Specific features of stainless steel compression elements
... of various types and qualities, whose resistance to corrosion is obtained with at least 10.5 percent of chromium and no more than 1.2 percent of carbon. The austenitic and duplex types of steel are most widely used in construction industry. Austenitic stainless steels contain iron, 16-28 percent of ...
... of various types and qualities, whose resistance to corrosion is obtained with at least 10.5 percent of chromium and no more than 1.2 percent of carbon. The austenitic and duplex types of steel are most widely used in construction industry. Austenitic stainless steels contain iron, 16-28 percent of ...
File
... good conductors of heat and electricity? • Valence e- in a metal are shared by all the metal atoms and can move freely, thus good conductors • e- absorb light and are ...
... good conductors of heat and electricity? • Valence e- in a metal are shared by all the metal atoms and can move freely, thus good conductors • e- absorb light and are ...
General - Department of Mechanical Engineering
... strength properties, however the ductility decreases. Ductility and strength may increase or decrease at temperatures above room temperature. This change depends on many factors, such as temperature and time of exposure. Materials shall be compatible with the thermal environment to which they are ...
... strength properties, however the ductility decreases. Ductility and strength may increase or decrease at temperatures above room temperature. This change depends on many factors, such as temperature and time of exposure. Materials shall be compatible with the thermal environment to which they are ...
A review on shape memory alloys with applications to morphing
... McFarland 1995, Brocca et al 2002). In stress-free conditions, some phase transformation temperatures (also called ‘critical temperatures’) can be identified as Ms, Mf, As, Af (M: martensite, A: austenite, s: start, f: final). For most SMAs, it results that Mf < Ms < As < Af. By varying the temperat ...
... McFarland 1995, Brocca et al 2002). In stress-free conditions, some phase transformation temperatures (also called ‘critical temperatures’) can be identified as Ms, Mf, As, Af (M: martensite, A: austenite, s: start, f: final). For most SMAs, it results that Mf < Ms < As < Af. By varying the temperat ...
Composite Materials
... expansion coefficient than metals Their thermal and electrical conductivities are lower than metals In addition MMCs have very high thermal deformation resistance which is especially important for spacecrafts • TDR = thermal conductivity / thermal expansion coefficient • Example – Magnesium has very ...
... expansion coefficient than metals Their thermal and electrical conductivities are lower than metals In addition MMCs have very high thermal deformation resistance which is especially important for spacecrafts • TDR = thermal conductivity / thermal expansion coefficient • Example – Magnesium has very ...
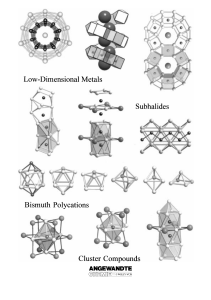
From the Metal to the Molecule
... intermetallic phases with bismuth. Previous studies suggest that this is a basic requirement for the formation of the corresponding subhalides. Consequently, we can speak of the partial oxidation of intermetallic phases. 2) The electronegativities of these transition metal elements hardly differ fro ...
... intermetallic phases with bismuth. Previous studies suggest that this is a basic requirement for the formation of the corresponding subhalides. Consequently, we can speak of the partial oxidation of intermetallic phases. 2) The electronegativities of these transition metal elements hardly differ fro ...
Formation Mechanism of Non-Metallic Inclusions in
... to compute the activity of elements in the liquid steel. This is based on Wagner’s formalism but it is more accurate for the steel baths where solutes are not at infinite dilution. This is the reason why Janke’s formalism is better than the Wagner’s one in the study of stainless steels, as they are ...
... to compute the activity of elements in the liquid steel. This is based on Wagner’s formalism but it is more accurate for the steel baths where solutes are not at infinite dilution. This is the reason why Janke’s formalism is better than the Wagner’s one in the study of stainless steels, as they are ...
The d- and f- Block Element Block Elements The d- and f
... of the groups 3-12 in which the d orbitals are progressively filled in each of the four long periods. The elements constituting the f -block are those in which the 4 f and 5 f orbitals are progressively filled in the latter two long periods; these elements are formal members of group 3 from which th ...
... of the groups 3-12 in which the d orbitals are progressively filled in each of the four long periods. The elements constituting the f -block are those in which the 4 f and 5 f orbitals are progressively filled in the latter two long periods; these elements are formal members of group 3 from which th ...
CHROMIUM
... Chromium is a steel-grey lustrous metal which can take a high polish. It is relatively hard but malleable and many of its applications are due to its excellent corrosion resistance. Chromium becomes rapidly passivated by oxygen when exposed to air. This stable oxide layer has a spinel structure ...
