragments. Heavy elements such as uranium or plutonium release energy when fissioned. fissile material A material consisting of atoms whose nuclei ...
... Any isotope created by the fission of a heavy element. Fission products are usually radioactive. fission The splitting of the nucleus of an element into fragments. Heavy elements such as uranium or plutonium release energy when fissioned. fissile material A material consisting of atoms whose nuclei ...
1 0 +1 0 - davis.k12.ut.us
... occur in nature, can you list a few? Technetium, neptunium, elements 93 and up… 10. How can synthetic elements be produced? Bombarding an atom with a neutron or alpha particle. 11. A 5.0 g sample of Lead-210 decays to approximately .63 g. How much time has passed if Lead-210 has a half-life of 22 ye ...
... occur in nature, can you list a few? Technetium, neptunium, elements 93 and up… 10. How can synthetic elements be produced? Bombarding an atom with a neutron or alpha particle. 11. A 5.0 g sample of Lead-210 decays to approximately .63 g. How much time has passed if Lead-210 has a half-life of 22 ye ...
Basics of Nuclear Physics and Fission
... Many heavy nuclei emit an energetic alpha particle when they decay. For instance uranium-238 decays into thorium-234 with a half-life of almost 4.5 billion years by emitting an alpha particle: 92-uranium-238 ====> 90-thorium-234 + alpha particle (nucleus of 2-helium-4) The mass number of uranium-238 ...
... Many heavy nuclei emit an energetic alpha particle when they decay. For instance uranium-238 decays into thorium-234 with a half-life of almost 4.5 billion years by emitting an alpha particle: 92-uranium-238 ====> 90-thorium-234 + alpha particle (nucleus of 2-helium-4) The mass number of uranium-238 ...
Nuclear reactions: fission and fusion
... redistribution of electric charge within a nucleus. Gamma rays are essentially very energetic X - rays; the distinction between the two is not based on their intrinsic nature but rather on their origins. X rays are emitted during atomic processes involving energetic electrons. Gamma radiation is emi ...
... redistribution of electric charge within a nucleus. Gamma rays are essentially very energetic X - rays; the distinction between the two is not based on their intrinsic nature but rather on their origins. X rays are emitted during atomic processes involving energetic electrons. Gamma radiation is emi ...
Alpha Beta Fission Fusion
... In 1902, Frederick Soddy proposed the theory that "radioactivity is the result of a natural change of an isotope of one element into an isotope of a different element." Nuclear reactions involve changes in particles in an atom's nucleus and thus cause a change in the atom itself. All elements heavie ...
... In 1902, Frederick Soddy proposed the theory that "radioactivity is the result of a natural change of an isotope of one element into an isotope of a different element." Nuclear reactions involve changes in particles in an atom's nucleus and thus cause a change in the atom itself. All elements heavie ...
AC Geophysical Science Study Guide
... 6. What are the major obstacles to building a fusion-powered electric-producing power plant? Why is a fusion reactor not in use? 7. Why does uranium not yield energy if fused? 8. What are two advantages and two disadvantages to the eventual use of fusion reactors? 9. Calculate the amount of energy l ...
... 6. What are the major obstacles to building a fusion-powered electric-producing power plant? Why is a fusion reactor not in use? 7. Why does uranium not yield energy if fused? 8. What are two advantages and two disadvantages to the eventual use of fusion reactors? 9. Calculate the amount of energy l ...
A Conceptual Introduction to Chemistry, First Edition
... Critical mass – The smallest amount of fissionable material necessary to support a continuing chain reaction ...
... Critical mass – The smallest amount of fissionable material necessary to support a continuing chain reaction ...
solutions - Physicsland
... 11. The proton “bullets” need enough momentum to overcome the electric force of repulsion they experience once they get close to the atomic nucleus. 14.The fusing of hydrogen atoms into helium atoms suggests that the electric force of repulsions can be overcome by the strong nuclear force if two ato ...
... 11. The proton “bullets” need enough momentum to overcome the electric force of repulsion they experience once they get close to the atomic nucleus. 14.The fusing of hydrogen atoms into helium atoms suggests that the electric force of repulsions can be overcome by the strong nuclear force if two ato ...
printable version
... b. Beta emission occurs in isotopes that have too many neutrons ( more neutrons than protons in small atoms) like Cobalt – 60 ( 27 protons and 33 neutrons) c. Isotopes that have too few neutrons (like nitrogen-13) would attain stability by electron capture or positron emission ...
... b. Beta emission occurs in isotopes that have too many neutrons ( more neutrons than protons in small atoms) like Cobalt – 60 ( 27 protons and 33 neutrons) c. Isotopes that have too few neutrons (like nitrogen-13) would attain stability by electron capture or positron emission ...
Physics: Principles and Applications, 6e Giancoli
... 19) An atom has 98 protons and 249 nucleons. If it undergoes alpha decay, what are the number of protons and neutrons, respectively, in the daughter nucleus? A) 100, 245 B) 94, 247 C) 96, 245 D) 100, 249 20) An element with atomic number 6 undergoes β- decay. Its atomic number is now A) 7. B) 6. C) ...
... 19) An atom has 98 protons and 249 nucleons. If it undergoes alpha decay, what are the number of protons and neutrons, respectively, in the daughter nucleus? A) 100, 245 B) 94, 247 C) 96, 245 D) 100, 249 20) An element with atomic number 6 undergoes β- decay. Its atomic number is now A) 7. B) 6. C) ...
1 + - crypt
... What isotopes are used for fission reactions? Copy Figure 3 on page 203 explain how a nuclear fission reactor produces steam to drive the turbines of a power station. Your account should include the purpose of the moderator, control rods and coolant. Copy and answer question (b) on page 203. Copy th ...
... What isotopes are used for fission reactions? Copy Figure 3 on page 203 explain how a nuclear fission reactor produces steam to drive the turbines of a power station. Your account should include the purpose of the moderator, control rods and coolant. Copy and answer question (b) on page 203. Copy th ...
nuclear chemistry - Wood County Schools
... Positron Emission: Medium-level radiation from the emission of a positron, which is the same as an electron, only with a positive charge, converting a proton into a neutron. Electron Capture: When an atom takes in an electron, converting a proton into a neutron. Fission: The splitting of an atomic n ...
... Positron Emission: Medium-level radiation from the emission of a positron, which is the same as an electron, only with a positive charge, converting a proton into a neutron. Electron Capture: When an atom takes in an electron, converting a proton into a neutron. Fission: The splitting of an atomic n ...