Chapter 25 Radioactivity
... Uranium, plutonium, oxygen, sodium, krypton, carbon Isotopes: atoms of the same element with different numbers of neutrons i.e 1214C and 1213C each has 12 protons which is the Atomic number. The mass number varies. It is the sum of the protons and neutrons. ...
... Uranium, plutonium, oxygen, sodium, krypton, carbon Isotopes: atoms of the same element with different numbers of neutrons i.e 1214C and 1213C each has 12 protons which is the Atomic number. The mass number varies. It is the sum of the protons and neutrons. ...
CHEMISTRY 120A FALL 2006
... ultra-violet regions of the spectrum), and then various laser-based pump-probe techniques for studying photo-dissociation and photo-chemistry in general. Quantum mechanical perturbation theory will be applied to Nuclear Magnetic Resonance (NMR) spectroscopy, showing how the characteristic multiplet ...
... ultra-violet regions of the spectrum), and then various laser-based pump-probe techniques for studying photo-dissociation and photo-chemistry in general. Quantum mechanical perturbation theory will be applied to Nuclear Magnetic Resonance (NMR) spectroscopy, showing how the characteristic multiplet ...
the original file
... 1. how to draw resonance structures 2. meaning of conjugated vs isolated pi bonds 3. what an orbital is 4. be able to draw MO diagrams for allyl radical and cation and benzene, such as the one in Fig. 10.2 but you dont need to know how the MOs look, just the relative energy levels and how to put in ...
... 1. how to draw resonance structures 2. meaning of conjugated vs isolated pi bonds 3. what an orbital is 4. be able to draw MO diagrams for allyl radical and cation and benzene, such as the one in Fig. 10.2 but you dont need to know how the MOs look, just the relative energy levels and how to put in ...
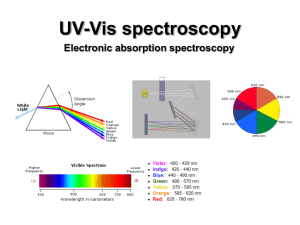
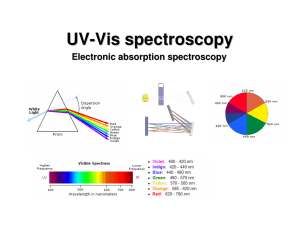
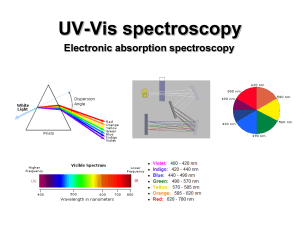
UV-Vis (electronic) spectroscopy
... electronic excitation (10-16 s). Since the nuclei do not move during the excitation, the internuclear distances remain constant and “the most probable component of an electronic transition involves only the vertical transitions”. ...
... electronic excitation (10-16 s). Since the nuclei do not move during the excitation, the internuclear distances remain constant and “the most probable component of an electronic transition involves only the vertical transitions”. ...
Over 99% of the known mass of the universe is composed of two
... of the nucleon to allow comparison with theory. Only then can we obtain a quantitative understanding of the nucleon’s substructure, ultimately providing a description of atomic nuclei from first principles. Nucleon spin structure is characterized by two dimensionless functions known as g1 and g2 . P ...
... of the nucleon to allow comparison with theory. Only then can we obtain a quantitative understanding of the nucleon’s substructure, ultimately providing a description of atomic nuclei from first principles. Nucleon spin structure is characterized by two dimensionless functions known as g1 and g2 . P ...
Nuclear Fission and Fusion Notes
... for ¾ of the sample to decay? c. Ra 222 has a half life of 3.82days. How long does it take for 15/16 of the sample to decay? ...
... for ¾ of the sample to decay? c. Ra 222 has a half life of 3.82days. How long does it take for 15/16 of the sample to decay? ...
IB-ATOMIC-AND-NUCLEAR-PHYSICS-DEFINITIONS
... NUCLEON: The particles in the nucleus (proton or neutron). NUCLEON NUMBER, A: The number of nucleons in the nucleus. PROTON NUMBER, Z: The number of protons in the nucleus. NEUTRON NUMBER, N: N=A-Z RADIOACTIVE DECAY: A random and spontaneous process in which unstable nuclei emit a particle (disinteg ...
... NUCLEON: The particles in the nucleus (proton or neutron). NUCLEON NUMBER, A: The number of nucleons in the nucleus. PROTON NUMBER, Z: The number of protons in the nucleus. NEUTRON NUMBER, N: N=A-Z RADIOACTIVE DECAY: A random and spontaneous process in which unstable nuclei emit a particle (disinteg ...
Muhammad Danish - Chemistry Department
... class of organotin compounds as they show different sets of 1H and 13C-NMR signals for Rgroups attached to Sn(IV) and only one set of signals for carboxylate ligand. A similar dichotomy is observed 119Sn-NMR spectra1,2. Two types of signals are only possible if there are two environments for the R-g ...
... class of organotin compounds as they show different sets of 1H and 13C-NMR signals for Rgroups attached to Sn(IV) and only one set of signals for carboxylate ligand. A similar dichotomy is observed 119Sn-NMR spectra1,2. Two types of signals are only possible if there are two environments for the R-g ...