... Chromium is a steel-grey lustrous metal which can take a high polish. It is relatively hard but malleable and many of its applications are due to its excellent corrosion resistance. Chromium becomes rapidly passivated by oxygen when exposed to air. This stable oxide layer has a spinel structure ...
Ni recovery using KOH, NaOH, and NH4OH in the presence of
... During production of the platinum group metals (PGMs) in South Africa, base metals are leached first. A significant amount of iron is also co-leached, which in turn is removed by precipitation as ammonium jarosite. Substantial amounts of nickel have been found entrained in this precipitate, which is ...
... During production of the platinum group metals (PGMs) in South Africa, base metals are leached first. A significant amount of iron is also co-leached, which in turn is removed by precipitation as ammonium jarosite. Substantial amounts of nickel have been found entrained in this precipitate, which is ...
s-BLOCK ELEMENTS - einstein classes
... Ionic salt-like hydrides formed at high temperature by Ca, Sr and Ba. ...
... Ionic salt-like hydrides formed at high temperature by Ca, Sr and Ba. ...
Materials - Hodder Education
... compression but weak in tension. This means it is very hard to change their shape, but they are easily snapped. They are electrical and thermal insulators and have very high melting points. They are chemically unreactive and do not react with oxygen, water or acids. Ceramics are crystalline compound ...
... compression but weak in tension. This means it is very hard to change their shape, but they are easily snapped. They are electrical and thermal insulators and have very high melting points. They are chemically unreactive and do not react with oxygen, water or acids. Ceramics are crystalline compound ...
Chemical Equations
... C6H12O6 + O2 H2O + CO2 Start with either C or H. Oxygen appears in every formula; save it till last. ...
... C6H12O6 + O2 H2O + CO2 Start with either C or H. Oxygen appears in every formula; save it till last. ...
C5 Chemicals of the Natural Environment SOW
... The crystal structure of metals is not obvious and the great diversity in properties of metals can make it difficult to see that one theory can explain all the variations. Poor diagrams can give a picture of the metallic bond that bears no relation to the incompressible nature of metals. ...
... The crystal structure of metals is not obvious and the great diversity in properties of metals can make it difficult to see that one theory can explain all the variations. Poor diagrams can give a picture of the metallic bond that bears no relation to the incompressible nature of metals. ...
FABRICATION OF A DISORDERED AND NANO
... alloying has also been successfully utilized in order to extend the solid solubilities in many commercially important alloy systems. Many intermetallic compounds which have high melting points and are difficult to elaborate by conventional processing techniques, could be simply synthesized with a ho ...
... alloying has also been successfully utilized in order to extend the solid solubilities in many commercially important alloy systems. Many intermetallic compounds which have high melting points and are difficult to elaborate by conventional processing techniques, could be simply synthesized with a ho ...
Chapter 6: Mechanical Properties
... • Large hardness means: --resistance to plastic deformation or cracking in compression. --better wear properties. apply known force ...
... • Large hardness means: --resistance to plastic deformation or cracking in compression. --better wear properties. apply known force ...
Chemistry Revision Checklist F4 2017 (inc F3)
... State the type of compound present, given a chemical name ending in -ane, -ene, -ol, or -oic acid or a molecular structure Describe the concept of homologous series as a ‘family’ of similar compounds with similar chemical properties due to the presence of the same functional group Describe the gener ...
... State the type of compound present, given a chemical name ending in -ane, -ene, -ol, or -oic acid or a molecular structure Describe the concept of homologous series as a ‘family’ of similar compounds with similar chemical properties due to the presence of the same functional group Describe the gener ...
Interfacial Reaction between Alumina and Cu
... shown in Fig. 2. The chemical composition in at.-% corresponding to each spectrum is 4.2 Cu, 49.0 Ti, 0.8 Al, 46.0 O for phase 1, and 27.5 Cu, 28.2 Ti, 5.5 Al, 38.8 O for phase 2. From the chemical analyses, phase 1 is TiO with a small amount of copper and aluminum, and phase 2 is a complex oxide. T ...
... shown in Fig. 2. The chemical composition in at.-% corresponding to each spectrum is 4.2 Cu, 49.0 Ti, 0.8 Al, 46.0 O for phase 1, and 27.5 Cu, 28.2 Ti, 5.5 Al, 38.8 O for phase 2. From the chemical analyses, phase 1 is TiO with a small amount of copper and aluminum, and phase 2 is a complex oxide. T ...
TOPIC 12. THE ELEMENTS
... may interact with existing clouds of gas, ice and dust to eventually form new stars and planets such as our solar system. Our sun was not one of the original stars in the universe but is probably a second or third generation star, formed in part from the energy and residues released by previous supe ...
... may interact with existing clouds of gas, ice and dust to eventually form new stars and planets such as our solar system. Our sun was not one of the original stars in the universe but is probably a second or third generation star, formed in part from the energy and residues released by previous supe ...
Mg-based compounds for hydrogen and energy storage
... hydrogen molecules into adsorbed atomic species and vice versa. It is not necessary to have pure Ni in the system, and Mg–Ni alloys also show these enhanced sorption kinetics [26–28]. For instance, enhanced hydrogen sorption rates, while retaining a high storage capacity of about 6 wt% hydrogen, wer ...
... hydrogen molecules into adsorbed atomic species and vice versa. It is not necessary to have pure Ni in the system, and Mg–Ni alloys also show these enhanced sorption kinetics [26–28]. For instance, enhanced hydrogen sorption rates, while retaining a high storage capacity of about 6 wt% hydrogen, wer ...
Ag−Ni alloy nanoparticles for electrocatalytic reduction of benzyl
... The electrocatalytic activity of the as-prepared Ag−Ni NPs was tested for the reduction of PhCH2Cl in CH3CN+(C2H5)4NBF4 solution. Figure 3(a) shows the CVs of PhCH2Cl on bare GC electrode, 5Ag1Ni, pure Ag and Ni electrodes at 50 mV/s. The Ni electrode shows no reduction peak which is so far consider ...
... The electrocatalytic activity of the as-prepared Ag−Ni NPs was tested for the reduction of PhCH2Cl in CH3CN+(C2H5)4NBF4 solution. Figure 3(a) shows the CVs of PhCH2Cl on bare GC electrode, 5Ag1Ni, pure Ag and Ni electrodes at 50 mV/s. The Ni electrode shows no reduction peak which is so far consider ...
Subject Materials for Chemistry
... broken glasses are add to the raw materials and then heated to 14000C (1673K) in the furnace. Cullet lowers the melting point of the batch. The following chemical reactions takes place to form molten glass, over molten glass scum is formed. The scum is skimmed off. ...
... broken glasses are add to the raw materials and then heated to 14000C (1673K) in the furnace. Cullet lowers the melting point of the batch. The following chemical reactions takes place to form molten glass, over molten glass scum is formed. The scum is skimmed off. ...
Physical Properties of the NbC Carbide
... metallic and ionic character. This fact explains the observed physical properties: high hardness and high melting points are properties typical of solids with ionic or covalent bonding, while thermal and electrical conductivity are characteristics of metallic bonding. The electronic structure of the ...
... metallic and ionic character. This fact explains the observed physical properties: high hardness and high melting points are properties typical of solids with ionic or covalent bonding, while thermal and electrical conductivity are characteristics of metallic bonding. The electronic structure of the ...
Granta Design • CES Edupack 2009 • Durability - CORE
... Calcium bicarbonate (Steel, Cast Iron), polyphosphate (Cu, Zn, Al, Fe), calcium hydroxide (Cu, Zn, Fe), sodium silicate (Cu, Zn, Fe), sodium chromate (Cu, Zn, Pb, Fe), potassium dichromate (Mg), sodium nitrite (Monel), benzoic acid (Fe), calcium and zinc metaphosphates (Zn). ...
... Calcium bicarbonate (Steel, Cast Iron), polyphosphate (Cu, Zn, Al, Fe), calcium hydroxide (Cu, Zn, Fe), sodium silicate (Cu, Zn, Fe), sodium chromate (Cu, Zn, Pb, Fe), potassium dichromate (Mg), sodium nitrite (Monel), benzoic acid (Fe), calcium and zinc metaphosphates (Zn). ...
Alloy

An alloy is a mixture of metals or a mixture of a metal and another element. Alloys are defined by metallic bonding character. An alloy may be a solid solution of metal elements (a single phase) or a mixture of metallic phases (two or more solutions). Intermetallic compounds are alloys with a defined stoichiometry and crystal structure. Zintl phases are also sometimes considered alloys depending on bond types (see also: Van Arkel-Ketelaar triangle for information on classifying bonding in binary compounds).Alloys are used in a wide variety of applications. In some cases, a combination of metals may reduce the overall cost of the material while preserving important properties. In other cases, the combination of metals imparts synergistic properties to the constituent metal elements such as corrosion resistance or mechanical strength. Examples of alloys are steel, solder, brass, pewter, duralumin, phosphor bronze and amalgams.The alloy constituents are usually measured by mass. Alloys are usually classified as substitutional or interstitial alloys, depending on the atomic arrangement that forms the alloy. They can be further classified as homogeneous (consisting of a single phase), or heterogeneous (consisting of two or more phases) or intermetallic.